Summary
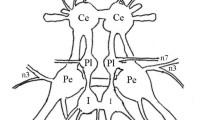
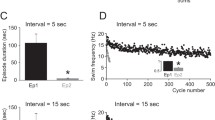
In the pteropodial mollusc Clione limacina, the rhythmic locomotor wing movements are controlled by the pedal ganglia. The locomotor rhythm is generated by two groups of interneurons (groups 7 and 8) which drive efferent neurons. In the present paper, the activity of isolated neurons, which were extracted from the pedal ganglia by means of an intracellular electrode, is described. The following results have been obtained: 1. Isolated type 7 and 8 interneurons preserved the capability for generation of prolonged (100–200 ms) action potentials. The frequency of these spontaneous discharges was usually within the limit of locomotor frequencies (0.5–5 Hz). By de- or hyperpolarizing a cell, one could usually cover the whole range of locomotor frequencies. This finding demonstrates that the locomotor rhythm is indeed determined by the endogenous rhythmic activity of type 7 and 8 interneurons. 2. Type 1 and 2 efferent neurons, before isolation, could generate single spikes as well as high-frequency bursts of spikes. These two modes of activity were also observed after isolating the cells. Thus, the bursting activity of type 1 and 2 neurons, demonstrated during locomotion, is determined by their own properties. Type 3 and 4 efferent neurons generated only repeated single spikes both before and after isolation. 3. The activity of the isolated axons of type 1 and 2 neurons did not differ meaningfully from the activity of the whole cells. Furthermore, in the isolated pedal commissure, we found units whose activity (rhythmically repeating prolonged action potentials) resembled the activity of type 7 and 8 interneurons. These units seemed to be the axons of type 7 and 8 interneurons. Thus, different parts of the cell membrane (soma and axons) have similar electric properties.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arshavsky YuI, Beloozerova IN, Orlovsky GN, Panchin YuV, Pavlova GA (1985a) Control of locomotion in marine mollusc Clione limacina. I. Efferent activity during actual and fictitious swimming. Exp Brain Res 58: 255–262
Arshavsky YuI, Beloozerova IN, Orlovsky GN, Panchin YuV, Pavlova GA (1985b) Control of locomotion in marine mollusc Clione limacina. II. Rhythmic neurons of pedal ganglia. Exp Brain Res 58: 263–272
Arshavsky YuI, Beloozerova IN, Orlovsky GN, Panchin YuV, Pavlova GA (1985c) Control of locomotion in marine mollusc Clione limacina. III. On the origin of locomotory rhythm. Exp Brain Res 58: 273–284
Arhavsky YuI, Beloozerova IN, Orlovsky GN, Panchin YuV, Pavlova GA (1985d) Control of locomotion in marine mollusc Clione limacina. IV. Role of type 12 interneurons. Exp Brain Res 58: 285–293
Arshavsky YuI, Orlovsky GN, Panchin YuV (1985e) Control of locomotion in marine mollusc Clione limacina. V. Photoinactivation of efferent neurons. Exp Brain Res 59: 203–205
Calabrese RL (1979) The roles of endogenous membrane properties and synaptic interaction in generating the heartbeat rhythm of the leech, Hirudo medicinalis. J Exp Biol 82: 163–176
Dagan D, Levitan IB (1981) Isolated identified neurons in cell culture. J Neurosci 1: 731–740
Dyakonova TL (1985) Two types of neurons differing by plastic properties: study of ionic mechanisms. Pavlov J Higher Nerv Activity 35: 552–560 (in Russian)
Friesen WO, Stent G (1978) Neuronal circuits for generating rhythmic movements. Ann Rev Biophys Bioeng 7: 37–61
Fuchs PA, Nicolls JG, Ready DF (1981) Membrane properties and selective connexions of identified leech neurons in culture. J Physiol (Lond) 316: 203–223
Gillette R, Gillette MU, Davis WJ (1980) Action-potential broadening and endogenously sustained bursting are substrates of command ability in feeding neuron of Pleurobranchaea. J Neurophysiol 43: 669–685
Grillner S, Wallen P, McClellan A, Sigwardt K, Williams T, Feldman J (1983) The neural generation of locomotion in the lamprey — an incomplete account. In: Roberts A, Roberts B (eds) Neural origin of rhythmic movements. Symp Soc Exp Biol, Brighton, pp 285–303
Hadley RD, Kater SB, Cohan CS (1983) Electrical synapse formation depends on interaction of mutually growing neurites. Science 221: 466–468
London JA, Merickel M (1979) Single cell isolation: a way to examine network interactions. Brain Res 179: 219–230
Maynard DM, Selverston AI (1975) Organization of the stomatogastric ganglion of the spiny lobster. IV. The pyloric system. J Comp Physiol 100: 161–182
Merickel M, Gray R (1980) Investigation of burst generation by the electrically coupled cyberchron network in the snail Helisoma using a single-electrode voltage clamp. J Neurobiol 11: 73–102
Miller JP, Selverston AI (1982a) Mechanisms underlying pattern generation in lobster stomatogastric ganglion as determined by selective inactivation of identified neurons. II. Oscillator properties of pyloric neurons. J Neurophysiol 48: 1378–1391
Miller JP, Selverston AI (1982b) Mechanisms underlying pattern generation in lobster stomatogastric ganglion as determined by selective inactivation of identified neurons. IV. Network properties of pyloric system. J Neurophysiol 48: 1416–1432
Rose RM, Benjamin PR (1981) Interneuronal control of feeding in the pond snail Lymnaea stagnalis. II. The interneuronal mechanism generating feeding cycles. J Exp Biol 92: 203–228
Satterlie RA (1985) Reciprocal inhibition and postinhibitory rebound produce reverberation in a locomotor pattern generator. Science 229: 402–404
Selverston AI, Miller JP (1980) Mechanisms underlying pattern generation in lobster stomatogastric ganglion as determined by selective inactivation of identified neurons. I. Pyloric system. J Neurophysiol 44: 1102–1121
Sigvardt KA, Grillner S, Wallén P, Dongen van AM (1985) Activation of NMDA receptors elicits fictive locomotion and bistable membrane properties in the lamprey spinal cord. Brain Res 336: 390–395
Stent GS, Thompson WJ, Calabrese RL (1979) Neural control of heartbeat in the leech and in some other invertebrates. Physiol Rev 59: 101–136
Swidchem H van (1979) On the endogenous bursting properties of “light yellow” neurosecretory cells in the freshwater snail Lymnaea stagnalis (L.) J Exp Biol 80: 55–67
Tazaki K, Cooke IM (1979) Spontaneous electrical activity and interaction of large and small cells in cardiac ganglion of the crab, Portunus sanguinolentus. J Neurophysiol 42: 975–999
Tazaki K, Cooke IM (1979b) Isolation and characterization of slow, depolarizing responses of cardiac ganglion neurons in the crab, Portunus sanguinolentus. J Neurophysiol 42: 1000–1021
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arshavsky, Y.I., Deliagina, T.G., Orlovsky, G.N. et al. Control of locomotion in marine mollusc Clione limacina . Exp Brain Res 63, 106–112 (1986). https://doi.org/10.1007/BF00235652
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00235652