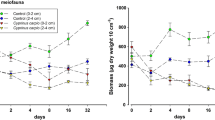
Summary
Experiments conducted on rocky bottoms at 7–11 m depth in the San Juan Archipelago, Washington assessed effects of canopies of understory kelps on growth of benthic suspension feeders, determined the mechanisms responsible for effects, and assessed the influence of kelp canopies on survivorship of benthic fauna. Kelp canopics influenced growth rates of diverse suspension feeders. At several sites the musselMytilus edulis, the barnacleBalanus glandula, and the serpulid polychaetePseudochitinopoma occidentalis grew faster on the bottom beneath kelp canopies than on nearby exposed substrata. The cheilostome bryozoanMembranipora membranacea showed a mixed response to kelp canopies, growing faster in exposed regions at one site, but faster beneath canopies at another. There were no differences in growth of 2 other species (the cheilostome bryozoanCheilopora praelonga and the spongeMyxilla incrustans) between kelp and no-kelp treatments; however, some processes influenced by plant canopies affected their growth. Specific mechanisms responsible for kelp effects on growth were assessed in a series of field experiments usingPseudochitinopoma, Membranipora, Cheilopora andMyxilla. Particulate deposition on the bottom, which is more intense beneath canopies, negatively affected growth of all 4 species. Kelps also reduced rates of flow and prevented devented development of microalgal turfs beneath the canopy.Pseudochitinopoma grew faster in the weaker flows below canopies and bothCheilopora andMyxilla grew faster where there were no microalgal turfs. These other effects of kelp canopies were at least as important to growth (in the cases ofCheilopora andMyxilla) or more important to growth (in the case ofPseudochitinopoma) than were the general, deleterious effects of higher sedimentation beneath canopies.
The lower growth rates caused by higher sedimentation beneath kelp canopies did not result in higher rates of animal mortality. Surprisingly, kelp canopies typically did not influence mortality due to predation. For 7 of 12 taxa, mortality rates did not differ between kelp-covered and exposed treatments. Significantly higher mortality occurred outside canopies for only 4 of 12 taxa, and for at least 2 of these 4 differences probably were not related to predation.Mytilus, a species rare at these depths, exhibited higher mortality beneath kelp canopies due to predation by crabs. Other macrophytes in fresh and salt water, as well as some benthic animals that create complex, 3-dimensional habitats, should influence benthic organisms and assemblages in ways analogous to the kelps acting through their effects on flow, particle transport, and shading.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Armitage P (1977) Statistical methods in medical research. Blackwell Scientific, Oxford, UK
Bernstein BB, Jung N (1979) Selective pressures and coevolution in a kelp canopy in Southern California Ecol Monogr 49: 335–355
Blundon JA, Kennedy VS (1982) Refuges for infaunal bivalves from blue crab.Calinectes sapidus (Rathburn), predation in Chesapeake Bay J Exp Mar Biol Ecol 65: 67–81
Bricelj VM, Epp J, Malouf RE (1987) Intraspecific variation in reproductive and somatic growth cycles of bay scallopsArgopecten irradians. Mar Ecol Prog Ser 36: 123–137
Brönmark C (1985) Interactions between macrophytes, epiphytes and herbivores: an experimental approach. Oikos 45: 26–30
Brönmark C (1988) Effects of vertebrate predation on freshwater gastropods: an exclosure experiment. Hydrobiologia 169: 363–370
Cancino JM, Muñoz J, Muñoz M, Orellana MC (1987) Effects of the bryozoanMembranipora tuberculata (Bosc.) on the photosynthesis and growth ofGelidium rex Santelices et Abbott. J Exp Mar Biol Ecol 113: 105–112
Carpenter SR, Lodge DM (1986) Effects of submersed macrophytes on ecosystem processes. Aquat Botn 26: 341–370
Carr MH (1989) Effects of macroalgal assemblages on the recruitment of temperate zone reef fishes. J Exp Mar Biol Ecol 126: 59–76
Coyer JA (1979) The invertebrate assemblage associated withMacrocystis pyrifera and its utilization as a food source by kelp forest fishes. Ph D, Univ. of Southern California
