Abstract
This chapter focuses on the shape, growth, and productivity of individual trees growing in inter- versus intraspecific environments. The individual tree senses and responds to the prevailing environmental conditions. The properties of the individuals determine the forest stand dynamics as individuals of different species interact with each other. Therefore, the level of the individual tree is most suitable for understanding competition, competition reduction through complementarity, and facilitation, which can result in the differences between structure dynamics and productivity of mixed compared with monospecific stands. The chapter shows how species mixing can modify the size development, persistence, and productivity of individual trees in mixed stands compared with members of the same species in neighbouring monospecific stands. Many of the beneficial tree mixing reactions result from complementary crown and root shape, spatially or temporally complementary resource exploitation, redistribution of resources, or modification of growth allocation and allometry introduced in this chapter.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Size growth and persistence are key requirements for a tree to compete and maximise fitness under the selective pressure in a forest stand. This chapter shows how species mixing can modify the size development, persistence, and productivity of individual trees in mixed stands compared with members of the same species in neighbouring monospecific stands. Living in association, both in intra- and interspecific neighbourhoods, entails pros and cons in comparison to solitary growth. An individual tree may be facilitated physically, e.g. by neighbours as they protect against storm breakage (von Lüpke and Spellmann 1997, 1999), sun scorch of bark (Assmann 1970), or snow sliding (Kuoch 1972; Mayer and Ott 1991, pp. 194–197; Strobel 1995). However, the meaning of neighbourhood is ambivalent, as it also entails competition when there are insufficient resources for all (Connell 1990). The interplay between facilitation and competition determines intraspecific and particularly interspecific coexistence (Larocque et al. 2013).
Mixing effects can be studied at various levels of organisation, e.g. at organ, individual, stand, or landscape level. Given that the individual tree forms the basic unit in forest stand ecology, this chapter focusses on the shape, growth, and productivity of individual trees growing in inter- versus intraspecific environments. The individual tree senses and responds to the prevailing environmental conditions. The properties of the individuals determine the forest stand dynamics as individuals of different species interact with each other (Smith and Smith 2009, p. 13). Therefore, the level of the individual tree is most suitable for understanding competition, competition reduction through complementarity, and facilitation, which can result in the differences between structure dynamics and productivity of mixed compared with monospecific stands discussed in Chaps. 4 and 5.
Understanding of the interspecific interactions between tree species can be improved through generalisation of the hologenome theory (Zilber-Rosenberg and Rosenberg 2008). According to this theory, a holobiont in a strict sense is a host organism in interaction with all associated microorganisms. More generally, all regular organisms which closely interact with each other (e.g. trees in mixed-species stands) can be perceived as holobiont-like systems and may also be studied as entities rather than focussing on the individual organisms (Matyssek and Lüttge 2012).
Tree species mixing may change the environmental conditions and thereby the physiology, morphology, and stand structure. As a result, the tree and stand growth also undergo changes. Because of the strong interaction between structural patterns and processes, the structure and morphology may reflect the results of mixing effects which are much more difficult to measure than the underlying physiological processes. To quantify mixing effects in this chapter, we compare the individuals’ behaviour in terms of tree shape and growth in mixed versus monospecific stands. We use the trees’ structure and growth in a monospecific stand as a reference for quantifying tree behaviour in neighbouring mixed stands.
Reaction patterns of shape, allometry, growth, and productivity at the individual tree level as presented in this chapter may contribute to understanding mixing effects. Further insight into the underlying mechanisms require analyses of the change in supply, capture, or use efficiency of resources in poly- versus monocultures (Binkley et al. 2004; Richards et al. 2010) as introduced in Chap. 3. We will set the growth of individual trees in relation to their required growing area (equivalent to tree stand area) and, in this way, link the tree level with the stand level and size distribution level (Chaps. 4 and 5).
6.1 Principles of Individual Tree Ontogeny
To explain inter- and intraspecific interactions between trees such as competition, competition reduction, and facilitation as well as their net effect on the growth of individual trees in mixed and monospecific stands, plant biology mostly starts with the ontogeny of a solitarily growing tree. The schematic Fig. 6.1a shows the unimodal course of the stem volume increment, ivs, and the s-shaped curve of stem volume, abbreviated to vs. These curves represent the characteristic individual tree ontogeny and were originally derived theoretically from basic relationships between tree assimilation and respiration (von Bertalanffy 1951). However, many works (e.g. Ryan and Yoder 1997; Yoder et al. 1994) show that it is rather nutrient limitation, genetic changes in meristem tissue, and hydraulic limitations that are behind the characteristic unimodal course of the stem volume increment, ivs, and the s-shaped curve of stem volume (see Boxes 6.1 and 6.2).
