Summary
Fifty-one methylotrophs were checked with respect to their ability of poly-β-hydroxybutyric acid (PHB) production from methanol. One of them, Pseudomonas sp. K, was chosen from its good growth on a minimum synthetic medium. Optimal temperature and pH for its growth were 30° C and 7.0, respectively. Concentrations of PO 3-4 and NH +4 in the medium should be kept at low levels. PHB formation was stimulated by deficiency of nutrient such as NH +4 , SO 2-4 , Mg2+, Fe2+ or Mn2+. Among them, nitrogen deficiency was chosen from its effectiveness and easiness for PHB accumulation.
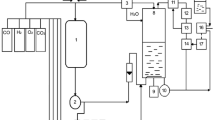
The microorganism was cultivated to produce a large amount of poly-β-hydroxybutyric acid (PHB) from methanol by means of microcomputer-aided fully automatic fed-batch culture technique. During the cultivation, temperature, dissolved oxygen concentration (DO), and methanol concentration in the culture broth were maintained at 30° C 2.5±0.5 ppm and 0.5±0.2 g/l, respectively. Other nutrients, nitrogen source and mineral ions, were also controlled to maintain their initial concentrations in the medium during cell growth phase. When the high cell concentration was achieved (160 g/l), feedings of ammonia and minerals were stopped and only methanol was supplied successively to accumulate PHB. At 175 h, high concentration of PHB (136 g/l) was obtained and total cell concentration became 206 g/l. DO must be maintained above the critical level during the PHB formation phase, too. PHB yield from methanol (g PHB/g methanol) was 0.18 and the maximum PHB content reached 66% of dry weight. Solid PHB produced by the strain had the melting point of 176° C and the average molecular weight of 3.0x105.
Article PDF
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Braunegg G, Sonnleitner B, Lafferty RM (1978) A rapid gas chromatographic method for the determination of poly-β-hydroxybutyric acid in microbial biomass. Eur J Appl Microbiol Biotechnol 6:29–37
Dawes EA, Senior PJ (1973) The role and regulation of energy reserve polymers in microorganism. Adv Microbial Physiol 10:203–253
Emeruwa AC, Hawirko RZ (1973) Poly-β-hydroxybutyrate metabolism during growth and sporulation of Clostridium botulinum. J Bacteriol 116:989–993
Heinzle E, Lafferty RM (1980) A kinetic model for growth and synthesis of poly-β-hydroxybutyric acid (PHB) in Alcaligenes eutrophus H 16. Eur J Appl Microbiol Biotechnol 11:8–16
Lafferty RM, Heinzle E (1977) Extraction of a thermoplastic from bacteria. Chem Rundsch 30:14–16
Pawell KA, Collinson BA, Richardson KR (1981) Japanese Patent 117793
Repaske R, Mayer R (1976) Dense autotrophic cultures of Alcaligenes eutrophus. Appl Environ Microbiol 32:592–597
Schlegel HG, Gottschalk G, Bartha R (1961) Formation and utilization of poly-β-hydroxybutyric acid by knallgas bacteria (Hydrogenomonas). Nature, London, 191:463–465
Senior PJ, Dawes EA (1973) The regulation of poly-β-hydroxybutyrate metabolism in Azotobacter beijerinckii. Biochem J 134:225–238
Sonnleitner B, Heinzle E, Braunegg G, Lafferty RM (1979) Formal kinetics of poly-β-hydroxybutyric acid (PHB) production in Alcaligenes entrophus H 16 and Mycoplana rubra R 14 with respect to the dissolved oxygen tension in ammonium-limited batch cultures. Eur J Appl Microbiol Biotechnol 7:1–10
Suzuki T, Mori H, Yamane T, Shimizu S (1985) Automatic supplementation of minerals in fed-batch culture to high cell mass concentration. Biotechnol Bioeng 27:192–201
Taylor IJ, Anthony C (1976) Acetyl-CoA production and utilization during growth of the facultative methylotroph Pseudomonas AM 1 on ethanol, malonate and 3-hydroxybutyrate. J Gen Microbiol 95:134–145
Ward AC, Rowley BI, Dawes EA (1977) Effect of oxygen and nitrogen limitation on poly-β-hydroxybutyrate biosynthesis in ammonium-grown Azotobacter beijerinckii. J Gen Microbiol 102:61–68
Yamane T, Matsuda M, Sada E (1980), Application of porous Teflon tubing method to automatic fed-batch culture of microorganisms. II. Automatic constant-value control of fed substrate (ethanol) concentration in semibatch culture of yeast. Biotechnol Bioeng 23:2509–2524
Yano T, Mori H, Kobayashi T, Shimizu S (1980) Reusability of broth supernatant as medium. J Ferment Technol 58:259–266
Yano T, Kobayashi T, Shimizu S (1981a) Control system of dissolved oxygen concentration employing a microcomputer. J Ferment Technol 59:295–301
Yano T, Kobayashi T, Shimizu S (1981b) Abstract of Annual Meeting of the Society of Fermentation Technology, Japan, p.4
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Suzuki, T., Yamane, T. & Shimizu, S. Mass production of poly-β-hydroxybutyric acid by fully automatic fed-batch culture of methylotroph. Appl Microbiol Biotechnol 23, 322–329 (1986). https://doi.org/10.1007/BF00257027
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00257027