Abstract
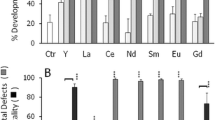
Bioassays were developed using sperm of a sea urchin (Echinometra mathaei), and sperm, embryos, and larvae of a bivalve species (Isognomon californicum). Sea urchin spawning was restricted to only a few months of the year and viability of sperm throughout the year varied from 5 to 75%. Sea urchin fertilization assays were affected by temporal variation in sperm viability. Spawning in this bivalve species occurs year-round and there is little temporal variation in sperm viability. Since stringent sperm:egg ratios are not required for the bivalve embryo and larval assays, these were less affected by variation in gamete quality. The relative sensitivity of the various assays were compared during exposure to three different metal pollutants: cadmium, copper, and tributyltin. Gametes and embryos were relatively resistant to cadmium toxicity, but larvae were very sensitive. With copper and tributyltin, sea urchin and bivalve fertilization assays were the least sensitive; and bivalve growth assays were the most sensitive, followed closely by the bivalve embryo assays. On the basis of sensitivity, ease and time required to conduct the assay, and salinity tolerance, the bivalve embryo assay was recommended as the overall single most reliable toxicity bioassay. However a multispecies, multidimensional approach using sperm fertilization assays as well as embryo assays should be employed, perhaps in a hierarchal manner. Larval growth assays were deemed too tedious and time consuming to be used routinely, but due to their high sensitivity, should still be considered as a valuable comparative tool.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Akberali HB, Trueman ER (1985) Effects of environmental stress on marine bivalve molluscs. Adv Mar Biol 22:101–198
ASTM (1985) Conducting static acute toxicity tests with larvae of four species of bivalve molluscs. In: Annual book of ASTM standards, Vol 11.04. E 724-80, pp 256–271
Beaumont AR, Budd MD (1984) High mortality of the larvae of the common mussel at low concentrations of tributyltin. Mar Pollut Bull 15:402–405
Calabrese A, Collier RS, Nelson DA, MacInnes JR (1973) The toxicity of heavy metals to embryos of the American oyster Crassostrea virginica. Mar Biol 18:162–166
Connor PM (1972) Acute toxicity of heavy metals to some marine larvae. Mar Pollut Bull 3:190–192
Coombs TL (1979) Cadmium in aquatic organisms. In: Webb M (ed) The chemistry, biochemistry, and biology of cadmium, Elsevier, North-Holland
Dinnel PA (1988) Adaptation of the sperm/fertilization bioassay protocol to Hawaiian sea urchin species. Final Report for State of Hawaii Department of Health, Honolulu, Hawaii, Contract # 88-405
Dinnel PA, Link JM, Stober QJ (1987) Improved methodology for a sea urchin sperm cell bioassay for marine waters. Arch Environ Contam Toxicol 16:23–32
Dinnel PA, Link JM, Stober QJ, Letourneau MW, Roberts WE (1989) Comparative sensitivity of sea urchin sperm bioassays to metals and pesticides. Arch Environ Contam Toxicol 18:748–755
Fasset DW (1980) Cadmium. In: Waldron HA (ed) Metals in the environment, Academic Press, London, pp 61–110
Goldberg ED (1986) TBT An environmental dilemma. Environment 28:17–20, 42–44
Holmes CW, Slade EA, McLerran CJ (1974) Migration and redistribution of zinc and cadmium in marine estuarine systems. Environ Sci Technol 8:255–259
Kelso DP (1970) A comparative morphological and ecological study of two species of the sea urchin genus Echinometra in Hawaii. PhD dissertation, Univ of Hawaii, Honolulu, Hawaii, 112 pp
Lee HH, Xu CH (1984) Effects of metals on sea urchin development: A rapid bioassay. Mar Pollut Bull 15:18–21
Martin M, Osborn KE, Billig P, Glickstein N (1981) Toxicities of ten metals to Crassostrea gigas and Mytilus edulis embryos and Cancer magister larvae. Mar Pollut Bull 12:305–308
Middaugh DP, Dean JM (1977) Comparative sensitivity of eggs, larvae and adults of the estuarine teleosts, Fundulus heteroclitus and Menidia menidia, to cadmium. Bull Environ Contam Toxicol 17:645–652
Nacci DE, Jackim E, Walsh R (1986) Comparative evaluation of three rapid marine toxicity tests: sea urchin early embryo growth test, sea urchin sperm cell toxicity test and Microtox. Environ Toxicol Chem 5:521–525
Nelson DA, Miller JE, Calabrese A (1988) Effect of heavy metals on bay scallops, surf clams, and blue mussels in acute and long-term exposures. Arch Environ Contam Toxicol 17:595–600
Pagano G, Esposito A, Giordano GG (1982) Fertilization and larval development in sea urchins following exposure of gametes and embryos to cadmium. Arch Environ Contam Toxicol 11:47–55
Phillips DJH (1977) The use of biological indicator organisms to monitor metal pollution in marine and estuarine environments—A review. Environ Pollut 13:281–317
Ringwood AH (1988) The effects of cadmium on larvae and adults of an Hawaiian bivalve, Isognomon californicum. PhD dissertation, Univ of Hawaii, Honolulu, Hawaii
— (1990) The relative sensitivities of different life stages of Isognomon californicum to cadmium toxicity. Arch Environ Contam Toxicol 19:338–340
Stebbing ARD (1985) Organotins and water quality—some lessons to be learned. Mar Pollut Bull 16:383–390
Waldhauer R, Matte A, Tucker RE (1978) Lead and copper in the waters of Raritan and lower New York Bays. Mar Pollut Bull 9:38–42
Watling HR (1982) Comparative study of the effects of zinc, cadmium, and copper on the larval growth of three oyster species. Bull Environ Contam Toxicol 28:195–201
Woelke CE (1972) Development of a receiving water quality bioassay criterion based on the 48-hour Pacific oyster, Crassostrea gigas embryo. Washington Dept Fish Tech Rep 9:1–93
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ringwood, A.H. Comparative sensitivity of gametes and early developmental stages of a sea urchin species (Echinometra mathaei) and a bivalve species (Isognomon californicum) during metal exposures. Arch. Environ. Contam. Toxicol. 22, 288–295 (1992). https://doi.org/10.1007/BF00212088
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00212088