Schematic representation of the development of a solitarily growing individual tree. (a) Growth of stem volume, ivs, and development of volume yield, vs, over tree age. (b) Allometric dependency of crown projection area and tree stand area, sa, on stem volume, vs. In this example, we assume that tree stand area and crown projection area are equally scaled in relation to tree volume (sa ∝ cpa ∝ v 1/2). Just the allometric factors, a, in cpa = a 1 × vs1/2 and sa = a 2 × vs1/2, which indicate the tree shape, differ (a 1 ≠ a 2). (c) Development of volume growth and volume yield per area. ivs/sa and vs/sa reflect the tree volume growth and yield per tree stand area (bold lines), while ivs/cpa and vs/cpa represent the tree growth and yield per crown projection area
Box 6.1 Characteristics of a Tree’s Crown Morphology, Crown Extension, and Growing Area
Description and analysis of tree structure, growth, and growth efficiency in this chapter are mainly based on the tree variables shown in Box Fig. 6.1-1. To compare tree morphology in mixed versus monospecific stands, the following ratios between tree organ sizes are frequently used:
-
h/d = the h–d ratio using h (m) and stem diameter at breast height (cm) addresses tree stability.
-
cd/d = the cd–d ratio with crown diameter (m) and stem diameter (cm) addresses the crown extension.
-
d r/d s = d r–d s ratio with diameter of the main root, d r, and stem diameter at breast height, d s, quantifies the root-stem relationship. When based on increment cores from stem and main roots, this relationship provides insight into the allocation principle between stem and main root (Fig. 6.18).
-
cpa/sa = this ratio between crown projection area, cpa, and tree stand area, sa, is a measure for the interlocking of neighbouring crowns.
(a–c) Essential tree characteristics. (a) Tree height, h; crown length, cl; height to crown base, hcb; crown diameter, cd; tree diameter at breast height, d 1.3; and stem volume, vs. Variable cpa represents the crown projection area. (b) Crown volume, cv; crown surface area, cs; diameter of the stem, d s = d1.3; and diameter of the three tallest roots, d r1 … d r3. (c) Tree stand area equivalent to growing area, sa, and crown projection area, cpa. Particularly in densely packed stands, cpa is often greater than sa
Most relationships between tree organ sizes change non-isometrically with tree size, i.e. when organ size x increases, organ y does not enlarge linearly (y = a + α × x) but according to the allometry equation (y = a × x α) with α ≠ 1. This means that the above ratios between organs, e.g. h/d or cd/d, show an ontogenetic drift with increasing size (see Box 6.3).
Raw density of wood (RDW) of species in boreal and temporal forests ranges mainly between 350 and 650 kg m−3 (Pretzsch 2009, p 67). RDW can be used to transform stem volume, vs, to stem mass, ms (ms = vs × RDW).
Box 6.2 The Rules by Kleiber (1947) and Rubner (1931) for Explaining the Ontogeny and the Growth and Yield Curves of Individual Trees
According to these rules, the unimodal course of the net growth (Fig. 6.1a) results from the difference between the anabolistic term (A = a × v 3/4) as minuend and the catabolistic term (R = b × v 1) as subtrahend (NG = a × v 3/4 − b × v 1). The variable A represents the gross assimilation, R the respiration, and NG the net growth of the tree. For the anabolistic term, the rule by Kleiber (1947) assumes that A scales with 3/4 over plant mass or volume. According to Rubner’s rule (1931), respiration increases linearly with mass or volume (plant mass ∝ plant volume). These two underlying allometric relationships are shown together in Box Fig. 6.2-1a and reflect that the net growth (grey area between the curves) is low for small trees, high for medium-sized trees, and low again for giants. This in turn reveals that plant size is a conflicting trait. It entails both supremacy over smaller neighbours and also low growth due to high expense for respiration. Intra- and interspecific interactions can affect both A and R and thus modify the trees’ development. Box Figure 6.2-1b shows the characteristic unimodal curve of the net growth, ivs, over tree volume, vs, which results from that in Box Fig. 6.2-1a. Starting with the unimodal ivs–vs relationship in Box Fig. 6.2-1b and assuming an initial stem volume for plant age 1 (e.g. v 1 = 0.001 m3), the course of volume increment, ivs, and the achieved volume, vs, can be derived (Box Fig. 6.2-1c). The development of ivs over age is the unimodal growth curve; the sigmoid development of vs over age results from integration of ivs.
