Summary
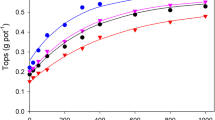
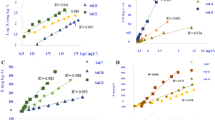
The effects of phosphate buffer capacity on the plant-availability of labile soil phosphate, when measured as intensity (I) or quantity (Q), are described and tested using results from a greenhouse experiment on 24 Sherborne soils. In multiple regression studies, phosphate buffer capacity with I or Q measurements as independent variables accounted for up to 94% of the variance in P uptake by ryegrass, the maximum buffer capacity being generally more useful than the equilibrium buffer capacity.
When the quantity of soil P is measured (Q), its availability (i.e. ease of desorption) to plant roots is inversely related to the Langmuir bonding energy parameter and the buffer capacity. When the intensity of soil P is measured (I), its availability (i.e. resistance to change) is directly related to the adsorption and buffer capacities. The levels of Q or I, therefore, which are optimal for plant uptake vary with the buffer capacity of the soil. There is little or no correlation between the adsorption capacity and the bonding energy in many soils and consequently phosphate buffer capacity is only poorly correlated with the total adsorption capacity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bache, B. W. and Williams, E. G., A phosphate sorption index for soils. J. Soil Sci. 22, 289–301 (1971).
Beckett, P. H. T. and White, R. E., Studies on the phosphate potentials of soils. III. The pool of labile inorganic phosphate. Plant and Soil 21, 253–282 (1964).
Cole, C. V. and Olsen, S. R., Phosphorus solubility in calcareous soils: II. Effects of exchangeable phosphorus and soil texture on phosphorus solubility. Soil Sci. Soc. Am. Proc. 23, 119–121 (1959).
Gunary, D., A new adsorption isotherm for phosphate in soil. J. Soil Sci. 21, 72–77 (1970).
Holford, I. C. R. and Mattingly, G. E. G., A model for the behaviour of labile phosphate in soil. Plant and Soil 44, 219–229 (1975).
Khasawneh, F. E., Solution ion activity and plant growth. Soil Sci. Soc. Am. Proc. 35, 426–436 (1971).
Mattingly, G. E. G., Russell, R. D. and Jephcott, B. M., Experiments on cumulative dressings of fertilisers on calcareous soils in south-west England. II. Phosphorus uptake by ryegrass in the greenhouse. J. Sci. Food Agr. 14, 629–637 (1963).
Olsen, S. R. and Watanabe, F. S., Diffusion of phosphorus as related to soil texture and plant uptake. Soil Sci. Soc. Am. Proc. 27, 648–653 (1963).
Russell, R. D., Experiments on cumulative dressings of fertilisers on calcareous soils in south-west England. I. Description of field experiments and soil analysis for phosphorus residues. J. Sci. Food Agr. 14, 622–628 (1963).
Schofield, R. K., Can a precise meaning be given to ‘available’ soil phosphorus. Soil and Fertilizers 18, 373–375 (1955).
White, R. E., Buffering capacity of soil on uptake of phosphorus by plants. Trans. 9th Intern. Congr. Soil Sci. 2, 787–794 (1968).
Williams, E. G., The intensity and quantity aspects of soil phosphate status and laboratory extraction values. Anales Edafol. Agrobiol. (Madrid) 26, 525–546 (1967).
Woodruff, J. R. and Kamprath, E. J., Phosphorus adsorption maximum as measured by the Langmuir isotherm and its relationship to phosphorus availability. Soil Sci. Soc. Am. Proc. 29, 148–150 (1965).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holford, I.C.R., Mattingly, G.E.G. Phosphate adsorption and availability plant of phosphate. Plant Soil 44, 377–389 (1976). https://doi.org/10.1007/BF00015889
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00015889