Abstract
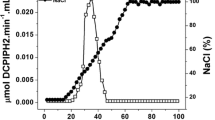
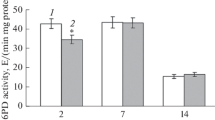
Investigations into physiological aspects of glycerol conversion to dihydroxyacetone (DHA) by Gluconobacter oxydans ATCC 621 were made. The activity levels of the enzymes involved in the three catabolic pathways previously known and the effects of specific inhibitors and uncoupling agents on cellular development, DHA synthesis, and cellular respiratory activity were determined. It was established that only two catabolic pathways are involved in glycerol dissimilation by this micro-organism. The only enzyme responsible for DHA production is membrane-bound glycerol dehydrogenase, which employs oxygen as the final acceptor of reduced equivalents without NADH mediation. The ketone is directly released into the culture broth. As the glycolytic and carboxylic acid pathways are absent, the pathway provided by the membrane-bound enzyme is indispensable for the energy requirements of G. oxydans. The cytoplasmic pathway, which begins by phosphorylation of glycerol followed by a dehydrogenation to dihydroxyacetone phosphate, allows growth of the bacterium. At the same time, the substrate transport mode was characterized as facilitated diffusion using radioactive [1(3)-3H]-glycerol. Concerning the DHA inhibition of microbial activity, the enzymatic study of the membrane-bound glycerol dehydrogenase showed the enzymatic origin of this phenomenon: a 50% decrease of the enzyme activity was observed in the presence of 576 mm DHA. The decrease in the rate of penetration of glycerol into cells in the presence of DHA indicates that growth inhibition is essentially due to the high inhibition exerted by the ketone on the substrate transport system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ameyama M, Matsushita K, Ohno O, Shinagawa E, Adachi O (1981) Existence of a novel prosthetic group PQQ, in membrane-bound electron transport chain-linked primary dehydrogenase of oxidative bacteria. FEBS Lett 130:179–183
Ameyama M, Shinagawa E, Matasushita K, Adachi O (1985) Solubilization, purification and properties of membrane-bound glycerol dehydrogenase from Gluconobacter industrius. Agric Biol Chem 49:1001–1010
Ameyama M, Matsushita K, Shinagawa E, Adachi O (1987) Sugar-oxidizing respiratory chain of Gluconobacter suboxydans. Evidence for a branched respiratory chain and characterization of respiratory chain-linked cytochromes. Agric Biol Chem 51:2943–295
Battle JL, Collon Y (1979) Dosage enzymatique en flux continu du glycérol dans les vins. Connaiss Vign Vin 1:45–50
Blazejak S, Sobcjak E (1988) Bioconversion of glycerol into dihydroxyacetone DHA using Gluconobacter xylinum. Acta Aliment Pol 14:207–216
Bobin MF, Martini MC, Cotte J (1984) Effect of color adjuvants on the tanning effect of dihydroxyacetone. J Soc Cosmet Chem 35:265–272
Bories A, Claret C, Soucaille P (1991) Kinetic study and optimisation of the production of dihydroxyacetone from glycerol using Gluconobacter oxydans. Process Biochem 26:243–248
Calmes D, Deal R (1972) Glycerol transport by Nocardia asteroïdes. Can J Microbiol 18:1703–1708
Claret C, Bories A, Soucaille P (1992) Glycerol inhibition of growth and dihydroxyacetone production by Gluconobacter oxydans. Curr Microbiol 25:149–155
Claret C, Bories A, Soucaille P (1993) Inhibitory effect of dihydroxyacetone on Gluconobacter oxydans. Kinetic aspects and expression by mathematical equations. J Ind Microbiol 11:105–112
De Ley J, Dochy R (1960) On the localisation of oxidase system in Acetobacter cells. Biochim Biophys Acta 40:277–289
De Ley J, Kersters K (1964) Oxidation of aliphatic glycols by acetic acid bacteria. Bacteriol Rev 28:164–180
Hauge J, King T, Cheldelin V (1955a) Alternate conversions of glycerol to dihydroxyacetone in Acetobacter suboxydans. J Biol Chem 214:1–9
Hauge J, King T, Cheldelin V (1955b) Oxidation of dihydroxyacetone via the pentoses cycle in Acetobacter suboxydans. J Biol Chem 214:11–26
Kersters K, De Ley J (1966) Polyols dehydrogenases of Gluconobacter. Methods Enzymol 9:170–177
King T, Cheldelin V (1952a) Phosphorylative and non phosphorylative oxidation in Acetobacter suboxydans. J Biol Chem 198:135–141
King T, Cheldelin V (1952b) Source of energy and the dinitrophenol effect in the growth in Acetobacter suboxydans. J Bacteriol 66:581–584
Matsushita K, Shinagawa E, Adachi O, Ameyama M (1987) Purification, characterization and reconstitution of cytochrome o-type oxidase from Gluconobacter suboxydans. Biochim Biophys Acta 394:304–312
Meybeck A (1977) A spectroscopic study of reaction products of dihydroxyacetone with aminoacids. J Soc Cosmet Chem 28:25–35
Reus M, Josic D, Popovic M, Brown W (1979) Viscosity of yeast suspension. Eur J Appl Microbiol Biotechnol 8:165–175
Richey D, Lin E (1972) Importance of facilitated diffusion for effective utilization of glycerol by Escherichia coli. J Bacteriol 112:784–790
Saheb S (1972) Perméation du glycérol et sporulation chez Bacillus subtilis. Can J Microbiol 18:1307–1313
Sanno Y, Wilson T, Lin E (1968) Control of permeation to glycerol in cells of Escherichia coli. Biochem Biophys Res Commun 32:344–349
Williams P, Rainbow C (1964) Enzymes of the tricarboxylic acids cycle in acetic acid bacteria. J Gen Microbiol 35:237–247
Yamada Y, Aida K, Uemura T (1969) Enzymatic studies on the oxidation of sugar and sugar alcohol: V. Ubiquinone of acetic acid bacteria and its relation to classification genera Gluconobacter and Acetobacter, especially of the so-called intermediate strains. J Gen Appl Microbiol 15:181–196
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Claret, C., Salmon, J.M., Romieu, C. et al. Physiology of Gluconobacter oxydans during dihydroxyacetone production from glycerol. Appl Microbiol Biotechnol 41, 359–365 (1994). https://doi.org/10.1007/BF00221232
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00221232