Abstract
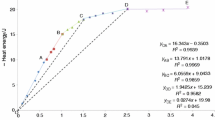
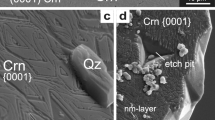
Hydrothermal syntheses were made mainly in the binary system SiO2-H2O in a temperature range between 300 ° C and 500 ° C and pressures from 0.2 kbar up to 4.0 kbar with various starting materials. In this way the transformation behavior of different amorphous silicas via cristobalite and keatite to quartz were observed. This behavior depends mainly on the parameters: pressure, temperature, run duration and state of the starting material. Four reaction paths have been observed: in most experiments the complete reaction sequence “amorphous silica→cristobalite→keatite→quartz” took place. Less often the reactions: “amorphous silica→cristobalite→quartz” and: “amorphous silica→keatite→quarts” were observed. Very few samples were found with a direct transition of amorphous silica into quartz at high pressures. A kinetic model is given in form of a pressure-temperature-time diagram of the system SiO2-H2O under hydrothermal conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alexander, G.B., Heston, W.M., Iler, H.K.: The solubility of amorphous silica in water. J. Phys. Chem. 58, 453–455 (1954)
Bettermann, P.: Petrographische und betontechnologische Untersuchungen zur Alkaliempfindlichkeit von Geschiebefeuerstein aus Schleswig-Holstein. Diplomarbeit Universität Kiel, p. 245, Kiel 1972
Bettermann, P., Ackermand, D.: Comparison flint-chert, based on chemical data. (abstract in: collected abstracts, 9th gen. meet., IMA, 1974) in press: J. Sed. Petrol. 1975
Bricker, O.P.: Stability constants and Gibbs free energies of formation of magadiite and kenyaite. Am. Mineralogist 54, 1026–1033 (1969)
Brousse, R.: Minéralogie et Pétrographie des roches volcaniques du massif du Mont Dore (auvergne). Bull. Soc. Franc. Mineral. 84, 131–186 (1961)
Campbell, A.S., Fyfe, W.S.: Hydroxyl-ion catalysis of the hydrothermal crystallization of amorphous silica, a possible high-temperature pH-indicator. Am. Mineralogist 45, 464–468 (1960)
Carr, R.M., Fyfe, W.S.: Some observations on the crystallization of amorphous silica. Am. Mineralogist 43, 908–916 (1958)
Correns, C.W.: Beiträge zur Petrographie und Genesis der Lydite, (Kieselschiefer). Mitt. preuss. geol. Landesanstalt, 18–36, Berlin 1924
Correns, C.W.: Zur Geochemie der Diagenese, 1. Das Verhalten von CaCO3 und SiO2. Geochim. Cosmochim. Acta, 1, 49–54 (1950)
Endell, J.: Röntgenographischer Nachweis kristalliner Zwischenzustände bei der Bildung von Cristobalit aus Kieselgur beim Erhitzen. Kolloid Z. 111, 19–22 (1948)
Ernst, W.G., Calvert, S.E.: An experimental study of the recrystallization of porcelanite and its bearing on the origin of some bedded chert. Am. J. Sci. 269A (Schairer-Volume), 114–133 (1969)
Eugster, H.P.: Hydrous sodium silicates from Lake Magadi, Kenya: precursors of bedded chert. Science 157, 1177–1180 (1967)
Eugster, H.P.: Inorganic bedded cherts from the Magadi area, Kenya. Contrib. Mineral. Petrol. 22, 1–31 (1969)
Flörke, O.W.: Strukturanomalien bei Tridymit und Cristobalit. Ber. Dt. Keram. Ges. 369–381 (1955a)
Flörke, O.W.: Zur Frage des “Hoch”-cristobalit in Opalen, Bentoniten und Gläsern. Neues Jahrb. Mineral. Monatsh. 217–223 (1955b)
