Abstract
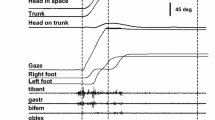
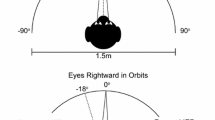
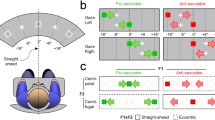
Gaze shifts vary in the extent of eye and head contribution; a large amplitude and/or an eccentric ocular orbital starting position alter the participation of head movement in the shift. The interval between eye onset and head onset determines compensatory counterrolling before and after the shift and the extent of vestibular ocular reflex reduction during the shift. The latency of eye saccades in the head-fixed condition was measured with respect to target amplitude and orbital position in order to establish base-line operations of these two variables as they apply to the headfree condition. Eye movements were measured during single-step saccades in nine young adult humans. The target step, hereafter called a jump, started from three possible fixation lights; e.g., rightward saccades started from the midline (0°) or from -20 or -40° left of the midline, with a maximum amplitude of 80°. The latency of saccades starting from the primary position increased with jump amplitude (amplitude-latency relation). When the eye started eccentrically, the latency was decreased (orbital position-latency relation), with the largest jump amplitudes most affected. These changes can be related to active eye-head coordination. Thus, with a leftward maximal orbital eccentricity, compensatory eye rotation would be impossible with a rightward head movement; however, incorporating the orbital position-latency relation, the forward ocular saccade is expedited by 90 ms. Conversely, with a primary starting position, the ocular component of an 80° gaze saccade could be slowed 125 ms by incorporating the amplitude-latency relation, thus facilitating a head contribution to the gaze shift. The orbital position and amplitude-latency relations were prominent in those subjects with habitually large head contributions to the gaze shift and minimal in individuals with typically small head contributions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baizer JS, Bender DB (1989) Comparison of saccadic eye movements in humans and macaques to single-step and double-step target movements. Vision Res 29: 485–495
Baloh RW, Honrubia V (1976) Reaction time and accuracy of the saccadic eye movements of normal subjects in a moving-target task. Aviat Space Environ Med 47: 1165–1167
Barnes GR (1979) Vestibulo-ocular function during co-ordinated head and eye movements to acquire visual targets. J Physiol (Lond) 287: 127–147
Backer W (1989) Metrics, In: Wurtz RH, Golberg ME (eds) The neurobiology of saccadic eye movements. Elsevier, Amsterdam, pp 13–67
Becker W, Jurgens R (1992) Gaze saccades to visual targets: does head movement change the metrics. In: Berthoz A, Graf W, Vidal P-P (eds) Head-neck sensory-motor system. Oxford University Press, Oxford, pp 427–433
Biguer B, Prablanc C, Jeannerod M (1984) The contribution of coordinated eye and head movements in hand pointing accuracy. Exp Brain Res 55: 462–469
Frost D, Poppel E (1976) Different programming modes of human saccadic eye movements as a function of stimulus eccentricity: indications of a functional subdivision of the visual field. Biol Cybern 23: 39–48
Fuller JH (1992a) Comparison of head movement strategy among mammals. In: Berthoz A, Graf W, Vidal P-P (eds) Head-neck sensory-motor system. Oxford University Press, Oxford, pp 101–112
Fuller JH (1992b) Head movement propensity. Exp Brain Res 92: 152–164
Fuller JH (1994) Ocular orbital position effects on saccadic amplitude/latency relation. Soc Neurosci Abstr 20: 234
Fuller JH, Maldonado H, Schlag J (1983) Vestibular-oculomotor interaction in cat eye-head movements. Brain Res 271: 241–250
Guitton D, Volle M (1987) Gaze control in humans: eye head coordination during orienting movements to targets within and beyond the oculomotor range. J Neurophysiol 58: 427–459
Hallet PE, Lightstone AD (1976) Saccadic eye movements towards stimuli triggered by prior saccades. Vision Res 16: 99–106
Laurutis VP, Robinson DA (1986) The vestibulo-ocular reflex during human saccadic eye movement. J Physiol (Lond) 373: 209–233
McIlwain JT (1986) Effects of eye position on saccades evoked electrically from superior colliculus of alert cats. J Neurophysiol 55: 97–112
Pelisson D, Prablanc C, Urquizar C (1988) Vestibuloocular reflex inhibition and gaze saccade control characteristics during eyehead orientation in humans. J Neurophysiol 59: 997–1013
Ron S, Berthoz A (1991) Eye and head coupled and dissociated movements during orientation to a double step visual target displacement. Exp Brain Res 85: 196–207
Thickbroom GW, Mastaglia FL (1985) Presaccadic “spike” potential: investigation of topography and source. Brain Res 339: 279–280
Tusa RJ, Becker JL (1989) Are saccade latencies based on the craniotopic location of the target? Soc Neurosci Abstr 15: 475.13
Zahn JR, Abel LA, Dell'Osso LF (1978) Audio-ocular response characteristics. Sens Processes 2: 32–37
Zambarbieri D, Schmid R, Magenes G, Prablanc C (1982) Saccadic responses evoked by presentation of visual and auditory targets. Exp Brain Res 47: 417–427
Zambarbieri D, Beltrami G, Versino M (1995) Saccade latency toward auditory targets depends on the relative position of the sound source relative to the eyes. Vision Res 35: 3305–3312
Zangemeister WH, Stark L (1981) Active head rotations and eyehead coordination. Ann NY Acad Sci 374: 540–559
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fuller, J.H. Eye position and target amplitude effects on human visual saccadic latencies. Exp Brain Res 109, 457–466 (1996). https://doi.org/10.1007/BF00229630
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00229630