Abstract
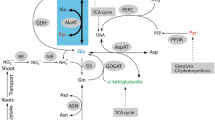
The aspartate-derived amino-acid pathway leads to the production of the essential amino-acids lysine, methionine, threonine and isoleucine. Aspartate kinase (AK) is the first enzyme in this pathway and exists in isoforms that are feedback inhibited by lysine and threonine. Two maize (Zea mays L.) threonine-overproducing, lysine-insensitive AK mutants (Ask1-LT19 and Ask2-LT20) were previously isolated. The present study was conducted to determine the map location of Ask2 and to examine the amino-acid profiles of the Ask mutants. The threonine-overproducing trait conferred by Ask2-LT20 was mapped to the long arm of chromosome 2. Both mutants exhibited increased free threonine concentrations (nmol/mg dry weight) over wild-type. The percent free threonine increased from approximately 2% in wild-type kernels to 37–54% of the total free amino-acid pool in homozygous mutant kernels. Free methionine concentrations also increased significantly in homozygous mutants. Free lysine concentrations were increased but to a much lesser extent than threonine or methionine. In contrast to previous studies, free aspartate concentrations were observed to decrease, indicating a possible limiting factor in threonine synthesis. Total (free plus protein-bound) amino-acid analyses demonstrated a consistent, significant increase in threonine, methionine and lysine concentrations in the homozygous mutants. Significant increases in protein-bound (total minus free) threonine, methionine and lysine were observed in the Ask mutants, indicating adequate protein sinks to incorporate the increased free amino-acid concentrations. Total amino-acid contents (nmol/kernel) were approximately the same for mutant and wild-type kernels. In five inbred lines both Ask mutations conferred the threonine-overproducing phenotype, indicating high expressivity in different genetic backgrounds. These analyses are discussed in the context of the regulation of the aspartate-derived amino-acid pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Azevedo RA, Arana JL, Arruda P (1990) Biochemical genetics of the interaction of the lysine plus threonine-resistant mutant Ltr*1 with opaque-2 maize mutant. Plant Sci 70:81–90
Azevedo RA, Smith RJ, Lea PJ (1992) Aspartate kinase regulation in maize: evidence for co-purification of threonine-sensitive aspartate kinase and homoserine dehydrogenase. Phytochemistry 31:373–374
Bright SWJ, Kueh JSH, Franklin J, Rognes SE, Miflin BJ (1982 a) Two genes for threonine accumulation in barley seeds. Nature 299:278–279
Bright SWJ, Miflin BJ, Rognes SE (1982 b) Threonine accumulation in the seeds of a barley mutant with an altered aspartate kinase. Biochem Genet 20:229–243
Bryan JK (1990 a) Advances in the biochemistry of amino-acid biosynthesis. In: Miflin BJ, Lea PJ (eds) The biochemistry of plants, vol. 16. Intermediary nitrogen metabolism. Academic Press, New York, pp 161–195
Bryan JK (1990 b) Differential regulation of maize homoserine dehydrogenase under physiological conditions. Plant Physiol 92:785–791
Bryan JK, Lissik EA, Matthews BF (1977) Changes in enzyme regulation during growth of maize. III. Intracellular localization of homoserine dehydrogenase in chloroplasts. Plant Physiol 59:673–679
Bourgin JP, Chupeau MC, Missonier C (1982) Amino-acid-resistant plants from tobacco cells selected in vitro. In: Earle ED, Demarly Y (eds) Variability in plants regenerated from tissue culture. Praeger, New York, pp 163–174
Cattoir-Reynaerts A, Degryse E, Verbruggen I, Jacobs M (1983) Selection and characterisation of carrot embyroid cultures resistant to inhibition by lysine plus threonine. Biochem Physiol Pflanzen 178:81–90
Cohen GN, Saint-Girons I (1987) Biosynthesis of threonine, lysine and methionine. In: Neidhardt FC (ed) Escherichia coli and Salmonella typhimirium: cellular and molecular biology. American Society for Microbiology, Washington, D.C., pp 429–444
Diedrick TJ, Frisch DA, Gengenbach BG (1990) Tissue culture isolation of a second mutant locus for increased threonine accumulation in maize. Theor Appl Genet 79:209–215
Dotson SB, Frisch DA, Somers DA, Gengenbach BG (1990) Lysineinsensitive aspartate kinase in two threonine-overproducing mutants in maize. Planta 182:546–552
Feinberg AP, Vogelstein B (1983) A technique for radiolabelling DNA restriction endonuclease fragments to high specific acitivity. Anal Biochem 132:6–13
Frankard V, Ghislain M, Negrutiu I, Jacobs M (1991) High-threonine-producer mutant of Nicotiana sylvestris (Spegg. and Comes). Theor Appl Genet 82:273–282
Frankard V, Ghislain M, Jacobs M (1992) Two feedback-insensitive enzymes of the aspartate pathway in Nicotiana sylvestris. Plant Physiol 99:1285–1293
