Abstract
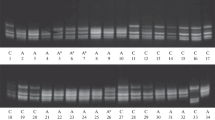
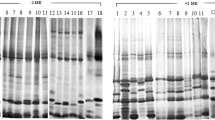
The molecular forms of β-amylase present in developing, mature, germinating and malted grains of barley (Hordeum vulgare L.), and in vegetative tissues, have been studied using Western-blot analyses and isoelectric focusing of isoenzymes. Five isoforms with different relative molecular masses (Mrs) could be recognised. The major isoform present in the mature grain, called isoform B, had an Mr of about 60 000. This was converted on malting or germination to two lower-Mr forms called C and D. Previous work (R. Lundgard and B. Svensson, 1986, Carlsberg Res. Commun. 51, 487–491) has shown that these result from partial proteolysis of isoform B. Isoenzyme analyses showed complex patterns of bands, with pIs between about 5.0 and 6.0. Two allelic types were present in the eight lines. A number of new bands with a range of pIs appeared during germination and malting.
An isoform with the same Mr as D and a minor low-Mr isoform (E) were present in young developing whole caryopses (8–12 d after anthesis), but not in older developing endosperms (14–21 d after anthesis). Isoenzyme analyses also showed different patterns of bands in these two tissues, while hybrid-dot analyses indicated the presence of separate populations of mRNAs. It is suggested that the early endosperm isoforms (D and E) are “green” β-amylases present in the pericarp and-or testa of the young caryopses.
Roots but not shoots or leaves also contained an isoform with the same Mr as D, although the pattern of isoenzymes differed from that present in the seed tissues.
The fifth isoform, A, was a diffuse high-Mr form present in small amounts in all seed and vegetative tissues, and may correspond to a constitutively expressed form.
These multiple molecular forms of β-amylase are discussed in relation to the recent report that β-amylase is encoded by two structural loci, with a total copy number of two to three per haploid genome (Kreis et al, 1988, Genet. Res. Camb. 51, 13–16).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Mr :
-
relative molecular mass
- SDS-PAGE:
-
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
References
Ainsworth, C.C., Gale, M.D., Baird, S. (1983) The genetics of β-amylase isozymes in wheat. Theor. Appl. Genet. 66, 39–49
Ainsworth, C.C., Miller, T.E., Gale, M.D. (1987) α-Amylase and β-amylase homoeoloci in species related to wheat. Genet. Res. Camb. 49, 93–103
Allison, M.J., Ellis, R.P. (1973) The inheritance of β-amylases in developing barley grain. Biochem. Genet. 10, 165–173
Briggs, D.E. (1973) Hormones and carbohydrate metabolism in germinating cereal grains. In: Biosynthesis and its control in plants, pp. 219–277, Milborrow, B.V., ed. Academic Press, London New York
Bunce, N., White, R.P., Shewry, P.R. (1985) Variation in estimates of molecular weights of cereal prolamins by SDS-PAGE. J. Cereal Sci. 3, 131–142
Daussant, J., Renard, H.A. (1987) Development of different α-amylase isozymes, having high and low isoelectric points, during early stages of kernel development in wheat. J Cereal Sci. 5, 13–21
Doll, H. (1980) A nearly non-functional mutant allele of the storage protein locus Hor-2 in barley. Hereditas 93, 217–222
Doll, H., Køie, B., Eggum, B.O. (1974) Induced high-lysine mutants in barley. Radiat. Bot. 14, 73–80
Festenstein, G.N., Hay, F.C., Shewry, P.R. (1987) Immunochemical relationships of the prolamin storage proteins of barley, wheat, rye and oats. Biochim. Biophys. Acta 912, 371–383
Gale, M.D., Law, C.N., Chojecki, A.J., Kempton, R.A. (1983) Genetic control of α-amylase production in wheat. Theor. Appl. Genet. 64, 309–316
Giese, H., Hejgaard, J. (1984) Synthesis of salt-soluble proteins in barley. Pulse-labelling study of grain filling in liquidcultured detached spikes. Planta 161, 172–177
