Summary
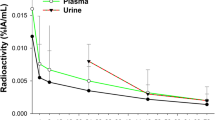
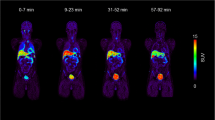
Of all the bleomycin-containing radiopharmaceuticals, bleomycin 57Co has proven the most useful whole-body tumor-imaging agent. We have studied its in vitro physicochemical properties and in vivo disposition in animals and man to optimize its use as a scanning agent. High-pressure liquid chromatographic analysis of the standard bleomycin 57Co preparation (1 unit bleomycin plus 1 mCi chloride 57Co showed it to contain 1% free chloride 57Co). Dialysis experiments showed that bleomycin 57Co does not dissociate as it diffuses through a dialysis membrane. In nine patients, bleomycin 57Co had a t 2/β1 of 3.4 h, a t 2/γ1 of 45.8 h, a Vd of 12.1 liters/m2 and a 24-h urinary excretion of 82.1% of the administered dose. In comparison, bleomycin, assayed by radioimmunoassay, had a terminal phase plasma t1/2 of only 4.0 h, a similar Vd (17.3 liters/m2), and a 24-h urinary excretion of only 44.8%. Bleomycin 57Co tumorto-plasma concentration ratios ranged from 14.1–23.8 at 1 day to 5.4 at 2 days after administration. Our finding that tumor imaging with bleomycin 57Co is best achieved at 24 h is well explained by its almost complete urinary elimination in the first few hours after administration and the peak tumor-to-plasma ratio achieved at 24 h. One disadvantage of bleomycin 57Co as a scanning agent is its very extended plasma t1/2 In rabbits chloride 57Co has the same prolonged plasma terminal elimination phase (t 2/γ1 ) as our standard bleomycin 57Co preparation, which contains chloride 57Co as a 1% impurity. Removal of this impurity prior to scanning or use of cold cobalt chloride to help eliminate it from the plasma might result in a shortened bleomycin 57Co plasma t 2/γ1 .
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alberts, D. S., Chen, H.-S. G., Liu, R., et al.: Bleomycin pharmacokinetics in man. I. Intravenous administration. Cancer Chemother. Pharmacol. 1, 177–181 (1978)
Broughton, A., Strong, J. E.: Radioimmunoassay of bleomycin. Cancer Res. 36, 1430–1434 (1977)
Grove, R. B., Eckelman, W. C., Reba, R. C.: Distribution of labeled bleomycin in normal and tumor-bearing mice. J. Nucl. Med. 14, 917–919 (1973)
Kahn, P. C., Milunsky, C., Dewanjee, M. K., et al.: The place of 57Co-belomycin scanning in the evaluation of tumors. Am. J. Roentgenol. 129, 267–273 (1977)
Kono, A., Matsushima, Y., Kojima, M., Maeda, T.: Cobalt chelate of bleomycin. I. Physicochemical properties and distribution in tumor bearing mice. Chem. Pharm. Bull. 25 (7), 1725–1731 (1977)
Maeda, T., Kohno, A., Kojima, M.: Tumor scanning with bleomycin 57Co. Radioisotopes 21, 436–438 (1972)
Metzler, C. M.: NONLIN: A computer program for parameter estimation in nonlinear situations. Kalamazoo, Michigan: Upjohn Co. 1969
Müller, W. E. G., Zahn, R. K.: Determination of the bleomycininactivating enzyme in biopsies. Gann 67, 425–430 (1976)
Nouel, J. P.: Radioactive metal-bleomycin complex for the diagnosis of cancer. In: Fundamental and clinical studies of bleomycin. Carter, S. K., Ichikawa, T., Mathé, G., and Umezawa, H. (eds.), p. 301–316. University Park Press 1976
Nouel, J. P., Renault, H., Robert, J., et al.: La bléomycine marquée au Co-57. Intérêt dans le diagnostic des tumeurs malignes et de leur extension. Nouv. Press Med. 1, 95–98 (1972)
Poulose, K. P., Watkins, A. E., Reba, R. C., et al.: Cobaltlabeled bleomycin — a new radiopharmaceutical for tumor localization. A comparative clinical evaluation with gallium citrate. J. Nucl. Med. 16, 839–841 (1975)
Rasker, J. J., Van de Poll, M. A. P. C., Beekhuis, H., et al.: Some experience with 57Co-labeled bleomycin as a tumor-seeking agent. J. Nucl. Med. 16, 1058–1069 (1975)
Robert, J., Renault, H., Rapin, J., Rudler, M., Nouel, J. P.: Métabolisme de la bléomycine marquée au cobalt-57 chez la souris. I. Distribution et cinétique. Therapie 28, 933 (1973a)
Robert, J., Renault, H., Rapin, J., Rudler, M., Nouel, J. P.: Métabolisme de la bléomycine marquée par un cation divalent. II. Etude des métabolites urinaires chez le rat et comparaison de l'élimination tissulaire du cation seul on chélaté par la bléomycin. Therapie 28, 941 (1973b)
Sedman, A. J., Wagner, J. G.: CSTRIP: A Fortran IV computer program for obtaining initial polyexponential parameter estimates. J. Pharm. Sci. 65, 1006–1010 (1976)
Strong, J. E., Broughton, A., Crooke, S. T.: Specificity of antisera produced against bleomycin. Cancer Treat. Rep. 61 (8), 1509–1512 (1977)
Umezawa, H.: Recent studies on bleomycin. Lloydia 10 (1), 67–81 (1977)
Van de Poll, M. A. P. C., Versluis, A., Rasker, J. J., Jurjens, H., Woldring, M. G.: Labelling of bleomycin with cobalt-57, indium-111, technetium-99m, mercury-197, lead-203, and copper-67. Nucl. Med. 15 (2), 86–90 (1976)
Woolfenden, J. M., Alberts, D. S., Hall, J. N., et al.: Cobalt-57 bleomycin for imaging head and neck tumors. Cancer (in press, May 1979)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alberts, D.S., Chen, HS.G., Woolfenden, J.M. et al. Pharmacokinetics of bleomycin in man. Cancer Chemother. Pharmacol. 3, 33–40 (1979). https://doi.org/10.1007/BF00254417
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00254417