(a–c) Derivation of the unimodal growth and s-shaped yield development of a tree over age from the allometric rules by Kleiber (1947) and Rubner (1931). (a) Net growth (grey area between the curves, NG) is the difference between gross assimilation (concave line, A = a × v 3/4) and respiration (straight line, R = b × v 1). (b) Development of net plant growth, ivs = NG, over plant volume, vs. The latter curve results from the relationships between A and vs and between R and vs in Box Fig. 6.2-1a. (c) Course of plant volume, vs, and plant volume increment, ivs, over age, developed from the relationship between ivs and vs in Box Fig. 6.2-1b assuming an initial plant volume of 0.001 m3
While the shown unimodal course of the stem volume increment, ivs, and the s-shaped curve of stem volume, abbreviated to vs, are a good basis and reference for analysing any external effects such as intra- and interspecific competition and facilitation on tree growth, their causal explanation has been improved. Ryan and Waring (1992) showed that respiration of older and taller trees generally declines as growth declines (i.e. R < < b × v 1) because more than half of the respiration is growth respiration. Even maintenance respiration of big trees typically goes down (Yoder et al. 1994). Many works (e.g. Ryan and Yoder 1997) show that it is rather nutrient limitation, genetic changes in meristem tissue, and hydraulic limitations that are behind the characteristic unimodal course of the stem volume increment, ivs, and the s-shaped curve of stem volume. Although it is temptingly simple, the theoretical explanation of individual tree ontogeny by von Bertalanffy (1951) based on the rules by Kleiber (1947) and Rubner (1931) is increasingly called into question by empirical findings.
Figure 6.1a represents the growth through ‘the plant’s eye view’, i.e. the development of growth and size. The production ecology represented by the ‘forester’s eye view’ asks for the growth and volume per growing area, i.e. for the productivity. To transition from tree growth to tree growth per growing area, we apply the allometric relationship between stem volume and tree growing area (sa ∝ vs1/2) which is shown in Fig. 6.1b (West et al. 2009). By dividing the increment, ivs, and volume, vs, by the required growing area, we get the growth per unit area (productivity ivs/sa) and standing volume per unit area (standing stock vs/sa) of the tree (Fig. 6.1c).
Note that the transition from tree growth to tree productivity is based on growing area, sa, and not crown projection area, cpa. Unless cpa = sa, crown area productivity (ivs/cpa) differs from growing area productivity (ivs/sa) (Gspaltl et al. 2012). Particularly in mixed stands, cpa is often larger than sa as illustrated in Fig. 6.1b. In this case, ivs/cpa << ivs/sa, i.e. upscaling from tree to area level based on crown projection area underestimates tree productivity. The transition from tree growth to tree growth per area means that the time point of culmination and inflexion shifts towards younger tree ages as the required stand area of a tree increases with size (compare broken vertical lines in Fig. 6.1c and a).
The depicted unimodal and s-shaped development courses of a solitary tree in terms of growth per plant and growth per unit area (Fig. 6.1a and c) form the reference for the subsequent introduction to the effects of competition and facilitation.
