Abstract
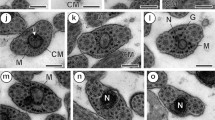
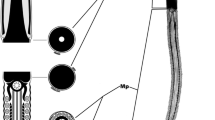
Spermatozoa and some stages of spermatogenesis were studied for four species of aplousobranch ascidians. Spermatozoa of Clavelina lepadiformis (Müller) (family Clavelinidae) are plesiomorphous in that they have apical acrosomal vesicles and a moderately elongated, cylindrical nucleus. The elongated mitochondrion is twisted ea. 11/2 times around the nucleus. In their ultrastructural morphology, C. lepadiformis sperm conform in some respects to the least-derived ascidian sperm, those of the phlebobranch ascidian Ciona intestinalis L.; however, the sperm of Clavelina lepadiformis have two apomorphies not shared with those of Ciona sp.: (1) the mitochondrion of Clavelina lepadiformis is long and spiralled along the entire nucleus rather than being comparatively compact and not at all helical; (2) the mitochondrial cristae are elongated parallel to the long axis of the nucleus, whereas in Ciona sp. sperm the cristae are unmodified. In Distaplia sp., Aplidium sp. and Synoicum pulmonaria (Ellis and Solander) the spermatozoa are more derived and consist of a proximal cylindrical and a distal corkscrew-like part. The mitochondrion in Distaplia sp. and Aplidium sp. contains electron-dense material and extends in a long thread around the nucleus. In S. pulmonaria the mitochondrion surrounds the anterior part of the nucleus in mature spermatozoa, and an elongated, dense structure displaying fine striation is enclosed in the mitochondrion in late spermatids. The sperm ultrastructural morphology observed in this study is consistent with the majority view that clavelinids are closest to the ancestral ascidian but is also consistent with other conclusions, particularly that the Cionidae are closest to the stem ascidian.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Literature cited
Afzelius, B. A., Franzén, Å. (1971). The spermatozoon of the jellyfish Nausithoe. J. Ultrastruct. Res. 37: 186–199
Berrill, N. J. (1936). Studies in tunicate development. V. Evolution and classification. Phil. Trans. R. Soc. (Ser. B) 226: 43–70
Brien, P. (1948). Embranchement des tuniciers. Traité Zool. 11: 553–894
Burighel, P., Martinucci, G. B., Magri, F. (1985). Unusual structures in the spermatozoa of the ascidians Lissoclinum perforatum and Diplosoma listerianum (Didemnidae). Cell Tissue Res. 241: 513–521
Cloney, R. A., Abbott, L. C. (1980). The spermatozoa of ascidians: acrosome and nuclear envelope. Cell Tissue Res. 206: 261–270
Cotelli, R., De Santis, R., Rosati, F., Monroy, A. (1980). Acrosome differentiation in the spermatogenesis of Ciona intestinalis. Dev. Growth Differentiation 22: 561–569
Flood, P. R., Afzelius, B. A. (1978). The spermatozoon of Oikopleura dioica Fol (Larvacea, Tunicata). Cell Tissue Res. 191: 27–37
Franzén, Å., (1956). On spermiogenesis, morphology of the spermatozoon and biology of fertilization among invertebrates. Zool. Bidr. Upps. 31: 354–482
Franzén, Å. (1958). On sperm morphology and acrosome filament formation in some Annelida, Echiuroidea, and Tunicata. Zool. Bidr. Uppsala 33: 1–28
Franzén, Å. (1970). Phylogenetic aspects of the morphology of spermatozoa and spermiogenesis. In: Baccetti, B. (ed.) Comparative spermatology. Academic Press, New York, p. 29–46
Franzén, Å. (1976). The fine structure of spermatid differentiation in a tunicate, Corella parallelogramma (Muller). ZOON 4: 115–120
Franzén, Å. (1977). Sperm structure with regard to fertilization biology and phylogenetics. Verh. dt. zool. Ges. 1977: 123–138
Franzén, Å. (1983). Ultrastructural studies of spermatozoa in three bivalve species with notes on evolution of elongated sperm nucleus in primitive spermatozoa. Gamete Res. 7: 199–214
Fukumoto, M. (1979). Tube-like structures in mitochondria of tunicate (Pyura vittata) spermatids. J. Ultrastruct. Res. 68: 1–5
