Summary
-
1.
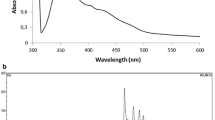
Cells of the extremely halophilic bacteria, Halobacterium cuti-rubrum and H. halobium grown in a chemically defined medium (BSMK) were red due to the presence of bacterioruberins (maxima, 370, 388, 494 and 527 nm). Adding 0.1% glycerol to BSMK stimulated growth, but cells rapidly lost bacterioruberins becoming greyish purple in the stationary phase. Acetone extracts of these cells were yellow with a broad absorption band at 360–390 nm, partly attributable to retinal. In BSMK medium with or without glucose, the bacterioruberin concentration increased until maximal growth was reached, then fell rapidly.
-
2.
In complex medium (CM) cells formed less bacterioruberins than in BSMK. Adding 0.1% glycerol to CM stimulated growth but did not change pigmentation; adding glucose only slightly stimulated growth but greatly increased bacterioruberin formation. Exposure to visible light did not affect growth or pigmentation.
-
3.
Addition of glycerol or glucose to BSMK increased the formation (relative to squalene) of dihydrosqualene, tetrahydrosqualene, and vitamin MK8. Higher levels of these compounds were found in cells grown in CM than in BSMK. Though glycerol decreased the formation of bacterioruberins it increased the formation of β-carotenes. Glucose increased the formation both of bacterioruberins and β-carotene. A preliminary hypothesis to account for the effects of nutrients on pigmentation is presented.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blaurock, A. E., Stoeckenius, W.: Structure of the purple membrane. Nature New Biol. 233, 152–155 (1971).
Brown, R. H., Stevenson, J.: Variation in the composition of a purified membrane fraction from Halobacterium salinarium with the phase of growth. Antonie v. Leeuwenhoek 37, 89–99 (1971).
Gochnauer, M. B., Kushner, D. J.: Growth and nutrition of extremely halophilic bacteria. Canad. J. Microbiol. 15, 1157–1165 (1969).
Hubbard, R., Bownds, D., Yoshizawa, T.: The chemistry of visual photoreception. Cold Spring Harb. Symp. quant. Biol. 30, 301–315 (1965).
Jensen, S. L.: The constitution of some bacterial carotenoids and their bearings on biosynthetic problems, p 157. Trondheim: Bruns 1962.
Kelly, M., Norgard, S., Liaaen-Jensen, S.: Bacterial carotenoids. XXXI. C50-carotenoids. 5. Carotenoids of Halobacterium salinarium, especially bacterioruberin. Acta chem. scand. 24, 2169–2182 (1970)
Kushwaha, S. C., Pugh, E. L., Kramer, J. K. G., Kates, M.: Isolation and identification of dehydrosqualene and C40-carotenoid pigments in Halobacterium cutirubrum. Biochim. biophys. Acta (Amst.) 260, 492–506 (1972).
Lowry, O. H., Rosebrough, N. J., Farr, S. N., Randall, R. J.: Protein measurement with the folin phenol reagent. J. biol. Chem. 193, 265–275 (1951).
Nandy, S. C., Sen, S. N.: Effect of environment of growth and carotenogenesis of Sarcina litoralis. Canad. J. Microbiol. 9, 601–611 (1963).
Oesterhelt, D., Stoeckenius, W.: Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nature New Biol. 233, 149–152 (1971).
Sehgal, S., Gibbons, N. E.: Effect of some metal ions on the growth of Halobacterium cutirubrum. Canad. J. Microbiol. 6, 165–169 (1960).
Tornabene, T. G., Kates, M., Gelpi, E., Oro, J.: Occurrence of squalene, di- and tetrahydrosqualene and vitamin MK8 in an extremely halophilic bacterium, Halobacterium cutirubrum. J. Lipid Res. 10, 294–303 (1969).
Vogel, A. I.: Practical organic chemistry, pp. 161–175 London: Longmans, Green 1956.
Wald, G., Brown, P. K.: Synthesis and bleaching of rhodopsin. Nature (Lond.) 177, 174–176 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gochnauer, M.B., Kushwaha, S.C., Kates, M. et al. Nutritional control of pigment and isoprenoid compound formation in extremely halophilic bacteria. Archiv. Mikrobiol. 84, 339–349 (1972). https://doi.org/10.1007/BF00409082
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00409082