Summary
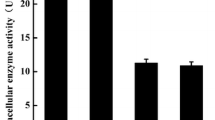
The β-casein specific cell wall proteolytic system of Lactococcus lactis subsp. cremoris P8-2-47 contains a metal-independent X-prolyl-dipeptidyl-aminopeptidase. Suitable substrates for its assay are Gly-Pro-nitroanilide and Ala-Pronitroanilide. It is suggested that the function of the enzyme is to cleave the proline-rich sequences of β-casein, as shown by the degradation of β-casomorphin. It is a serine proteinase with a monomer molecular mass of about 90 000 daltons, a temperature optimum of 45°–50°C, and a pH optimum of about 7.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andresen A, Geis A, Krusch U, Teuber M (1984) Plasmidmuster milchwirtschaftlich genutzter Starterkulturen. Milchwissenschaft 39:140–143
Bockelmann W (1987) Das proteolytische System von Milchsäurebakterien. PhD. Thesis, University of Kiel
Boven A van, Tan PST, Konings WN (1988) Purification and characterization of a dipeptidase from Streptococcus cremoris WG2. Appl Environ Microbiol 54:43–49
Exterkate FA (1975) An introductory study of the proteolytic system of Streptococcus cremoris strain HP. Neth Milk Dairy J 29:303–318
Exterkate FA (1976) The proteolytic system of a slow lactic acid producing variant of Streptococcus cremoris HP. Neth Milk Dairy J 30:3–8
Exterkate FA, de Veer GJCM (1987) Partial isolation and degradation of caseins by cell wall proteinase(s) of Streptococcus cremoris HP. Appl Environ Microbiol 53:1482–1486
Geis A, Bockelmann W, Teuber M (1985) Simultaneous extraction and purification of a cell wall associated peptidase and β-casein specific protease from Streptococcus cremoris AC1. Appl Microbiol Biotechnol 23:79–84
Kok J, van Dijl JM, van der Vossen JMBM, Venema G (1985) Cloning and expression of a Streptococcus cremoris proteinase in Bacillus subtilis and Streptococcus lactis. Appl Environ Microbiol 50:94–101
Kok J, Leenhouts KJ, Haandrikman AJ, Ledebeer AM, Venema G (1988) Nucleotide sequence of the cell wall proteinase gene of Streptococcus cremoris WG2. Appl Environ Microbiol 54:231–238
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Meyer J, Jordi R (1987) Purification and characterization of X-prolyl-dipeptidyl-aminopeptidase from Lactobacillus lactis and from Streptococcus thermophilus. J Dairy Sci 70:738–745
Miller CG, Mackinnon K (1974) Peptidase mutants of Salmonella typhimurium. J Bact 120:355–363
Monnet V, Le Bars D, Gripon JC (1986) Specificity of a cell wall proteinase from Streptococcus lactis NCDO 763 towards bovine β-casein. FEMS Microbiol Lett 36:127–131
Sedmak JJ, Grossberg SE (1977) A rapid, sensitive, and versatile assay for protein using Coomassie Brilliant Blue G250. Anal Biochem 79:544–552
Terzaghi BE, Sandine WE (1975) An improved medium for lactic streptococci and their bacteriophages. Appl Microbiol 29:807–813
Thomas TD, Pritchard GG (1987) Proteolytic enzymes of dairy starter cultures. FEMS Microbiol Rev 46:245–268
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kiefer-Partsch, B., Bockelmann, W., Geis, A. et al. Purification of an X-prolyl-dipeptidyl aminopeptidase from the cell wall proteolytic system of Lactococcus lactis subsp. cremoris . Appl Microbiol Biotechnol 31, 75–78 (1989). https://doi.org/10.1007/BF00252531
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00252531