6.2 Interaction Between Trees in Inter- Versus Intraspecific Environments
Suppose that of three equally sized trees established on similar sites, one tree grows solitarily, one in medium, and the third in maximum density with neighbours of the same species (Fig. 6.2a, trees 1–3). When we choose the solitary tree (tree 1) as a reference and set its growth rate to 1.0, what will the growth rate of the trees growing in medium and high stand density be and what does this depend on? On the one hand, the individual growing area becomes smaller from trees 1 to 3 so that the competition for resources (water, light, nutrients) increases. Helms (1998, p 34) defined competition as ‘… the extent to which each organism maximizes fitness by both appropriating contested resources from a pool not sufficient for all, and adapting to the environment altered by all participants…’. However, living in association with neighbours may also have advantages over solitary growth. For example, neighbours may protect against windthrow (Knoke and Hahn 2007; Griess and Knoke 2011), improve the soil conditions (Augusto et al. 2002; Heinsdorf 1999), or prevent nearest neighbours from sun scorch of bark or drought (Assmann 1970; Pretzsch et al. 2012b). The latter abiotic or biotic improvement of the neighbours’ living conditions is called facilitation (Vandermeer 1992). Vandermeer (1992, p. 9) defined facilitation as ‘… the process in which two individual plants or two populations of plants interact in such a way that at least one exerts a positive effect on the other. Double facilitation is equivalent to mutualism’. In Fig. 6.2a, the trees’ growth rates (indicated by the white envelopes of trees 1–3) when growing in solitary conditions and in medium and high density reflect the net effect of competition and facilitation on tree growth. In relation to the growth of the solitary tree 1, the interaction between competition and facilitation results in a net facilitation under medium density (Fig. 6.2a, envelope tree 2 > envelope tree 1) and a net competition effect (Fig. 6.2a, envelope tree 3 < envelope tree 1) in the case of high density.
Schematic representation of the effect of competition and facilitation in (a) a monoculture and (b) a mixed stand on tree growth under solitary growing conditions (left) and in medium (centre) and high stand density (right). The outer hulls of the crown bodies (white envelopes) represent the current growth rate of the trees in question. From left to right (from trees 1 to 3 and 4 to 6, respectively) competition becomes stronger as does facilitation. As a result, trees 2 and 5 encounter medium density but can have higher growth rates (white envelopes) than trees 1 and 4 under solitary conditions
Analysing, understanding, modelling, and prognosticating monocultures often entail a thorough consideration of the effect of competition between neighbouring trees but mostly ignores facilitation. A plant may be facilitated by its neighbour through improvement of environmental factors (non-resource) or resource supply or a combination of both. Examples for improvement of environmental factors are slowing down the wind speed and avoidance of storm breakage, shading and avoidance of sunburn, providing a barrier against spread of insects, suppression of forest floor vegetation and competing weeds, and protection against snowslides or browsing. This kind of facilitation does not entail direct costs in terms of resources for the benefactor. In contrast, when facilitation is based on an improved resource supply, the benefactor may lose resources while the beneficiary gains.
Competition and facilitation act simultaneously. It is difficult to separate them. In field experiments in monospecific stands, the net effect of the interaction between facilitation and competition on tree structure and growth rather than their particular effects is accessible through measurement. To simplify matters, the interplay between competition and facilitation is first illustrated for monospecific stand conditions. In Fig. 6.3 we set the growth in solitary conditions as the reference (1.0-line) and show three basic patterns of how facilitation and competition can modify this solitary growth. Remember that in terms of absolute growth, a solitary tree follows the trajectories shown in Fig. 6.1 and that we ask here how neighbourhood might modify this generic trajectory.
Schematic representation of the modification of solitary growth (1.0-line) through facilitation and competition in dependence on stand density in monocultures. The graphs show three essential patterns of how net facilitation and net competition result from the interplay between facilitation only and competition only. (a) Facilitation only has the upper hand in relation to competition only so that net facilitation is effective over the whole range of stand densities. (b) Competition only has the upper hand in relation to facilitation only so that net competition is effective over the whole range of stand densities. (c) The balance between competition only and facilitation only results in net facilitation under low stand density and net competition under high stand densities
Figure 6.3a assumes that competition only increases from low to high stand densities and that facilitation also only increases with density. Given that in this theoretical case, the balance between the negative effect of competition and the positive effect of facilitation on growth is always positive, we consider net facilitation (facilitation only minus competition only) over the whole range of stand densities. Figure 6.3b represents the opposite case where competition only always has the upper hand and net competition prevails over the whole range of stand densities. These two reaction patterns with net facilitation or rather net competition over the whole range of densities tend to be exceptions. Figure 6.3c represents the most frequently observed reaction pattern, where net facilitation dominates under low densities and net competition is predominant in dense stands. In sparsely stocked stands, competition is still low enough to be compensated or even overcompensated by facilitation. Such low density conditions, where facilitation may have the upper hand, are frequently observed in grassland ecosystems which are too poor for the development of closed forests (Callaway and Walker 1997; Canham et al. 1994, 2004). But they are less relevant in forest ecosystems (Forrester 2013). As stand density increases, growth mostly declines, can hardly be balanced by facilitation, and causes mostly net competition. However, this consideration highlights that although not easy to detect and to separate from competition, facilitation may also play a role in forest stands with high stand density. The facilitative effect of medium density compared with high density or solitary growth can be revealed by Nelder trials (Nelder 1962) where the tree growth can only be at optimum between the extremes (Uhl et al. 2015).
