Abstract
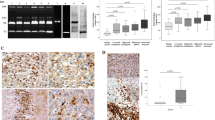
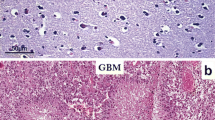
Matrix metalloproteinases play an important regulatory role in tissue morphogenesis, cell differentiation and motility, and tumor cell invasiveness. We have recently demonstrated elevated activity of the 92 kDa type IV collagenase (MMP-9) in human glioblastoma and in the present study examine the relative amounts of MMP-9 protein and mRNA in human gliomas and as well as the distribution of MMP-9 in human glioma tumors in vivo. Using an enzyme-linked immunosorbent assay for the quantitative determination of MMP-9 protein, we found that levels were significantly higher in malignant astrocytomas, especially in glioblastoma multiforme, than in normal brain tissues and low-grade gliomas. In addition, the amount of MMP-9 mRNA, as determined by northern blot analysis was higher in anaplastic astrocytomas and glioblastoma multiforme than in normal brain tissue and low-grade gliomas. Immunocytochemical staining for MMP-9 showed strong cytoplasmic immunoreactivity in the tumor cells and the proliferating endothelial cells of glioblastoma multiforme and anaplastic astrocytomas. The staining intensity was lower in low-grade astrocytomas, and was undetectable or very low in normal brain astrocytes. The results indicate that expression of MMP-9 is dramatically upregulated in highly malignant gliomas and correlates with the highly malignant progression of human gliomas in vivo, and support a role for the MMP-9 in facilitating the invasiveness seen in malignant gliomas in vivo.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bjerkvig R, Laerum OD and Mella O, 1986, Glioma cell interactions with fetal rat brain aggregates in vitro and with brain tissue in vivo. Cancer Res, 46, 4071–9.
Russel DC and Rubinstain LJ, 1989, Pathology of Tumors of the Nervous System, 5th ed. London: Edward Arnold.
Gleave JR, 1986, Surgery for primary brain tumors. In: Blehen NM, ed. Tumors of the Brain. Berlin: Springer-Verlag, pp. 101–120.
Liotta LA, 1984, Tumor invasion and metastases: role of the basement membrane. Am J Pathol, 117, 339–48.
Kwaan HC, 1992, The plasminogen-plasmin system in malignancy. Cancer Metastasis Rev, 11, 291–311.
Goldfarb RH and Liotta LA, 1986, Proteolytic enzymes in cancer invasion and metastasis. Semin Thromb Hemostasis, 12, 294–307.
Liotta LA, Steeg PS and Stetler-Stevenson WG, 1991, Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell, 64, 327–36.
Woessner JF, 1991, Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J, 5, 2145–54.
Nagase H, Ogata Y, Suzuki K, Enghild JJ and Salvensen G, 1991, Substrate specificities and activation mechanisms of matrix metalloproteinases. Biochem Soc Trans, 19, 715–8.
Sato H and Seiki M, 1993, Regulatory mechanism of 92-kDa type IV collagenase gene expression which is associated with invasiveness of tumor cells. Oncogene, 8, 395–405.
Stetler-Stevenson WG, Krutzsch HC and Liotta LA, 1989, Tissue inhibition of metalloproteinase (TIMP-2). A new member of the metallproteinase inhibitor family. J Biol Chem, 264, 17374–8.
Hibbs MS, Hoidal JR and Kang AH, 1987, Expression of a metalloproteinase that degrades native type V collagen and denatured collagens by cultured human alveolar macrophages. J Clin Invest 80, 1644–50.
Liotta LA and Stetler-Stevenson WG, 1990, Metalloproteinases and cancer invasion. Semin Cancer Biology, 1, 99–106.
Schultz RM, Silberman S, Persky B, Bahkoski AS and Carmichael DF, 1988, Inhibition by human recombinant tissue inhibitor of metallproteinases of human amnion invasion and lung colonization by murine B16-F10 melanoma cells. Cancer Res, 48, 5539–45.
Wang M and Stearns ME, 1988, Blocking of collagenase secretion by estramustine during in vitro tumor cell invasion. Cancer Res, 48, 6262–71.
Paganetti PA, Caroni P and Schwab ME, 1988, Glioblastoma infiltration into central nervous system tissue in vitro: involvement of a metalloproteinase. J Cell Biol, 107, 2281–91.