Flörke, O.W.: Die Kristallarten des SiO2 und ihr Umwandlungsverhalten. Ber. Dt. Keram. Ges. 38, 89–97 (1961)
Flörke, O.W.: Untersuchungen an amorphem und mikrokristallinem SiO2. Chem. Erde 22, 91–110 (1962)
Flörke, O.W.: Die Modifikationen des SiO2. Fortschr. Mineral. 44, 181–230 (1967)
Flörke, O.W.: Transportation and deposition of SiO2 with H2O under supercritical conditions. Kristall und Technik 7, 159–166 (1972)
Flörke, O.W., Jones, J.B., Schmincke, H.U.: Z. Krist. Laves-Festschrift, in press. (1975)
Flörke, O.W., Jones, J.B., Segnit, E.R.: Opal-CT crystals. Neues Jahrb. Mineral. Mh. '75.8, 369–377 (1975)
Fyfe, W.S., McKay, D.S.: Hydroxyl ion catalysis of the crystallization of amorphous silica at 330 ° C and some observations on the hydrolysis of albite solutions. Am. Mineral. 47, 83–89 (1962)
Greenwood, R.: Thermal behavior of SiO2-X and its relation to the natural silica minerals. Am. Mineral. 52, 1662–1668 (1967)
Hamilton, D.L., Henderson, C.M.B.: The preparation of silicate compositions by a gelling method. Mineral. Mag. 36, 832–838 (1968)
Harder, H.: Experimente zur “Ausfällung” der Kieselsäure. Geochim. Cosmochim. Acta 29, 429–444 (1965)
Harder, H., Flehming, W.: Bildung von Quarz aus verdünnten Lösungen bei niedrigen Temperaturen. Naturwissenschaften 54, 140 (1967)
Harder, H., Flehming, W.: Quarzsynthese bei tiefen Temperaturen. Naturwissenschaften 295–305 (1969)
Harder, H., Menschel, G.: Quarzbildungen am Meeresboden. Naturwissenschaften 54, 561 (1967)
Heydemann, A.: Untersuchungen über die Bildungsbedingungen von Quarz im Temperaturbereich zwischen 100 ° C und 250 ° C. Beitr. Mineral Petrog. 10, 242–259 (1964)
Hofmann, U., Rothe, A.: Über die Veränderung des Quarzes beim trockenen Mahlen. Z. Anorg. Allgem. Chem. 357, 196–201 (1968)
Jones, J.B., Segnit, E.R.: The nature of opal. I. Nomenclature and constituent phases. J. Geol. Soc. Australia 18, 57–68 (1971)
Kameyama, T., Naka, S.: Effect of water on transformation of amorphous silica to coesite. J. Am. Ceram. Soc. 57, 499 (1974)
Keat, P.P.: A new crystalline silica. Science 120, 328–330 (1954)
Keat, P.P.: The transformation of silicic acid to quartz and the synthesis of a new crystalline silica. Ph. D., Thesis, Rutgers University, New Jersey 1956
Konnert, J.H., Karle, J., Ferguson, G.A.: Crystalline ordering in silica and germania glasses. Science 179, 177–178 (1973)
Li, C.T.: Transformation mechanism between high-quartz and keatite phases of LiAlSi2O6 composition. Acta Cryst. B 27, 1132–1140 (1971)
Li, C.T.: The role of lithium in stabilizing some high-temperature silica phases, Z. Krist. 138, 216–236 (1973)
Luth, W.C., Tuttle, O.F.: Externally heated cold-seal pressure vessels for use to 10000 bars and 750 ° C. Am. Mineral. 48, 1401–1403 (1963)
Mackenzie, F.T., Gees, R.: Quartz: Synthesis at earth-surface conditions. Science 173, 533–535 (1971)
Maglione, G., Servant, M.: Signification des silicates de sodium et des cherts néoformés dans les variations hydrologiques et climatiques holocènes du bassin tschadien. Compt. Rend. 277, 1721–1724 (1973)
Mizutani, S.: Transformation of silica under hydrothermal conditions. J. Earth Sci. [Nagova Univ.] 14, 56–88 (1966)
