Summary
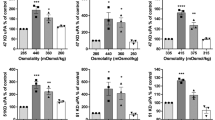
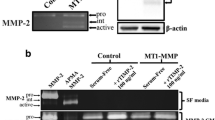
The invasive nature of human gliomas represents a major factor in preventing their total resection. The exact nature of the underlying mechanisms of tumor cell invasion are still unclear. In this study, we have quantitatively assayed a glioblastoma cell line for its ability to migrate through a polycarbonate filter coated with matrigel which contains a complex of multiple basement membrane components. At 48 h the glioblastoma cell line (U251) showed a rate of invasiveness of 42% and also dependent on the concentration of matrigel. The U251 cell line produced a urokinase type plasminogen activator and a 92-KDa type IV collagenase. Both enzymes were inhibited by the addition of uPA and 92-KDa type IV collagenase antibodies. Those same antibodies reduced the invasion rate of U251 cells from 42% to 12 and 21%, respectively. Similarly, the addition of ɛ-aminocaproic acid (a plasmin inhibitor) or tissue inhibitor of metalloprotease (TIMP2, a collagenase inhibitor) reduced the invasiveness of U251 cells from 42% to 14% and 10%, respectively. Additionally, the other two glioblastoma cell lines (LG11, UWR1) and astrocytes showed a rate of invasiveness at 41%, 61% and 12%, respectively. Finally, the addition of hyaluronic acid to the matrigel, a constituent of brain extracellular matrix, enhanced the rate of invasion. These findings provide evidence for the role of serine proteases and metalloproteases in facilitating the invasion of extracellular matrix components by glioblastoma cell line and suggest a therapeutic role for protease inhibitors in attempting to minimize the invasive propensity of gliomas.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dano K, Andreasen PA, Grondahl-Hansen J, Kristensen B, Nielsen LS, Skriver L: Plasminogen activators, tissue degradation and cancer. Adv Cancer Res 44: 146–239, 1985
Poole AR, Tiltman KJ, Recklies AD, Stoker TAM: Differences in secretion of the proteinase cathepsin B at the edges of human breast carcinomas and fibroadenomas. Nature 273: 545–547, 1978
Recklies AD, Mort JS, Poole AR: Secretion of a thiol proteinase from mouse mammary carcinomas and its characterization. Cancer Res 42: 1026–1032, 1982
Sloane BF, Honn KV, Sadler JC, Turner WA, Kimpson JJ, Taylor JD: Cathepsin B activity in B16 melanoma cells: A possible marker for metastatic potential. Cancer Res 42: 980–986, 1982
Robertson DM, Williams DC:In vitro evidence for neutral collagenase activity in an invasive mammalian tumor. Nature 221: 259–260, 1969
Dresden MH, Heilman SA, Schmidt JD: Collagenolytic enzymes in human neoplasms. Cancer Res 32: 993–996, 1972
Liotta LA, Abe S, Gehron-Robey P, Martin GR: Preferential digestion of basement membrane collagen by an enzyme derived from a metastatic murine tumor. Proc Natl Acad Sci USA 76: 2268–2272, 1979
Liotta LA, Tryggvason K, Garbisa S, Hart I, Foltz CM, Shafie S: Metastatic potential correlates with enzymatic degradation of basement membrane collagen. Nature (Lond) 284: 67–68, 1980
Eisenbach L, Segal S, Feldman M: Proteolytic enzymes in tumor metastasis. II. Collagenase type IV activity in subcellular fractions of cloned tumor cell populations. J Natl Cancer Inst 74: 87–93, 1985
Noguchi PD, Johnson JB, O'Donnel R, Petricciani JD: Chick embryonic skin as a rapid organ culture assay for cellular neoplasia. Science 199: 1980–1983, 1978
Mareel M, Kint J, Meyvisch C: Methods of study of the invasion of malignant C3H mouse fibroblasts into embryonic chick heartin vitro. Virch Arch B Cell Pathol 30: 95–111, 1979