The combined effect of competition and facilitation becomes even more complex in mixed stands where trees differ in terms of traits and niches. Suppose the above trees in solitary and medium- and high-density constellations grow in mixed as opposed to monospecific stands (Fig. 6.2b, trees 4–6). In this example, 50% of the neighbours belong to a second species, i.e. competition and facilitation become interspecific and modified compared with monospecific stand conditions.
Reduced competition and an even stronger facilitation caused by species mixing might result in an even more obvious growth maximum under medium stand density (tree 5, envelope tree 5 >> envelope tree 4) compared with growth rates under solitary conditions or growth rates under maximum density.
Figure 6.4a indicates this shift in the growth-density relationship due to a reduction in competition (e.g. by spatial or temporal niche complementarity) and an increase in facilitation (e.g. tapping nutrients in deep soil layers which are favourable for species 1) through admixture of trees of species 2. Such a reaction pattern can cause a widening of the density range with net facilitation (horizontal arrow). In other words, the range where net competition has the upper hand narrows. For mixed-species stands as in monospecific stands, a net competition which is normal for trees growing in medium or high stand density does not exclude the effect of facilitation. In both cases, competition merely has the upper hand over facilitation. The relationship between the net tree growth in mixture and in monoculture (vertical arrows in Fig. 6.4a) reflects the mixing effect in terms of tree growth.
Schematic representation of the modification of solitary growth (1.0-line) through facilitation and competition in dependence on stand density in mixed-species stands. (a) Broadening of the range of net facilitation (horizontal arrow pointing right) because of additional facilitation and competition reduction through species mixing. (b) Increasing net facilitation (horizontal arrow pointing right) due to the increase in facilitation from mild (+) to harsh (−) environmental conditions
Facilitation effects are widely held to be strong under harsh environmental conditions and weaker under favourable conditions (Brooker et al. 2008; Callaway and Walker 1997). Accordingly, the density range with net facilitation may increase as site quality decreases (see Fig. 6.4b curves referred to as +, ±, −). Note that it is also possible for the net curve to remain the same although its components change in the case that the positive and negative changes in competition and facilitation cancel each other out.
While in plant ecology the net effect of competition and facilitation is mostly related to solitary plant growth (Callaway and Walker 1997; Belsky and Canham 1994; Forrester 2013), in closed forests we are interested in the mixing effects under mean or high stand density. In field experiments in monospecific stands, the net effect of the interaction between facilitation and competition rather than their specific effects are accessible through measurement. To complicate the situation, measurements of solitary plant or tree growth which are required as a reference are scarcely available, except from very rare experiments like the outer Nelder circles (Nelder 1962; Uhl et al. 2015) or spacing and thinning experiments, which include plots with open-growing trees. So, in most cases only growth of medium to high density plants is measured, and it is difficult to quantify whether net facilitation or competition has the upper hand because of the lack of reference measurements. The same restriction applies for quantification of net facilitation or net competition in mixed stands in relation to solitary plant growth (according to the definition of facilitation and competition). However, what can instead be easily detected in field experiments is the change in the net curve from monospecific to mixed stands (see upwards shift of the net curves in the centre of Fig. 6.4a and b marked with vertical arrows).
The relationship between the tree growth in mixed versus monospecific stands indicates the net mixing effect in closed stands. Of special interest is to what extent mixing increases or reduces net competition of trees in closed stands rather than whether the growth-density relationship shows net facilitation or net competition of a solitary tree. The former would mean an increase or decrease in productivity of the species in mixed versus monospecific stands. This practically relevant effect is represented by the relationship between mixed and monospecific stand performance in the right branch or even at the right end of the net growth-density relationship in Fig. 6.4.