Crowder LB, Cooper WE (1982) Habitat structural complexity and the interaction between bluegills and their prey. Ecology 63: 1802–1813
Dayton PK (1985) The ecology of kelp communities. Ann Rev Ecol Syst 16: 215–245
Dayton PK, Tegner MJ (1984) The importance of scale in community ecology: a kelp forest example with terrestrial analogs. In: Price PW, Slobodchikoff CN, Gaud WS (eds) A new ecology: novel approaches to interactive systems, John Wiley & Sons, New York, USA, pp 457–481
Dennison WC, Alberte RS (1982) Photosynthetic responses ofZostera marina L. (Eelgrass) to in situ manipulations of light intensity. Oecolgia 55: 137–144
Dennison WC, Alberte RS (1985) Role of daily light period in the depth distribution ofZostera marina (Eelgrass). Mar Ecol Prog Ser 25: 51–61
Duggins DO, Eckman JE, Sewell AT (1990) Ecology of understory kelp environments. II. Effects of kelps on recruitment of benthic invertebrates. J Exp Mar Biol Ecol 143: 27–45
Duggins DO, Simenstad CA, Estes JA (1989) Magnification of secondary production by kelp detritus in coastal marine ecosystems. Science 245: 170–173
Ebeling AW, Laur DR (1985) The influence of plant cover on surf perch abundance at an offshore temperate reef. Env Biol Fish 12: 169–179
Ebeling AW, Laur DR (1988) Fish populations in kelp forests without sea otters: effects of severe storm damage and destructive sea urchin grazing. In: VanBlaricom G, Estes J (eds), The community ecology of sea otters, Springer-Verlag, New York, USA, pp 169–191
Eckman JE (1987) The role of hydrodynamics in recruitment, growth and survival ofArgopecten irradians (L.) andAnomia simplex (D'Orbigny) within eelgrass meadows. J Exp Mar Biol Ecol 106: 165–192
Eckman JE, Nowell ARM, Jumars PA (1981) Sediment destabilization by animal tubes. J Mar Res 39: 361–374
Eckman JE, Duggins DO, Sewell AT (1989) Ecology of understory kelp environments. I. Effects of kelps on flow and particle transport near the bottom. J Exp Mar Ecol 129: 173–187
Fauchald K, Junars PA (1979) The diet of worms: a study of polychaete feeding guilds. Oceanogr. Mar Biol Ann Rev 17: 193–284
Feder HM (1959) The food of the starfish,Pisaster ochraceus, along the California Coast. Ecology 40: 721–724
Fréchette M, Butman CA, Geyer WR (1989) The importance of boundary-layer flows in supplying phytoplankton to the benthic suspension feeder,Mytilus edulis L. Limnol Oceanogr 34: 19–36
Gaines SD, Roughgarden J (1987) Fish in offshore kelp forests affect recruitment to intertidal barnacle populations. Science 235: 479–481
Gerrish N, Bristow JM (1979) Macroinvertebrate associations with aquatic macrophytes and artificial substrates. J Great Lakes Res 5: 69–72
Gilinsky E (1984) The role of fish predation and spatial heterogeneity in determining benthic community structure. Ecology 65: 455–468
Griffiths RJ (1981) Population dynamics and growth of the bivalveChoromytilus meriodionalis (Kr.) at different tidal heights. Estuar Coastal Shelf Sci 12: 101–118
Grizzle RE, Lutz RA (1989) A statistical model relating horizontal seston fluxes and bottom sediment characteristics to growth ofMercenaria mercenaria. Mar Biol 102: 85–93
Harvell CD, Caswell H, Simpson P (1990) Density effects in a colonial monoculture: experimental studies with a marine bryozoan (Membranipora membranacea L.). Oecologia 82: 227–237
Heck KL Jr., Thoman TA (1981) Experiments on predator-prey interactions in vegetated aquatic habitats. J Exp Mar Biol Ecol 53: 125–134
Hershey AE (1985) Effects of predatory sculpin on the chironomid communities in an Arctic lake. Ecology 66: 1131–1138
Hilbish TJ (1985) Demographic and temporal structure of an allele frequency cline in the musselMytilus edulis. Mar Biol 86: 163–171
Hutchinson GE (1985) A treatise on limnology, vol. III. Limnological botany. John Wiley & Sons, New York
Jackson GA (1984) Internal wave attenuation by coastal kelp stands. J Phys Oceanogr 14: 1300–1306
Jackson GA (1986) Interaction of physical and biological processes in the settlement of planktonic larvae. Bull Mar Sci 39: 202–212
Jackson GA, Winant CD (1983) Effects of a kelp forest on coastal currents. Cont Shelf Res 2: 75–80