Kramer RH, Nicolson GL: Interactions of tumor cells with vascular endothelial cell monolayers: A model for metastatic invasion. Proc Natl Acad Sci USA 76: 5704–5709, 1979
Nicolson GL: Metastatic tumor cell attachment and invasion assay utilizing vascular endothelial cell monolayers. J Histochem Cytochem 30: 214–220, 1982
Roos E, Van De Pavert IV, Middelkoop OP: Infiltration of tumor cells into cultures of isolated hepatocytes. J Cell Sci 47: 385–397, 1981
Kuettner KE, Pauli BU, Soble LW: Morphological studies on the resistance of cartilage to invasion by osteosarcoma cellsin vitro andin vivo. Cancer Res 38: 277–287, 1978
Hart IR, Fidler IJ: Anin vitro assay for tumor cell invasion. Cancer Res 38: 3218–3224, 1978
Poste G, Doll J, Hart IR, Fidler IJ:In vitro selection of murine B16 melanoma variants with enhanced tissue-invasive properties. Cancer Res 40: 1636–1644, 1980
Armstrong PB, Quigley JP, Sidebottom E: Transepithelial invasion and intramesenchymal infiltration of the chick embryo chorioallantois by tumor cell lines. Cancer Res 42: 1826–1837, 1982
Ossowski L, Reich E: Antibodies to plasminogen activator inhibit human tumor metastasis. Cell 35: 611–619, 1983
Starkey JR, Hosick HL, Stanford DR, Liggitt HD: Interaction of metastatic tumor cells with bovine lens capsule basement membrane. Cancer Res 44: 1585–1594, 1984
Liotta LA, Lee CW, Morakis DJ: New method for preparing large surfaces of intact human basement membrane for tumor invasion studies. Cancer Lett 11: 141–152, 1980
Thorgeirsson UP, Liotta LA, Kalebic T, Margulies I, Thomas K, Russo RG: Effect of natural protease inhibitors and a chemoattractant on tumor cell invasionin vitro. J Natl Cancer Inst 69: 1049–1054, 1982
Hince TA, Roscoe JP: Differences in pattern and level of plasminogen activator production between a coloned cell line from an ethylnitrosourea-induced glioma and one from normal adult rat brain. J Cell Physiol 104: 199–207, 1980
Dano K, Dabelsteen E, Nielsen DS, Kaltoft K, Wilson EL, Zeuthen J: Plasminogen-activating enzyme in cultured glioblastoma cells: An immunofluorescence study with monoclonal antibody. J Histochem Cytochem 30: 1165–1170, 1982
Sawaya R, Highsmith R: Plasminogen activator activity and molecular weight patterns in human brain tumors. J Neurosurg 68: 73–79, 1988
Sawaya R, Ramo OJ, Shi ML: Biological significance of tissue plasminogen activator content in brain tumors. J Neurosurg 76: 480–486, 1991
Rutka JT, Gibiin JR, Apodaca G, Dearmond SJ, Stern R, Rosenblum ML: Inhibition of growth and induction of differentiation in a malignant human glioma cell line by normal leptomeningeal extracellular matrix proteins. Cancer Res 47: 3515–3622, 1987
Apodaca G, Rutka JT, Bouhana K, Berens ME, Giblin JR, Rosenblum JL, McKerrow JH, Banda MJ: Expression of metalloproteinases and metalloproteinase inhibitors by fetal astrocytes and glioma cells. Cancer Res 50: 2322–2329, 1990
Rucklidge GJ, Lund-Johansen M, Milne G, Bjerkvig R: Isolation and characterization of a metalloproteinase secreted by rat glioma cells in serum-free culture. Biochem Biophys Res Commun 172: 546–550, 1990
Lund-Johansen M, Rucklidge GJ, Milne G, Bjerkvig RA: Metalloproteinase capable of destroying cultured brain tissue isolated from rat glioma cells. Anti-Cancer Res 11: 1001–1006, 1991
Bjerkvig R, Laerum OD, Mella O: Glioma cell interactions with fetal rat brain aggregatesin vitro and with brain tissuein vivo. Cancer Res 46: 4071–4079, 1986
Bjerkvig R, Tonnesen A, Laerum OD, Backlund EO: Multicellular tumor spheroids from human gliomas maintained in organ culture. J Neurosurg 72: 463–475, 1990