Fukumoto, M. (1981). The spermatozoa and spermiogenesis of Perophora formosana (Ascidia) with special reference to the striated apical structure and the filamentous structures in the mitochondrion. J. Ultrastruct. Res. 77: 37–53
Fukumoto, M. (1983). Fine structure and differentiation of the acrosome-like structure in solitary ascidians, Pyura haustor and Styela plicata. Dev. Growth Differentiation 25: 503–515
Fukumoto, M. (1984). Fertilization in ascidians: acrosome fragmentation in Ciona intestinalis spermatozoa. J. Ultrastruct. Res. 87: 252–262
Fukumoto, M. (1985). Acrosome differentiation in Molgula manhattensis (Ascidiacea, Tunicata). J. Ultrastruct. Res. 92: 158–166
Fukumoto, M. (1986). The acrosome in ascidians I. Pleurogona. Int. J. Invert. Reprod. Dev. 10: 335–346
Fukumoto, M. (1990). Morphological aspects of ascidian fertilization. Zool. Sci. 7: 989–998
Garstang, W. (1928). The morphology of the Tunicata, and its bearing on the phylogeny of the Chordata. Q. Jl. microsc. Sci. 72: 51–187
Hawkins, C. L., Kott, P., Parry, D. L., Swinehart, J. H. (1983). Vanadium content and oxidation state related to ascidian phylogeny. Comp. Biochem. Physiol. 76B: 555–558
Holland, L. Z. (1988). Spermatogenesis in the salps Thalia democratica and Cyclosalpa affinis (Tunicata: Thaliacea): an electron microscope study. J. Morph. 198: 189–204
Holland, L. Z. (1989). Fine structure of spermatids and sperm of Dolioletta gegenbauri and Doliolum nationalis (Tunicata: Thaliacea): implications for tunicate phylogeny. Mar. Biol. 101: 83–95
Holland, L. Z. (1990). Spermatogenesis in Pyrosoma atlanticum (Tunicata: Thaliacea: Pyrosomatida): implications for tunicate phylogeny. Mar. Biol. 105: 451–470
Holland, L. Z. (in press). The phylogenetic significance of tunicate sperm morphology. In: Baccetti, B. (ed.) Sixth International Congress of Spermatology, Siena, Italy
Holland, L. Z., Gorsky, G., Fenaux, R. (1988). Fertilization in Oikopleura dioica (Tunicata, Appendicularia): acrosome reaction, cortical reaction and sperm-egg fusion. Zoomorphology 108: 229–243
Kott, P. (1969). Antarctic Ascidiacea. Antarctic Res. Ser. 13: 1–239
Lambert, C. C., Epel, D. (1979). Calcium-mediated mitochondrial movement in ascidian sperm during fertilization. Devl Biol. 69: 296–304
Schabtach, E., Ursprung, H. (1965). The fine structure of the sperm of a tunicate, Ascidia nigra. J. exp. Zool. 159: 357–366
Tuzet, O., Bogaraze, D., Lafargue, F. (1972). Recherches ultrastructurales sur la spermiogenese de Diplosoma listerianum (Milne-Edwards, 1841) et Lissoclinum pseudoleptoclinum (Von Drasche, 1883) ascidies, composées, aplousobranches. Annls Sci. nat. (sér. Zool.) (12th series) 14: 177–190
Tuzet, O., Bogaraze, D., Lafargue, F. (1974). La spermatogenese de Polysyncratum lacazei, 1872 et Trididemnum cereum Giard 1872 (Ascidies composées, Aplousobranches). Bull. biol. Fr. Belg. 108: 151–167
Villa, L. (1981). An electron microscope study of spermiogenesis and spermatozoa of Molgula impura and Styela plicata (Ascidiacea, Tunicata). Acta Embryol. Morph. exp. (N.S.) 2: 69–85
Wirth, U. (1984). Die Struktur der Metazoen-Spermien und ihre Bedeutung für die Phylogenetik. Verh. naturw. Ver. Hamb. 27: 295–362
Woollacott, R. M. (1977). Spermatozoa of Ciona intestinalis and analysis of ascidian fertilization. J. Morph. 152: 77–88
Young, J. Z. (1950). The life of vertebrates. Clarendon Press, Oxford
Author information
Authors and Affiliations
Additional information
Communicated by T. Fenchel, Helsingør
Rights and permissions
About this article
Cite this article
Franzén, Å. Spermatozoan ultrastructure and spermatogenesis in aplousobranch ascidians, with some phylogenetic considerations. Marine Biology 113, 77–87 (1992). https://doi.org/10.1007/BF00367641
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00367641