Mosebach, R.: Die Löslichkeit der gefällten Kieselsäure und des Kieselglases in Wasser bei höheren Temperaturen und Drucken. Neues Jahrb. Mineral., Abhandl. 89, 1–26 (1956)
Mosebach, R.: Theromodynamic behavior of quartz and other forms of silica in pure water at elevated temperatures and pressures with conclusion on their mechanism of solution. J. Geol. 65, 347–363 (1957)
Nagasima, N.: Structure analysis of silicon dioxide films formed by oxidation of silane. J. Appl. Phys. 43, 3378–3386 (1972)
Nagelschmidt, G., Gordon, R.L., Griffin, O.G.: Surface of finely-ground silica. Nature 169, 539 (1952)
Okamoto, G., Okura, T., Goto, T.: Properties of silica in water. Geochim. Acta 12, 123–132 (1957)
Olesch, M.: Synthese und Stabilität trioktaedrischer Calciumglimmer (Clintonit, Xanthophyllit, Brandisit). Dissertation Ruhr Universität, p. 197, Bochum 1973
Robarick, E.: Synthese und kristallographische Charakterisierung von SiO2-X. Diplomarbeit, Ruhr-universität, p. 67, Bochum 1974
Schrader, R. Wissing, R., Kubsch, H.: Zur Oberflächenchemie von mechanisch aktiviertem Quarz. Z. Anorg. Allgem. Chem. 365, 191–198 (1969)
Shropshire, J., Keat, P.P., Vaughan, P.A.: The crystal structure of keatite, a new form of silica. Z. Krist. 112, 400–413 (1959)
Sievers, R.: Silica solubility, 0–200 ° C, and the diagenesis of siliceous sediments. J. Geol. 70, 127–150 (1962)
Siffert, B., Wey, R.: Etude quantitative de la transformation de la silice en quartz entre 230 ° et 350 ° C en présence de bases alcalines. Silicates Ind. 32, 415–421 (1967)
Simon, I.: Infra-red studies of glass. In: Modern aspects of the vitreous state, (J.D. Mackenzie, ed.), p. 120–151. London: Butterworth 1960
Simon, I., McMahon, H.O.: Study of the structure of quartz, cristobalite and vitreous silica by reflection in infrared. J. Chem. Phys. 21, 23–30 (1953)
Skinner, B.J., Appleman, D.E.: Melanophlogite, a cubic polymorph of silica. Am. Mineral. 48, 854–867 (1963)
Tarr, W.A.: The origin of chert and flint. Univ. Missouri Studies, 1, (2), 1926
Tröger, W.E.: Optische Bestimmung der gesteinsbildenden Minerale. Teil 2 Textband, 2. Auflage, p. 822, Stuttgart: Nägele und Obermiller 1969
Tuttle, O.F.: Two pressure vessels for silicate-water studies. Geol. Soc. Am. Bull. 60, 1727–1729 (1949)
Wetzel, W.: Sedimentpetrographische Studien I. Feuerstein. Neues Jahrb. Mineral. 47, 39–78 (1923)
Wetzel, W.: Die Entstehungsgeschichte verschiedener Arten von Kreidefeuersteinen, Z. D. DGG, Bd. 89 (1973)
Wetzel, W., Wetzel, O., Deflandre, G.: Die Feuersteine der Kreidezeit (Kieselsäure als Versteinerungsmittel). Umschau 45, 275–279 (1941)
Wetzel, W.: Selektive Verkieselung. N. Jb. Paläont., Abh. 105, 1–10 (1957)
Wey, R., Siffert, B.: Quelques observations quantitatives sur la transformation de la silice amorphe en quartz et cristobalite. Silicates Industriels 9, 419–424 (1961)
Wilson, M.J., Russell, J.D., Tait, J.M.: A new interpretation of the structure of disordered α-cristobalite. Contrib. Mineral. Petrol. 47, 1–6 (1974)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bettermann, P., Liebau, F. The transformation of amorphous silica to crystalline silica under hydrothermal conditions. Contr. Mineral. and Petrol. 53, 25–36 (1975). https://doi.org/10.1007/BF00402452
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00402452