Engebraaten O, Bjerkvig R, Lund-Johansen M, Pedersen PH, Wester K, Backlund EO, Laerum OD: Interaction between fetal rat brain tissue and brain tumor biopsiesin vitro. Acta Neuropathol 81: 130–140, 1990
Engebraaten O, Bjerkvig R, Berens ME: Effect of alkyl-hysophospholipid on glioblastoma cell invasion into fetal rat brain tissuein vitro. Cancer Res 51: 1713–1719, 1991
Kleinman HK, McGarvey ML, Hassell JR, Star VL, Cannon FB, Laurie GW, Martin GR: Basement membrane complexes with biological activity. Biochemistry 25: 312–318, 1986
Albini A, Iwamoto Y, Kleinman HK, Martin GR, Aaronson SA, Kozlowski JM, McEwan RN: A rapidin vitro assay for quantitating the invasive potential of tumor cells. Cancer Res 47: 3238–3245, 1987
Reich R, Thompson EW, Iwamoto Y, Martin GR, Deason JR, Fuller GC, Miskin R: Effects of inhibitors of plasminogen activator, serine proteinases and collagenase IV on the invasion of basement membranes by metastatic cells. Cancer Res 38: 3307–3312, 1988
Meissauer A, Kramer MD, Hofmann M, Erkell LJ, Jacob E, Schirrmacher V, Brunner G: Urokinase-type and tissue type plasminogen activators are essential forin vitro invasion of human melanoma cells. Exp Cell Res 192: 453–459, 1991
Deutsch DG, Mertz ET: Plasminogen purification from human plasma by affinity chromatography. Science 170: 1095–1096, 1970
Repesh LA: A newin vitro assay for quantitations of tumor cell invasion. Invasion Metastasis 9: 192–208, 1989
Mohanam S, Sawaya R, McCutcheon I, Ali-Osman F, Boyd D, Rao JS: Modulation ofin vitro invasion of human glioblastoma cells by urokinase type plasminogen activator receptor antibody. Cancer Res 53: 4143–4147, 1993
Schlechte W, Brattain M, Boyd D: Invasion of extracellular matrix by cultured colon cancer cells. Dependence on urokinase receptor display. Cancer Commun 2: 173–179, 1990
Mosmann T: Rapid calorimetric assay for cellular growth and survival.: Application to proliferation and cytotoxicity assays. J Immunol Method 65: 55–63, 1983
Stetler-Stevenson WG, Krutzsch HC, Liotta LA: Tissue inhibitor of metalloproteinase (TIMP-2): A new member of the metalloproteinase inhibitor family. J Biol Chem 264: 17376–17378, 1989
Rao JS, Steck PA, Mohanam S, Stetler-Stevenson WG, Liotta LA, Sawaya R: Elevated levels ofMr 92, 000 type IV collagenase in human brain tumors. Cancer Res 53: 2209–2211, 1993
Laemmli UK: Cleavage of structural proteins during the assembly of the bed of bacteriophage T-4. Nature (Lond) 227: 680–685, 1970
Nakajima M, Latan D, Baig M, Carralero RM, Wood WR, Hendrix MJC, Lotan R: Inhibition of retionic acid of type IV collagenolysis and invasion through reconstituted basement membrane by metastatic rat mammary adenocarcinoma cells. Cancer Res 49: 1698–1706, 1989
Nakajima M, Morekawa K, Fabra A, Bucana CD, Fidler IJ: Influence of organ environment on extracellular matrix degradation activity and metastasis of human colon carcinoma cells. J Natl Cancer Invest 82: 1890–1898, 1990
Mork SJ, Laerum OD, De Ridder L: Invasiveness of tumors of the central nervous system. In: Mareel MM, Caiman KC (eds) Invasion Experimental and Clinical Implications, pp 79–125, Oxford. Oxford University Press, 1984
Russell DS, Rubinstein LJ: Pathology of tumors of the nervous system. London Edward Arnold, 1989
Bergman BL, Scott RW, Bajpai A, Watts S, Baker JB: Inhibition of tumor cell-mediated extracellular matrix destruction by a fibroblast proteinase inhibitor, protease nexin I. Proc Natl Acad Sci USA 83: 996–1000, 1986
