Abstract
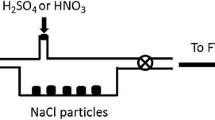
A theoretical model describing the general interaction between atmospheric trace gases, such as S02, NH3, C02 and 02, chemical reactant gaseous product H2SO4 and hydrometeors containing NaCl is proposed to study a possible mechanism for HCl production in non-precipitating cloud and the determination of the pH value of cloud droplets.
Four different cloud droplet distributions have been used to estimate the upper limit of the amount of gaseous HCl released into the atmosphere resulting from the evaporation of cloud droplets. It is shown that the acid production and the amount of HCl released depend on the following factors: (a) the temperature of the cloud; (b) the oxidation rates; (c) the ambient concentration of SO2, NH3, and H2SO4; (d) the life cycle of the cloud; and (e) the liquid content of the cloud.
This proposed chemical model also predicts a pH value spectrum depending on the cloud droplet distribution. Field measurements for the dependence of pH value on particle size and spatial distribution of gaseous HCl are recommended.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Junge, C. E.: 1963, Air Chemistry and Radioactivity, Academic Press, New York and London.
Rossby, C. G. and Egner, H.: 1955, Tellus VII, 118.
Stolarski, R. S. and Cicerone, R. J.: 1974, Canadian J. Chem. 52, 1610.
Duce, R. A.: 1969, J. Geophys. Res. 74, 4597.
Cauer, H.: 1938, Der Balneologes 5, 409.
Kohler, H. and Bath, M.: 1953, Nova acta Regiol Societatics Scient, Upsaliensis 15, 7, 24.
Yost, D. M. and Russell, H., Jr.: 1944, Systematic Inorganic Chemistry, Prentice Hall, inc., New York, 42.
Robbins, R. C., Cadle, R. D., and Eckhardt, D. L.: 1959, J. of Meteorology 16, 53.
Davis, D. D.: 1974, Can. J. Chem. 52, 1405.
Cadle, R. D.: 1972, J. Colloid Interface Sci. 39, 25.
Scott, W. D. and Hobbs, P. V.: 1967, J. Atmos. Sci. 24, 54.
Eastman, R. C. and Hobbs, P. V.: 1974, J. Atmos. Sci. 31, 1586
McKay, H. A. G.: 1971, Atmospheric Environment 5, 7.
Van der Heuval, A. P. and Mason, B. J.: 1963, Quart. J. R. Meteor. Soc. 89, 271.
Fuller, E. C. and Crist, R. H.: 1941, J. Am. Chem. Soc. 63, 1644.
Miller, J. M. and de Pena, R. G.: 1972, J. Geophys. Res. 77, 5905.
Castleman, A. W., Jr., Davis, R. E., Munkelwitz, H. R., Tang, I. N., and Wood, W. P.: 1976, J. of Chem. Kinetics, to be published.
Handbook of Geophysics: 1961, Macmillan, New York.
Whitley, K. T., Clark, W. E., Marple, V. A., Sverdrup, G. M., Sem, G. J., Willeke, K., Liu, B. Y. H., and Pui, D. Y. H.: 1972, Aerosols and Atmospheric Chemistry, Academic Press, New York.
Arnason, G. and Greenfield, R. S.: 1972, J. Atmos. Sci. 29, 342.
Woodcock, A. H.: 1957, Tellus 9, 521.
Eriksson, E.: 1959, Tellus II, 375.
Mordy, W.: 1959, Tellus II, 16.
Warner, J.: 1959, J. Atmos. Sci. 26, 1272.
Junge, C. E.: 1951, Ber. Deut. Wetterdienstes 35, 261.
Fletch, N. H.: 1952, The Physics of Rain Clouds, Cambridge University Press, 59.
Paltridge, G. W.: 1974, J. Atmos. Sci. 31, 1571.
Author information
Authors and Affiliations
Additional information
The National Center for Atmospheric Research is sponsored by the National Science Foundation.
Rights and permissions
About this article
Cite this article
Yue, G.K., Mohnen, V.A. & Kiang, C.S. A mechanism for hydrochloric acid production in cloud. Water, Air, and Soil Pollution 6, 277–294 (1976). https://doi.org/10.1007/BF00182870
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00182870