Summary
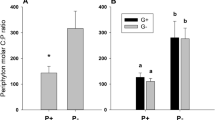
Benthic algal assemblages are regulated by both abiotic (e.g., nutrient) and biotic (e.g., grazing) constraint. The objective of this study was to determine how changes in these two factors affected the structure of an algal assemblage in an ephemeral stream. Coverslips were incubated for 21 days in enclosures containing one of three nutrient environments (ambient, phosphorus-enriched, or phosphorus and nitrogen enriched) and one of four densities of the snail Gonibasis (0, 40, 80, or 120 snails/m2) and examined directly to enumerate the algal assemblage. The effect of grazing on algal biomass was dependent on the nutrient environment. An overstory of diatoms was susceptible to removal by grazing and was not strongly affected by nutrient enrichment. An understory of Stigeoclonium was more resistant to grazing and responded strongly to nutrient enrichment only in the presence of grazers. Snail grazers may mediate nutrient availability to the understory indirectly by removing overlying cells or by direct excretion of nutrients. Multiple interactions occur between benthic herbivores and algae, and, as shown here, some of them are positive and involve modifications of the nutrient environment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
American Public Health Association, American Water Works Association, and Water Pollution Control Federation (1985) Standard methods for the examination of water and wastewater, 16th ed. Washington, D.C.
Bothwell ML (1985) Phosphorus limitation of lotic periphyton growth rates: an intersite comparison using continuous flow troughs (Thompson River System, British Columbia). Limnol Oceanogr 30:527–542
Carrick HJ, Lowe RL, Rotenberry JT (1988) Guilds of benthic algae along nutrient gradients: Relationships to algal community diversity. J N Am Benthol Soc 7:117–128
Cuker BE (1983) Grazing and nutrient interactions in controlling the activity and composition of the epilithic algal community of an arctic lake. Limnol Oceanogr 28:133–141
Fairchild GW, Lowe RL, Richardson WB (1985) Algal periphyton growth on nutrient-diffusing substrates: an in situ biossay. Ecology 66:465–472
Fairchild GW, Sherman JW (1989) Nutrient control of periphyton growth in softwater lakes. Bull N Am Benthol Soc 6 (1):121
Friedl FE (1974) Nitrogen excretion by the freshwater snail Lymnaea stagnalis jugularis Say. Comp Biochem Physiol 49:617–622
Grimm NB (1988) Role of macroinvertebrates in nitrogen dynamics of a desert stream. Ecology 69:1884–1893
Hart DD (1985) Grazing insects mediate algal interactions in a stream benthic ocmmunity. Oikos 44:40–46
Hill WR, Knight AW (1988) Nutrient and light limitation in two northern Californian streams. J Phycol 24:125–132
Kesler DH (1981) Periphyton grazing by Amnicola limosa: an enclosure-exclosure experiment. J Freshwater Ecol 1:51–59
Lamberti GA, Ashkenas LR, Gregory SV, Steinman AD (1987) Effects of three herbivores on periphyton communities in laboratory streams. J N Am Benthol Soc 6:92–104
Lamberti GA, Gregory SV, Ashkenas LR, Steinman AD, McIntire CD (1989) Productive capacity of periphyton as a determinant of plant-herbivore interactions in streams. Ecology 70:1840–1856
Lamberti GA, Resh VH (1983) Stream periphyton and insect herbivores: an experimental study of grazing by a caddisfly population. Ecology 64:1124–1135
Lentner M, Bishop T (1986) Experimental design and analysis. Valley Book Co., Blacksburg, VA
McAuliffe JR (1984) Resource depression by a stream herbivore: effects on distributions and abundances of other grazers. Oikos 42:327–333
McCormick PV (1990) Direct and indirect effects of consumers on benthic algae in an ephemeral stream food web. Can J Fish Aquat Sci 47:2057–2065
McCormick PV, Stevenson RJ (1989) Effects of snail grazing on benthic algal community structure in different nutrient environments. J N Am Benthol Soc 8:162–172
Patrick R (1970) Benthic stream communities. Am Sci58: 546–549
Pringle CM, Paaby-Hansen P, Vaux PD, Goldman CR (1986) In situ nutrient assays of periphyton growth in a lowland Costa Rican stream. Hydrobiology 134:207–213
Riber HH, Wetzel R (1987) Boundary-layer and internal diffusion effects on phosphorus fluxes in lake periphyton. Limnol Oceanogr 32:1181–1194
SAS Institute (1985) SAS user's guide: Statistics, 5th ed. Cary, NC
Sokal RR, Rohlf FJ (1981) Biometry, 2nd ed. W.H. Freeman and Co., New York
Steinman AD, McIntire CD (1987) Effects of irradiance on the community structure and biomass of algal assemblages in laboratory streams. Can J Fish Aq Sci 44:1640–1648
Steinman AD, Gregory SV, Lamberti GA, Ashkenas LR (1987) Effects of herbivore type and density on the taxonomic structure and physiognomy of algal assemblages in laboratory streams. J Am Benthol Soc 6:175–188
Steinman AD, McIntire CD, Gregory SV, Lamberti GA (1989) Effects of irradiance and grazing on lotic algal assemblages. J Phycol 25:478–485
Stevenson RJ (1984) Procedures for mounting algae in a syrup medium. Trans An Microsc Soc 103:320–321
Stevenson RJ, Glover RM (1990) Interactive effects of algal density and current on nutrient diffusion into periphyton mats. Bull N Am Benthol Soc 7:74
Stockner JG, Shortreed KRS (1978) Enhancement of autotrophic production by nutrient addition in a coastal rainforest stream on Vancouver Island. J Fish Res Bd Canada 35:28–34
Sumner WT, McIntire CD (1982) Grazer-periphyton interactions in laboratory streams. Arch Hydrobiol 93:135–157
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McCormick, P.V., Stevenson, R.J. Grazer control of nutrient availability in the periphyton. Oecologia 86, 287–291 (1991). https://doi.org/10.1007/BF00317542
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00317542