Rao JS, Baker JB, Morantz RA, Kimler B, Evans R, Festoff BW: Serpin inhibitors of urokinase and thrombin in normal rat brain and the brain tumor. Elevated expression of PNIlike inhibitor in a gliosarcomain vitro andin vivo. Cancer Res 50: 5039–5064, 1990
Mignatti P, Robbins E, Rifkin DB: Tumor invasion through the human amniotic membrane. Requirement for a proteinase cascade 47: 487–498, 1986
Estreicher A, Wohlwend A, Belin D, Schleuring WD, Vassalli JD: Characterization of the cellular binding site for the urokinase-type plasminogen activator. J Biol Chem 264: 1180–1189, 1989
Axelrod JH, Reich R, Mishkin R: Expression of human recombinant plasminogen activators enhances invasion and experimental metastasis of H-ras-transformed NIH3T3 cells. Mol Cell Biol 9: 2133–2141, 1989
Nakajima M, Welch D, Belloni PW, Nicolson GL: Degradation of basement membrane type IV collagen and lung subendothelial matrix by rat mammary adenocarcinoma cell clones of differing metastatic potentials. Cancer Res 47: 4869–4876, 1987
Bonfil DR, Reddel RR, Ura H, Reich R, Fridman, Harris CC, Klein-Szanto AJP: Invasive and metastatic potential of av-Ha-ras-transformed human bronchial epithelial cell line. J Natl Cancer Inst 81: 587–594, 1989
Ura H, Bonfil RD, Reich R, Reddel R, Pfeifer A, Harris CC, Klein AJP: Expression of type IV collagenase and procollagen genes and its correlation with the tumorigenic, invasive and metastatic abilities of oncogene-transformed human bronchial epithelial cells. Cancer Res 49: 4615–4621, 1989
Gottesman M: The role of proteases in cancer. Sem Cancer Biol 1: 97–160, 1990
Alvarez OA, Carmichael DF, Declerck YA: Inhibition of collagenolytic activity and metastasis of tumor cells by a recombinant human tissue inhibitor of metalloproteinases. J Natl Cancer Inst 83: 589–595, 1990
Schultz RM, Silberman S, Persky G, Bajowski AS, Carmichael DF: Inhibition by human recombinant tissue inhibitor of metalloproteinases of human amnion invasion and lung colonization by murine B16-F10 melanoma cells. Cancer Res 48: 5539–5545, 1988
Bourdon M, Matthews TJ, Pizzo SV, Bigner DD: Immunohistochemical and biochemical characterization of a glioma associated extracellular matrix protein. J Cell Biochem 28: 183–195, 1985
McComb RD, Moul JM, Bigner DD: Distribution of type IV collagen in human gliomas: Comparison with fibronectin and glioma-mesenchymal matrix glycoprotein. J Neuropath Exp Neurology 46: 623–633, 1987
Ogawa K, Oguchi M, Nakashima Y, Yamabe H: Distribution of collagen type IV in brain tumors. An immunohistochemical study. J Neuro-Oncol 7: 357–366, 1989
Bjerkvig R, Laerum OD, Rucklidge GJ: Immunocytochemical characterization of extracellular matrix proteins expressed by cultured glioma cells. Cancer Res 49: 5426–5428, 1989
Rucklidge G, Bjerkvig R, Dean V, Robins, Mella O: Immunolocalization of extracellular matrix proteins during brain tumor invasion in BD 1 × rats. Cancer Res 49: 5419–5423, 1989
Paranjpe M, Engel L, Young N, Liotta LA: Activation of human breast carcinoma collagenases through plasminogen activator. Life Sci 26: 1223–1231, 1980
Reith A, Rucklidge GJ: Invasion of brain tissue by primary glioma. Evidence for the involvement of urokinase-type plasminogen activator as an activator of type IV collagenase. Biochem Biophys Res Commun 186: 348–354, 1992
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rao, J.S., Steck, P.A., Tofilon, P. et al. Role of plasminogen activator and of 92-KDa type IV collagenase in glioblastoma invasion using anin vitro Matrigel model. J Neuro-Oncol 18, 129–138 (1993). https://doi.org/10.1007/BF01050419
Issue Date:
DOI: https://doi.org/10.1007/BF01050419