Vaithilingam IS, McDonald W, Stroude EC, Cook RA and Del Maestro RF, 1992, Proteolytic activity during the growth of C6 astrocytoma in the murine spheroid implantation model. Can J Neurol Sci, 19, 17–22.
Apodaca G, Rutka JT, Bouhana K, et al. 1990, Expression of metalloproteinases and metalloproteinase inhibitors by fetal astrocytes and glioma cells. Cancer Res, 50, 2322–9.
Rao JS, Steck PA, Mohanam S, Stetler-Stevenson WG, Liotta LA and Sawaya R, 1993, Elevated levels of M, 92,000 type IV collagenase in human brain tumors. Cancer Res, 53, 2208–11.
Rao JS, Steck PA, Tofilon P, et al. 1994, Role of plasminogen activator and of 92-kDa type 1 IV collagenase in glioblastoma invasion using an in vitro matrigel model. J Neuro-Oncol, 18, 129–38.
Kleihus P, Burger PC and Schneithauer BW, 1993, The new WHO classification of brain tumours. Brain Pathol, 3, 255–68.
Nakajima M, Welch DR, Belloni PN and Nicolson GL, 1987, Degradation of basement membrane type IV collagen and lung subendothelial matrix by rat mammary adenocarcinoma cell and clones of differing metastatic potentials. Cancer Res, 47, 4869–76.
Juarez J, Clayman G, Nakajima M, et al. 1993, Role and regulation of expression of 92-kDa type-IV collagenase (MMP-9) in two invasive squamous-cellcarcinoma cell lines of the oral cavity. Int J Cancer, 55, 10–18.
Chomczynski P and Sacchi N, 1987, Single-step method of RNA isolation by acid quanidinium thiocyanatephenol-chloroform extraction. Anal Biochem, 162, 156–9.
Matrisian LM, 1992, The matrix-degrading metalloproteinases. Bioessays, 14, 455–63.
Salo T, Turpeenniemi-Hujanen T and Tryggvason K, 1985, Tumor-promoting phorbol esters and cell proliferation stimulate secretion of basement membrane (type IV) collagen-degrading metalloprotease by human fibroblasts. J Biol Chem, 260, 8526–31.
Salo T, Liotta LA, Keski J, Turpeenniemi-Hujanen T and Tryggvason K, 1982, Secretion of basement membrane collagen degradation enzyme and plasminogen activator by transformed cells-role in metastasis. Int J Cancer, 30, 669–73.
Bernhard EJ, Gruber SB and Muschel RJ, 1994, Direct evidence linking expression of matrix metalloproteinase (92-kDa gelatinase/collagenase) to the metastatic phenotype in transformed rat embryo cells. Proc Natl Acad Sci USA, 91, 4293–7.
Muller D, Breathnach R, Eagelmann A, et al. 1991, Expression of collagenase-related metalloproteinase genes in human lung or head and neck tumours. Int J Cancer, 48, 550–56.
Monteagudo C, Merino MJ, San-Juan J, Liotta LA and Stetler-Stevenson WG, 1990, Immunohistochemical distribution of type IV collagenase in normal, benign and malignant breast tissue. Am J Pathol, 136, 585–92.
Hewitt RE, Leach IH, Powe DG, et al. 1991, Distribution of collagenase and tissue inhibitor of metalloproteases (TIMP) in colorectal tumors. Int J Cancer, 49, 666–72.
Nakagawa T, Kubota T, Kubota M, et al. 1994, Production of matrix metalloproteinases and tissue inhibitor of metalloproteinases-1 by human brain tumors. J Neurosurg, 81, 69–77.
Okada Y, Tsuchiya H, Shimizu H, et al. 1990, Induction and stimulation of 92-kDa gelatinase/type IV collagenase production in osteosarcoma and fibrosarcoma cell lines by tumor necrosis factor α. Biochem Biophys Res Commun, 171, 610–17.
Shima I, Sasaguri Y, Kusukawa J, et al. 1993, Production of matrix metalloproteinase 9 (92-kDa gelatinase) by human oesophageal squamous cell carcinoma in response to epidermal growth factor. Br J Cancer, 67, 721–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rao, J.S., Yamamoto, M., Mohaman, S. et al. Expression and localization of 92 kDa type IV collagenase/gelatinase B (MMP-9) in human gliomas. Clin Exp Metast 14, 12–18 (1996). https://doi.org/10.1007/BF00157681
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00157681