Abstract
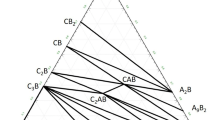
Phase relations in the lime-rich portion of the system CaO-B2O3-SiO2 have been studied by microscopy, infrared spectroscopy and X-ray powder diffraction of heated mixtures and quenched charges. Extensive solid solution of B2O3 in Ca2SiO4 occurs along the Ca2SiO4-Ca3B2O6 boundary, which has been studied in detail. It contains a ternary compound, Ca11B2Si4O22, which is stable to liquidus temperatures, melting incongruently to Ca2SiO4 and liquid at 1420 °C. Ca11B2Si4O22forms a eutectic with Ca3B2O6 at 1400 °C and, in the ternary system, with CaO and Ca3B2O6 at 1390 °C.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
G. A. Rankin and F. E. Wright, Amer. J. Sci. 31 (1915) 1.
A. Muan and E. F. Osborn, “Phase Equilibria Among Oxides in Steelmaking”, American Iron and Steel Institute, New York (Addison Wesley, Reading, MA, 1965) Library of Congress, 64-25807.
R. W. Nurse, in “The Dicalcium Silicate Phase”, Proceedings of the Third International Symposium on the Chemistry of Cement, London (1952) pp. 56–90.
J. Welch and W. Gutt, J. Amer. Ceram. Soc. 42 (1959) 11.
E. T. Carlson, J. Res. Nat. Bur. Stand. 9 (1932) 825.
E, P. Flint and L. S. Wells, ibid. 17 (1936) 727.
K. Suzuki, H. Kazutaka and J. Hideo, J. Ceram. Soc. Jpn 79 (1971) 199.
A. A. Colville and P. A. Colville, Amer. Mineral. 62 (1977) 1003.
J. V. Smith, I. L. Karle, H. Haptman and J. Karle, Acta Crystallogr. 13 (1960) 454.
R. F. Kevtsova and N. V. Belov, Sou. Phys. Crystallogr. 5 (1961) 659.
F. P. Glasser and K. Mohan, Cement Conc. Res. 7 (1977) 1.
E. G. Shame and F. P. Glasser, Brit. Ceram, Trans. J. 86 (1987) 13.
K. Suzuki and I. Hira, J. Ceram. Soc. Jpn 78 (1970) 189.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fletcher, J.G., Glasser, F.P. Phase relations in the system CaO-B2O3-SiO2 . JOURNAL OF MATERIALS SCIENCE 28, 2677–2686 (1993). https://doi.org/10.1007/BF00356203
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00356203