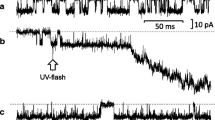
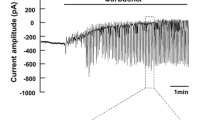
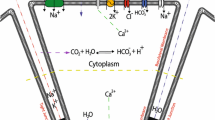
Abstract
Knowledge of luminal and basolateral acinar cell membrane areas of the secretory endpieces is a prerequisite for a detailed quantitative analysis of the ion transport involved in secretion of the primary saliva. In the present study, these areas were estimated in rat parotid acinar cells using standard stereological methods. A total of 480 micrographs — obtained by random sampling from eight glands from four rats — were analysed at a final magnification of 40000x. Expressed per unit cell volume, the area of the luminal acinar cell membrane was: 0.125 μm2 · μm−3 (SEM=0.027 μm2 · μm−3, n=4 animals) and the area of the basolateral membrane was: 1.54 μm2 · μm−3 (SEM=0.085 μm2 · μm−3, n=4 animals). These figures make it possible to perform a synthesis based upon different categories of experimental data, e.g. on ion fluxes, membrane potentials and single-channel conductances. Thus, we have estimated the density of open, low-conductance Cl− channels in the luminal membrane — which are not readily accessible for direct, patch-clamp analysis — to be approximately 18 channels per μm2 in the stimulated state.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berridge MJ, Oschman JL (1972) Transporting epithelia. Academic Press, New York
Bundgaard M, Frøkjær-Jensen J (1982) Functional aspects of the ultrastructure of terminal blood vessels: a quantitative study of consecutive segments of the frog mesenteric microvasculature. Microvasc Res 23:1–30
Bundgaard M, Zeuthen T (1982) Structure of Necturus gallbladder epithelium during transport at low external osmolarities. J Membr Biol 68:97–105
Cope GH, Williams MA (1973) Quantitative analyses of the constituent membranes of parotid acinar cells and of the changes evident after induced exocytosis. Z Zellforsch Mikrosk Anat 145:311–330
Fischer H, Kreusel KM, Illek B, Machen TE, Hegel U, Clauss W (1992) The outwardly rectifying Cl− channel is not involved in cAMP-mediated Cl− secretion in HT-29 cells: evidence for a very-low-conductance Cl− channel. Pflügers Arch 422:159–167
Gallacher DV, Morris AP (1986) A patch-clamp study of potassium currents in resting and acetylcholine-stimulated mouse submandibular acinar cells. J Physiol (Lond) 373:379–395
Gray MA, Harris A, Coleman L, Greenwel JR, Argent BE (1989) Two types of chloride channel on duct cells cultured from human fetal pancreas. Am J Physiol 257:C240-C251
Greger R, Schlatter E, Gögelein H (1985) Cl−-channels in the apical membrane of the rectal gland “induced” by cAMP. Pflügers Arch 403:446–448
Karnovsky MJ (1971) Use of ferrocyanide-reduced osmium tetroxide in electron microscopy. Proceedings of the 11th Annual Meeting of the American Society for Cell Biology, p 146a
Kunzelmann K, Kubitz R, Grolik M, Warth R, Greger R (1992) Small-conductance Cl−channels in HT29 cells: activation by Ca2+, hypotonic cell swelling and 8-Br-cGMP. Pflügers Arch 421:238–246
Lee RMKW (1984) A critical appraisal of the effects of fixation, dehydration and embedding on cell volume. In: Revel JP, Barnard T, Harris GH (eds) The science of biological specimen preparation. SEM Inc., AMF O'Hare, II 60666, pp 61–70
Lee SI, Turner RJ (1991) Mechanism of secretagogue-induced HCO −3 and Cl− loss from rat parotid acini. Am J Physiol 261:G111-G118
Martinez JR, Cassity N (1985) 36Cl fluxes in dispersed rat submandibular acini: effects of acetycholine and transport inhibitors. Pflügers Arch 403:50–54
Marty A, Tan YP, Trautmann A (1984) Three types of calcium-dependent channels in rat lacrimal glands. J Physiol (Lond) 357:293–325
Melvin JE, Kawaguchi M, Baum BJ, Turner RJ (1987) A muscarinic agonist-stimulated chloride efflux pathway is associated with fluid secretion in rat parotid acinar cells. Biochem Biophys Res Commun 145:754–759
Miller JE, Moczydlowski E, Latorre R, Phillips M (1985) Charybdotoxin, a protein inhibitor of single Ca2+-dependent K+ channels from mammalian skeletal muscle. Nature 313:316–318
Nauntofte B (1992) Regulation of electrolyte and fluid secretion in salivary acinar cells. Am J Physiol 263:G823-G837
Nauntofte B, Dissing S (1988) Cholinergic-induced electrolyte transport in rat parotid acini. Comp Biochem Physiol 90:739–746
Nauntofte B, Dissing S (1988) K+ transport and membrane potentials in isolated rat parotid acini. Am J Physiol 255:C508-C518
Nauntofte B, Dissing S (1989) Cholinergic-induced HCO −3 loss from rat parotid acini. Proc Finn Dent Soc 85:307–317
Nauntofte B, Poulsen JH (1986) Effects of Ca2+ and furosemide on Cl− transport and O2 uptake in rat parotid acini. Am J Physiol 251:C175-C185
Petersen OH (1986) Calcium-activated potassium channels and fluid secretion by exocrine glands. Am J Physiol 251:G1-G13
Reference deleted
Reference deleted
Poulsen JH, Kristensen LØ (1982) Is stimulation-induced uptake of sodium in rat parotid acinar cells mediated by a sodium/chloride co-transport system? In: Case RM, Garner A, Turnberg LA, Young JA (eds) Electrolyte and water transport across gastrointestinal epithelia. Raven, New York, pp 199–208
Poulsen JH, Nauntofte B (1990) Role of anion transport in secretion of primary saliva. In: Wong PYD, Young JA (eds) Epithelial secretion of water and electrolytes. Springer, Berlin Heidelberg New York, pp 143–155
Roberts ML, Petersen OH (1978) Membrane potential and resistance changes induced in salivary gland acinar cells by microiontophotoretic application of acetylcholine and adrenergic agonists. J Membr Biol 39:297–312
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na-K-ATPase in chloride transport. Am J Physiol 233:F298-F306
Tamarin A, Sreebny LM (1965) The rat submaxillary salivary gland. A correlative study by light and electron microscopy. J Morphol 117:295–352
Tennyson VM, Pappas GD (1968) The fine structure of the choroid plexus: adult and development stages. Prog Brain Res 29:63–85
Weibel ER, Bolender RP (1973) Stereological techniques for electron microscopic morphometry. In: Hayat MA (ed) Principles and techniques of electron microscopy, vol 3. Van Nostrand-Reinhold, New York, pp 237–296
Wong MMY, Foskett JK (1991) Oscillations of cytosolic sodium during calcium oscillations in exocrine acinar cells. Science 254:1014–1016
Young YA, Van Lennep EW (1978) The morphology of salivary glands. Academic Press, London
Reference deleted
Young JA, Cook DI, Van Lennep EW, Roberts M (1987) Secretion by salivary glands. In: Johnson LR, Christensen J, Jackson M, Jacobsen E, Walsh J (eds) Physiology of the gastrointestanl tract, vol 2, 2nd Ed, Raven, New York, pp 773–815
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Poulsen, J.H., Bundgaard, M. Quantitative estimation of the area of luminal and basolateral membranes of rat parotid acinar cells: some physiological applications. Pflugers Arch. 429, 240–244 (1994). https://doi.org/10.1007/BF00374318
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374318