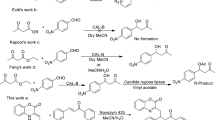
Summary
Doubly enantioselective lipase-catalyzed esterification of racemic acids and alcohols was proceeded in n-hexane. The enantioselectivities of the lipase toward both of racemic substrates were affected reciprocally. It showed that the active site of the lipase was quite flexible.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chen, C.S., Fujimoto, Y., Girdaukas, G and Sih, C.J. (1982) J. Am. Chem. Soc., 104, 7294–7299.
Chen, C. S. and Sih, C. J. (1989) Angew. Chem. Int. Ed. Engl. 28, 695–707.
Chen, C.S., Wu, S. H., Girdaukas, G and Sih, C.J. (1987) J. Am. Chem. Soc., 109, 2812–2817.
Holmberg, E. and Hult, K. (1992) Biocatalysis, 5, 289–256.
Kirchner, G., Scollar, M.P. and Klibanov, A.M. (1985) J. Am. Chem. Soc., 107, 7072–7076.
Klibanov, A. M. Acc. Chem. Res. (1990) 23, 114–120.
Sih, C. J. and Wu, S. H. (1989) Topics Stereochem. 19, 63–125.
Sonnet, P. E.(1987) J. Org. Chem. 52, 3477–3479.
Theil, H., Kunath, A. and Schick, H. (1992) Tetrahedron Lett, 33, 3457–3460.
Wong, C. H. Science. (1989) 244, 1145–1152.
Wu, S.H., Chu, F.Y. and Wang, K.T. (1991) BioMed. Chem. Lett, 1, 339–342.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, PY., Wu, SH. & Wang, KT. Double enantioselective esterification of racemic acids and alcohols by lipase from Candida cylindracea . Biotechnol Lett 15, 181–184 (1993). https://doi.org/10.1007/BF00133020
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00133020