Summary
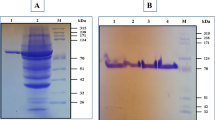
A pullulan-hydrolysing enzyme of Micrococcus sp. 207 was purified to an electrophoretically homogenous state by chromatography on DEAE-Toyopearl, α-cyclodextrin-Sepharose and Asahipak GS-520P. The purified enzyme was free of α-amylase activity. The molecular weight of the enzyme as estimated by SDS-PAGE was 120,000 and the pI value as determined by isoelectric focusing was 4.9. The enzyme was most active at pH 8.0 and 50°C. The enzyme was activated by the addition of CaCl2, but its thermoresistance increased after removing free Ca2+ ions. The enzyme could hydrolyse the α-1,6-linkages of amylopectins, glycogens and pullulan and the K m value for pullulan was about 0.018%. Pullulan at concentrations above 0.012% inhibited the enzyme activity and the activity was competitively inhibited by cyclodextrins.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abdullah M, French D (1966) Reversible action of pullulanase. Nature 210:200
Bender H, Wallenfels K (1961) Untersuchungen an Pullulan. II. Spezifischer Abbau durch ein bakterielles Enzym. Biochem Z 334:79–94
Dixon M, Webb EC (1979) Enzymes, 3rd edn. Longman, London, pp 350–368
Gunja-Smith Z, Marshall JJ, Smith EE, Whelan WJ (1970) A glycogen-debranching enzyme from Cytophaga. FEBS Lett 12:96–100
Hyun HH, Zeikus JG (1985) Biochemical characterization of thermostable extracellular pullulanase and glucoamylase from Clostridium thermohydrosulfuricum. Appl Environ Microbiol 49:1168–1173
Kimura T, Horikoshi K (1988) Isolation of bacteria which can grow at both high pH and low temperature. Appl Environ Microbiol 54:1066–1067
Kimura T, Horikoshi K (1989a) Carbohydrate use by alkalopsychrotrophic bacteria. Agric Biol Chem 53:2019–2020
Kimura T, Horikoshi K (1989c) Production of amylase and pullulanase by an alkalopsychrotrophic Micrococcus sp. Agric Biol Chem 53:2963–2968
Kimura T, Horikoshi K (1989c) Purification and characterization of α-amylases of an alkalopsychrotrophic Micrococcus sp.(in Japanese). Nippon Nôgeikagaku Kaishi 63:380
Marshall JJ (1973) Inhibition of pullulanase by schardinger dextrins. FEBS Lett 37:269–273
Nakamura N, Watanabe K, Horikoshi K (1975) Purification and some properties of alkaline pullulanase from a strain of Bacillus no. 202–1, an alkalophilic microorganism. Biochim Biophys Acta 397:188–193
Nakamura N, Sashihara N, Nagayama H, Horikoshi K (1989) Characterization of pullulanase and α-amylase activities of a Thermus sp. AMD 33. Starch 41:112–117
Plant AR, Morgan HW, Daniel RM (1986) A highly stable pullulanase from Thermus aquaticus YT-1. Enzyme Microb Technol 8:668–672
Robyt JF, Whelan WJ (1972) Reducing value methods for maltodextrins: 1. Chain-length dependence of alkaline 3,5-dinitrosalicylate and chain-length independence of alkaline copper. Anal Biochem 45:510–516
Sakai S (1981) Production and usage of maltose (in Japanese). J Jpn Soc Starch Sci 28:72–78
Takasaki Y (1976) Purifications and enzymatic properties of ß-amylase and pullulanase from Bacillus cereus var. mycoides. Agric Biol Chem 40:1523–1530
Ueda S, Nanri J (1967) Production of isoamylase by Escherichia intermedia. Appl Microbiol 15:492–496
Walker GJ (1968) Metabolism of the reserve polysaccharide of Streptococcus mitis: some properties of a pullulanase. Biochem J 108:33–40
Yagisawa M, Kato K, Koba Y, Ueda S (1972) Pullulanase of Streptomyces sp. no. 280. J Ferment Technol 50:572–579
Yokobayashi K, Misaki A, Harada T (1970) Purification and properties of Pseudomonas isoamylase. Biochim Biophys Acta 212:458–469
Author information
Authors and Affiliations
Additional information
Offprint requests to: K. Horikoshi
Rights and permissions
About this article
Cite this article
Kimura, T., Horikoshi, K. Characterization of pullulan-hydrolysing enzyme from an alkalopsychrotrophic Micrococcus sp.. Appl Microbiol Biotechnol 34, 52–56 (1990). https://doi.org/10.1007/BF00170923
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170923