Kaufmann KW (1981) Fitting and using growth curves. Oecologia 49: 293–299
Kennelly SJ (1987) Physical disturbances in an Australian kelp community. II. Effects of understorey species due to differences in kelp cover. Mar Ecol Prog Ser 40: 155–165
Kennelly SJ (1989) Effects of kelp canopies on understorey species due to shade and scour. Mar Ecol Prog Ser 50: 215–224
Khailov KM, Burlakova ZP (1969) Release of dissolved organic matter by marine seaweeds and distribution of their total organic production to inshore communities. Limnol Oceanogr 14: 521–527
Klumpp DW, Nichols PD (1983) Utilization of the seagrassPosidonia australis) as food by the rock crabNectocarcinus integrifons (Latreille) (Crustacea: Decapoda: Portunidae). Mar Biol Lett 4: 331–339
Laur DR, Ebeling AW (1983) Predator-prey relationships in surfperches. Environ Biol Fish 8: 217–229
Leonard AB, (1989) Functional response inAntedon mediterranea (Lamarck) (Echinodermata: Crinoidea): the interaction of prey concentration and current velocity on a passive suspension feeder. J Exp Mar Biol Ecol 127: 81–103
Leonard AB, Strickler JR, Holland ND (1988) Effects of current speed on filtration during suspension feeding inOligometra serripinna (Echinodermata: Crinoidea). Mar Biol 97: 111–126
Linley EAS, Newell RC, Bosma SA (1981) Heterotrophic utilization of mucilage released during fragmentation of kelp (Ecklonia maxima andLaminaria pallida). I. Development of microbial communities associated with the degradation of kelp mucilage. Mar Ecol Prog Ser 4: 31–41
McDonald JH, Koehn RK (1988). The musselsMytilus galloprovincialis andM. trossulus on the Pacific coast of North America. Mar Biol 99: 111–118
McFadden CS (1986) Colony fission increases particle capture rates of a soft coral: advantages of being a small colony. J Exp Mar Biol Ecol 103: 1–20
Mann KH (1973) Seaweeds: their productivity and strategy for growth. Science 182: 975–981
Mann KH (1988) Production and use of detritus in various freshwater, estuarine and coastal marine ecosystems. Limnol Oceanogr 33: 910–930
Mauzey KP (1966) Feeding behavior and reproductive cycles inPisaster ochraceus. Biol Bull 131: 127–144
Nelson WG (1979) Experimental studies of selective predation on amphipods: consequences for amphipod distribution and abundance. J Exp Mar Biol Ecol 38: 225–245
Nowell ARM, Jumars PA, Eckman JE (1981) Effects of biological activity on the entrainment of marine sediments. Mar Geol 42: 133–153
Okamura B (1984) The effects of ambient flow velocity, colony size and upstream colonies on the feeding success of bryozoa. I.Bugula stolonifera Ryland, an arborescent species. J Exp Mar Biol Ecol 83: 179–193
Okamura B (1985) The effects of ambient flow velocity, colony size and upstream colonies on the feeding success of bryozoa. II.Conopeum reticulum (Linnaeus), an encrusting species. J Exp Mar Biol Ecol 89: 69–80
Orth RJ (1977) The importance of sediment stability in seagrass communities. In: Coull BC (ed), Ecology of marine benthos, Univ. South Carolina Press, Columbia, USA pp 281–300
Orth RJ, Heck KL Jr., von Montfrans J (1984) Faunal communities in seagrass beds: a review of the influence of plant structure and prey characteristics on predator-prey relationships. Estuaries 7: 339–350
Page HM (1986) Differences in population structure and growth rate of the stalked barnaclePollicipes polymerus between a rocky headland and an offshore oil platform. Mar Ecol Prog Ser 29: 157–164
Paine RT (1966) Food web complexity and species diversity. Am Natur 100: 65–75
Paine RT (1980) Food webs: linkage, interaction strength and community infrastructure. J Anim Ecol 49: 667–685
Palumbi SR (1985) Spatial variation in an algal-sponge commensalism and the evolution of ecological interactions. Am Natur 126: 267–274
Patterson MR (1984) Patterns of whole colony prey capture in the octocoral,Alcyonium siderium. Biol Bull 167: 613–629
Peterson CH (1982) Clam predation by whelks (Busycon spp.): experimental tests of the importance of prey size, prey density and seagrass cover. Mar Biol 66: 159–170
Peterson CH (1986) Enhancement ofMercenaria mercenaria densities in seagrass beds: is pattern fixed during settlement season or altered by subsequent differential survival? Limnol Oceanogr 31: 200–205