Frisch DA, Gengenbach BG, Tommey AM, Sellner JM, Somers DA, Myers DE (1991) Isolation and characterization of dihydrodipicolinate synthase from maize. Plant Physiol 96:444–452
Gardiner JM, Coe EH, Melia-Hancock S, Hoisington DA, Chao S (1993) Development of a core RFLP map in maize using an immortalized F2 population. Genetics 134:917–930
Ghislain M, Frankard V, Jacobs M (1990) Dihydrodipicolinate synthase of Nicotiana sylvestris, a chloroplast-localized enzyme of the lysine pathway. Planta 180:480–486
Giovanelli J, Mudd SH, Datko A (1989) Aspartokinase of Lemna paucicostata Hegelm. 6746. Plant Physiol 90:1577–1583
Hibberd KA, Green CE (1982) Inheritance and expression of lysine plus threonine resistance selected in maize tissue culture. Proc Natl Acad Sci USA 79:559–563
Hibberd KA, Walter T, Green CE, Gengenbach BG (1980) Selection and characterization of a feedback-insensitive tissue culture of maize. Planta 148:183–187
Karchi H, Shaul O, Galili G (1993) Seed-specific expression of a bacterial desensitized aspartate kinase increases the production of seed threonine and methionine in transgenic tobacco. The Plant J 3:721–727
Knecht R, Chang J-Y (1986) Liquid Chromatographic determination of amino-acids after gas-phase hydrolysis and derivitization with (Dimethylamino) azobenzenesulfonyl chloride. Anal Chem 58:2375–2379
Krishnaswamy S, Bryan JK (1983 a) Ligand-induced interconversions of maize homoserine dehydrogenase among different states. Arch Biochem Biophys 222:449–463
Krishnaswamy S, Bryan JK (1983 b) Characterization of ligand-induced states of maize homoserine dehydrogenase. Arch Biochem Biophys 227:210–224
Madison JT, Thompson JF (1976) Threonine synthetase from higher plants: stimulation by S-adenosylmethionine and inhibition by cysteine. Biochem Biophys Res Commun 71:684–691
Miao S, Duncan DR, Widholm JM (1988) Selection of regenerable maize callus cultures resistant to 5-methyl-DL-tryptophan, S-2- aminoethyl-L-cysteine and high levels of L-lysine plus L-threonine. Plant Cell Tiss Org Cult 14:3–14
Negrutiu I, Cattoir-Reynearts A, Verbruggen I, Jacobs M (1984) Lysine overproducer mutants with an altered dihydrodipicolinate synthase from protoplast culture of Nicotiana sylvestris (Spegazzini and Comes). Theor Appl Genet 68:11–20
Perl A, Shaul O, Galili G (1992) Regulation of lysine synthesis in transgenic potato plants expressing a bacterial dihydrodipicolinate synthase in their chloroplasts. Plant Mol Biol 19:815–823
Rognes SE, Lea PJ, Miflin BJ (1980) S-adenosylmethionine -a novel regulator of aspartate kinase. Nature 287:357–359
Rognes SE, Bright SWJ, Miflin BJ (1983) Feedback-insensitive aspartate kinase isoenzymes in barley mutants resistant to lysine plus threonine. Planta 157:32–38
Saghai-Maroof MA, Soliman KM, Jorgensen RA, Allard RW (1984) Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, and population dynamics. Proc Natl Acad Sci USA 81:8014–8019
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Laboratory, Cold Spring Harbor, New York
Shaul O, Galili G (1992 a) Threonine overproduction in transgenic tobacco plants expressing a mutant desensitized aspartate kinase of Escherichia coli. Plant Physiol 100:1157–1163
Shaul O, Galili G (1992 b) Increased lysine synthesis in tobacco plants that express high levels of bacterial dihydrodipicolinate synthase in their chloroplasts. The Plant J 2:203–209
Suiter KA, Wendell JF, Case JS (1983) Linkage-1. J Hered 74:203–204
Thoen A, Rognes SE, Aarnes H (1978) Biosynthesis of threonine from homoserine in pea seedlings. II. Threonine synthase. Plant Sci Lett 13:113–119
Wallsgrove RM, Lea PJ, Miflin BJ (1983) Intracellular localization of aspartate kinase and the enzymes of threonine and methionine biosynthesis in green leaves. Plant Physiol 71:780–784
Walter TJ, Connelly JA, Gengenbach BG, Wold F (1979) Isolation and characterization of two homoserine dehydrogenases from maize suspension cultures. J Biol Chem 254:1349–1355
Weisemann JM, Matthews BF (1993) Identification and expression of a cDNA from Daucus carota encoding a bifunctional aspartokinase-homoserine dehydrogenase. Plant Mol Biol 22:301–312
Wilson BJ, Gray AC, Matthews BF (1991) Bifunctional protein in carrot contains both aspartate kinse and homoserine dehydrogenase activities. Plant Physiol 97:1323–1328
Author information
Authors and Affiliations
Additional information
Communicated by F. Salamini
Rights and permissions
About this article
Cite this article
Muehlbauer, G.J., Gengenbach, B.G., Somers, D.A. et al. Genetic and amino-acid analysis of two maize threonine-overproducing, lysine-insensitive aspartate kinase mutants. Theoret. Appl. Genetics 89, 767–774 (1994). https://doi.org/10.1007/BF00223717
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00223717