Hejgaard, J. (1976) Free and protein-bound β-amylases of barley grain. Characterization by two-dimensional immunoelectrophoresis. Physiol. Plant. 38, 293–299
Hejgaard, J. (1978) ‘Free’ and ‘bound’ β-amylases during malting of barley. Characterization by two-dimensional immunoelectrophoresis. J. Inst. Brew. 84, 43–46
Hejgaard, J., Carlsen, S. (1977) Immunoelectrophoretic identification of a heterodimer β-amylase in extracts of barley grain. J. Sci. Food Agric. 28, 900–904
Ingversen, J., Køie, B., Doll, H. (1973) Induced seed protein mutant of barley. Experinetia 29, 1151–1152
Jonassen, I., Ingversen, J., Brandt, A. (1981) Synthesis of SP11 albumin, β-amylase and chymotrypsin inhibitor CI-1 on polysomes from the endoplasmic reticulum of barley endosperm. Carlsberg Res. Commun. 46, 175–181
Karlsson, K.E. (1976) Linkage studies of the lys gene in relation to some marker genes and translocations. In: Barley genetics III, pp. 536–541, Gaul, H., ed. Verlag Karl Thieming, München
Kawazu, T., Nakanishi, Y., Uozumi, N., Sasaki, T., Yamagata, H., Tsukagoshi, N., Udaka, S. (1987) Cloning andnucleotide sequence of the gene coding for enzymatically active fragments of the Bacillus polymyxa β-amylase. J. Bacteriol. 169, 1564–1570
Kreis, M., Williamson, M.S., Buxton, B., Pywell, J., Hejgaard, J., Svendsen, I. (1987) Primary structure and differential expression of β-amylase in normal and mutant barley. Eur. J. Biochem. 169, 517–525
Kreis, M., Williamson, M.S., Shewry, P.R., Sharp, P., Gale, M. (1988) Identification of a second locus encoding β-amylase on chromosome 2 of barley. Genet. Res. Camb 51, 13–16
Kruger, N.J., Hammond, J.B.W. (1988) Immunodetection of protein on “Western” blots using 125I-labeled protein A. In: Methods in molecular biology, vol. 3, Walker, J.M., ed. The Humana Press, Clifton, N. J., USA. In press
LaBerge, D.E., Marchylo, B.A. (1983) Heterogeneity of the β-amylase enzymes of barley. J. Am. Soc. Brew. Chem. 41, 120–124
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of heads of the bacteriophage T4. Nature 227, 681–685
Levi, C., Preiss, J. (1978) Amylopectin degradation in pea chloroplast extracts. Plant Physiol. 61, 218–220
Lundgard, R., Svensson, B. (1986) Limited proteolysis in the carboxy-terminal region of barley β-amylase. Carlsberg Res. Commun. 51, 487–491
Munck, L., Karlsson, K.E., Hagberg, A., Eggum, B.O. (1970) Gene for improved nutritional value in barley seed protein. Science 168, 985–987
Nummi, M., Vilhunen, R., Enari, T.-M., (1965) β-Amylase: I. β-Amylases of different molecular size in barley and malt. Eur. Brew. Conf. Proc. 10th Congr. Stockholm, pp. 51–61
Powling, A., Islam, A.K.M.R., Shepherd, K.W. (1981) Isozymes in wheat-barley hybrid derivative lines. Biochem. Genet. 19, 237–254
Rahman, S., Kreis, M., Forde, B.G., Shewry, P.R., Miflin, B.J. (1984) Hordein-gene expression during development of the barley (Hordeum vulgare) endosperm. Biochem. J. 223, 315–322
Rahman, S., Shewry, P.R., Miflin, B.J. (1982) Differential protein accumulation during barley grain development. J. Exp. Bot. 33, 717–728
Shewry, P.R., Franklin, J., Parmar, S., Smith, S.J., Miflin, B.J. (1983) The effects of sulphur starvation on the amino acid and protein compositions of barley grain. J. Cereal Sci. 1, 21–31
Shewry, P.R., Williamson, M.S., Kreis, M. (1987) Effects of mutant genes on the synthesis of storage components in developing barley endosperms. In: Developmental mutants in higher plants, pp. 97–118, Thomas, H. and Grierson, D., eds., Cambridge University Press, Cambridge, UK
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shewry, P.R., Parmar, S., Buxton, B. et al. Multiple molecular forms of β-amylase in seeds and vegetative tissues of barley. Planta 176, 127–134 (1988). https://doi.org/10.1007/BF00392488
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392488