Abstract
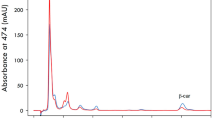
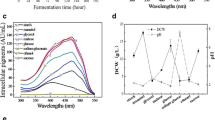
During fermenter cultivation of Phaffia rhodozyma on a grape juice medium, the presence of glucose initially delayed fructose utilization, although fructose was consumed before glucose depletion. Total pigment and astaxanthin production were growth associated and reached maximum values of 15.9 μg/ml and 9.8 μg/ml, respectively, after depletion of the carbon source. The total cellular pigment and astaxanthin content increased during the stationary growth phase due to a decrease in biomass, reaching final values of 2120 μg/g and 1350 μg/g, respectively, without the volumetric concentration in the culture changing. The final cell yield was 0.33 g/g sugar utilized. High sugar concentrations in shake-flasks as well as O2 limitation decreased the astaxanthin content of the cells. Addition of yeast extract to a grape juice minimal medium markedly increased the maximum specific growth rate, total pigment and astaxanthin content of the cells. An excess of ammonia decreased the intracellular astaxanthin content, which reached a maximal value in cultures with no residual glucose or ammonia.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
An, G.-H., Schuman, D.B. & Johnson, E.A. 1989 Isolation of Phaffia rhodozyma mutants with increased astaxanthin content. Applied and Environmental Microbiology 55, 116–124.
Bajpai, P.K., Bajpai, P. & Ward, O.P. 1992 Optimization of culture conditions for production of eicosapentaenoic acid by Mortierella elongata NRRL 5513. Journal of Industrial Microbiology 9, 11–18.
Britton, G. 1983 Carotenoids. In The Biochemistry of Natural Pigments, eds Benkovic, S.J., Elmore, D.T., Lewis, J., Muetterties, E.L., Schofield, K., Thomas, J.M. & Thrush, B.A., pp. 23–73. Cambridge: Cambridge University Press.
Chaney, A.L. & Marbach, E.P. 1962 Modified reagents for determination of urea and ammonia. Clinical Chemistry 8, 130–132.
Chien, Y.-H. & Jeng, S.-C. 1992 Pigmentation of kuruma prawn, Penaeus japonicus Bate, by various pigment sources and levels of feeding regimes. Aquaculture 102, 333–346.
Du Preez, J.C. & Van Der Walt, J.P. 1983 Fermentation of D-xylose to ethanol by a strain of Candida shehatae. Biotechnology Letters 5, 357–362.
Egli, T. & Fiechter, A. 1981 Theoretical analysis of media used in the growth of yeasts on methanol. Journal of General Microbiology 123, 365–369.
Gentles, A. & Haard, N.F. 1991 Pigmentation of rainbow trout with enzyme-treated and spray-dried Phaffia rhodozyma. The Progressive Fish-Culturist 53, 1–6.
Goodwin, T.W. 1971 Biosynthesis. In Carotenoids, ed Isler, O. pp. 577–636. Basel: Birkhäuser Verlag.
Haard, N.F. 1988 Astraxanthin formation by the yeast Phaffia rhodozyma on molasses. Biotechnology Letters 10, 609–614.
Hansson, L. & Dostálek, M. 1988 Effect of culture conditions on mycelial growth and production of gamma-linolenic acid by the fungus Mortierella ramanniana. Applied Microbiology and Biotechnology 28, 240–246.
Johnson, E.A., Conklin, D.E. & Lewis, M.J. 1977 The yeast Phaffia rhodozyma as a dietary pigment source for salmonids and crustaceans. Journal of the Fish Research Board of Canada 34, 2417–2421.
Johnson, E.A. & Lewis, M.J. 1979 Astaxanthin formation by the yeast Phaffia rhodozyma. Journal of General Microbiology 115, 173–183.
Johnson, E.A., Villa, T.G. & Lewis, M.J. 1980 Phaffia rhodozyma as an astaxanthin source in salmonid diets. Aquaculture 20, 123–134.
Johnson, V., Singh, M., Saini, V.S., Adhikari, D.K., Sista, V. & Yadav, N.K. 1992 Bioemulsifier production by an oleaginous yeast Rhodotorula glutinis IIP-30. Biotechnology Letters 14, 487–490.
Kreger-van Rij, H.J.W. 1984 The Yeasts: a Taxonomic Study, 3rd edn. Amsterdam: Elsevier Science.
Longo, E., Sieiro, C., Velázquez, J.B., Calo, P., Cansado, J. & Villa, T.G. 1992 Astaxanthin production from Phaffia rhodozyma. Biotech Forum Europe 9, 565–567.
Meyer, P.S., Du Preez, J.C. & Kilian, S.G. 1992 Chemostat cultivation of Candida blankii on sugar cane bagasse hemicellulose hydrolysate. Biotechnology and Bioengineering 40, 353–358.
Meyer, P.S., Du Preez, J.C. & Kilian, S.G. 1993 Selection and evaluation of astaxanthin-overproducing mutants of Phaffia rhodozyma. World Journal of Microbiology & Biotechnology 9, 514–520.
Petrik, M., Käppeli, O. & Fiechter, A. 1983 An expanded concept for the glucose effect in the yeast Saccharomyces uvarum: involvement of short-and long-term regulation. Journal of General Microbiology 129, 43–49.
Ratledge, C. 1986 Lipids. In Biotechnology, Vol. 4, eds Pape, H. & Rehm, H.-J. pp. 185–213. Weinheim: Verlag, Chemie.
Riegler, M., Käppeli, O. & Fiechter, A. 1983 The role of limited respiration in the incomplete oxidation of glucose by Saccharomyces cerevisiae. Journal of General Microbiology 129, 653–661.
Storebakken, T. & No, K.H. 1992 Pigmentation of rainbow trout. Aquaculture 100, 209–229.
Sullivan, S. & Levinson, M. 1992 GATT: the wrath of grapes. Washington threatens a trade war with Europe. Newsweek November 16, 42.
Van Zyl, C., Prior, B.A. & Du Preez, J.C. 1988 Production of ethanol from sugar cane bagasse hemicellulose hydrolyzate by Pichia stipitis. Applied Biochemistry and Biotechnology 17, 357–369.
Additional information
The authors are with the Department of Microbiology and Biochemistry, University of the Orange Free State, P.O. Box 339, Bloemfontein 9300, South Africa;
Rights and permissions
About this article
Cite this article
Meyer, P.S., du Preez, J.C. Astaxanthin production by a Phaffia rhodozyma mutant on grape juice. World Journal of Microbiology & Biotechnology 10, 178–183 (1994). https://doi.org/10.1007/BF00360882
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00360882