Peterson CH, Beal BF (1989) Bivalve growth and higher order interactions: Importance of density, site, and time. Ecology 70: 1390–1404
Peterson CH, Black R (1987) Resource depletion by active suspension feeders on tidal flats: influence of local density and tidal elevation. Limnol Oceanogr 32: 143–166
Peterson CH, Black R (1988) Responses of growth to elevation fail to explain vertical zonation of suspension-feeding bivalves on a tidal flat. Oecologia 76: 423–429
Peterson CH, Summerson HC, Duncan PB (1984) The influence of seagrass cover on population structure and individual growth rat of a suspension-feeding bivalve,Mercenaria mercenaria. J Mar Res 42: 123–138
Ryland JS, Stebbing ARD (1971) Settlement and oriented growth in epiphytic and epizoic bryozoans. In: Crisp DJ (ed), Fourth European marine biology symposium, Cambridge University Press, Cambridge, UK, pp 105–123
Schiel DR, Foster MS (1986) The structure of subtidal algal stands in temperate waters. Oceanogr Mar Biol Ann Rev 24: 265–307
Sculthorpe DC (1985) The biology of aquatic vascular plants, 2nd ed., Lubrecht & Cramer, Forestburg, New York, USA
Sebens KP (1984) Water flow and coral colony size: interhabitat comparisons of the octocoralAlcyonium siderium. Proc Nat Acad Sci USA 81: 5473–5477
Sebens KP (1987) The ecology of indeterminant growth in animals. Ann Rev Ecol Syst 18: 371–407
Seed R (1976) Ecolog. In: Bayne BL (ed), Marine mussels: their ecology and physiology, Cambridge University Press, Cambridge, UK, pp 13–65
Seed R, O'Connor RJ (1981) Community organization in marine algal epifaunas. Ann Rev Ecol Syst 12: 49–74
Shanks AL, Wright WG (1986) Adding teeth to wave action: the destructive effects of wave-borne rocks on intertidal organisms. Oecologia 69: 420–428
Sokal RR, Rohlf FJ (1981) Biometry, 2nd ed., W. H. Freeman & Company, San Francisco, California, USA
Stevenson JC (1988) Comparative ecology of submersed grass beds in freshwater, estuarine, and marine environments. Limnol Oceanogr 33: 867–893
Stoner AW (1980) The role of seagrass biomass in the organization of benthic macrofaunal assemblages. Bull Mar Sci 30: 537–551
Stoner AW (1982) The influence of benthic macrophytes on the foraging behavior of pinfish,Lagodon rhomboides (Linnaeus). J Exp Mar Biol Ecol 58: 271–284
Suchanek TH (1978) The ecology ofMytilus edulis L. in exposed rocky intertidal communities. J Exp Mar Biol Ecol 31: 105–120
Summerson HC, Peterson CH (1984) Role of predation in organizing benthic communities of a temperate-zone seagrass bed. Mar Ecol Prog Ser 15: 63–77
Thompson RJ (1984) The reproductive cycle and physiological ecology of the musselMytilus edulis in a subarctic, non-estuarine environment. Mar Biol 79: 277–288
Trager GC, Hwang J-S, Strickler JR (1990) Barnacle suspension-deeding in variable flow. Mar Biol 105: 117–127
Vadas RL (1977) Preferential feeding: an optimization strategy in sea urchins. Ecol Monogr 47: 337–371
Wethey DS (1984) Spatial pattern in barnacle settlement: day to day changes during the settlement season. J Mar Biol Assoc UK 64: 687–698
Winer BJ (1971) Statistical principles in experimental design, 2nd ed., McGraw-Hill, New York
Witman JD (1987) Subtidal coexistence: storms, grazing, and the zonation of kelps and mussels. Ecol Monogr 57: 167–187
Yoshioka PM (1982) The role of planktonic and benthic factors in the population dynamics of the bryozoanMembranipora membranacea. Ecology 63: 457–468
Young CM, Chia F-S (1984) Microhabitat associated variability in survival and growth of subtidal ascidians during the first 21 days after settlement. Mar Biol 81: 61–68
Young DK, Young MW (1978) Regulation of species densities of seagrass-associated macrobenthos: evidence from field experiments in the Indian River estuary, Florida. J Mar Res 36: 569–593
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eckman, J.E., Duggins, D.O. Life and death beneath macrophyte canopies: effects of understory kelps on growth rates and survival of marine, benthic suspension feeders. Oecologia 87, 473–487 (1991). https://doi.org/10.1007/BF00320409
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00320409