6.3 Basic Feedback Loop Between Growth, Structure, and Local Environment in Forest Stands
The tree–tree interaction illustrated by an example in the previous section can be conceptualised by the basic feedback loop between functioning, structure, environment, and functioning in forest stands (Fig. 6.5). In order to explain the emergence of intra- and interspecific interactions between neighbouring trees, a distinction between their functioning (e.g. germination, growth, defence, flowering, fruiting, senescence), their structure formation (e.g. body size, crown and root extension, crown space filling with foliage), and environment (resource supply, e.g. water, nutrient, or CO2 supply) and environmental factors (e.g. temperature, salinity, browsing) is helpful.
Feedback loop between stand structure, environmental conditions, and tree functioning in a two-species stand. The outer feedback loops (FSE loop) structure → environment → functioning → structure (bold arrows) are slow, and the inner loops (FS loop) environment → functioning → environment work faster. Further explanation is provided in the text
The conceptual model in Fig. 6.5 clarifies essential plant–plant interactions in monospecific and mixed-species communities. Trees interact with their environment in two ways, via structure and functioning (Hari 1985). The feedback between functioning and environment (FE loop) can be very rapid and temporary, e.g. defence substances are exuded quickly after or nearly simultaneously to the injury or pathogen attack and are reduced after stress release (within-stand environment versus external environmental drivers). Reduction of atmospheric CO2 concentration or soil water supply by roots and crowns of fast-reacting neighbours can immediately reduce photosynthesis or growth of slower-reacting trees.
The feedback between functioning, structure, and environment (FSE loop), in contrast, is slow and accumulative. The functioning changes the structure (e.g. tree size and structure) and, via tree and stand structures, the trees’ environment. The crown structure, for instance, determines where the water and mineral nutrients from the crown periphery drip to and to what extent neighbours are shaded. The pattern of water drip and shading determines where the roots and crowns of neighbours grow and where they forage. This in turn determines their morphological structure. Trees develop and maintain their root, stem, and crown structure for decades and affect and adapt to their surroundings rather permanently.
The feedback between environment, growth, and structure may be relatively clear in monospecific stands where the species apply similar tricks and traits when appropriating contested resources, adapting to the environment, and modifying the stand structure. In mixed stands with two species as shown in Fig. 6.5, or with even more species, interactions and their effects on growth, structure, and environment may be more varied. Mixed-species modify their environment to their own benefit, e.g. by overtopping the neighbouring crowns or penetrating neighbouring roots in order to improve access to contested resources and acclimation to the environment altered by their neighbours. The principle feedback remains the same when neighbours belong to different species; however, their tricks and traits to modify their environment may be different, so that a broader range of reaction patterns, structures, and changes to the environment may occur.
The subsequent analyses of tree structure and tree growth in mixed versus monospecific stands address the two key elements of the feedback loop. Long-term experiments provide retrospective information on growth and structure but rarely time series on environmental variables such as light profile, nutrient supply, or water uptake of trees in mixed versus monospecific stands. Therefore, the effect of mixing on the local environment is less known and understood than the effect on structure and growth. However, future approaches will close this knowledge gap to enhance understanding of the feedback and underlying mechanism as a whole (Binkley et al. 2004; Pretzsch et al. 2015).
6.4 Growth and Yield at the Tree Level
Any changes to the local environmental conditions in mixed stands compared with monospecific stands primarily determine tree functioning in terms of growth (Fig. 6.5). This section will deal with these growth responses to mixing. Reduction or increase of the growth rate due to mixing can modify the tree and stand structure. In addition, the local environment may considerably modify the growth allocation in terms of tree morphology and thereby the tree and stand structure. Such structural changes which have a feedback effect on the local environment will be the subject of Sect. 6.5.
6.4.1 Dominant Rather Than Open-Grown Trees as Reference
Grassland plant ecology mostly uses solitarily growing plants as a reference for detecting facilitation or competition effects (Canham et al. 1994, 2004; Forrester 2014). When association with neighbours increases plant growth above the level of solitary plants, this indicates facilitation. When the opposite occurs, this serves as evidence of competition. Using open-grown trees as a reference for mixing effects such as facilitation or competition reduction is barely feasible given that these are rare in woodlands and, when situated in distant open land, are mostly incomparable in terms of site characteristics.
In Fig. 6.6 we therefore use dominant trees (d) in monocultures as a reference instead of open-grown trees (o). The former have lower growth rates than open-grown trees, but long-term experiments in monocultures provide a sound database for predicting the potential growth of dominant trees in forest stands in dependence on the site conditions. In the following, we use the unimodal diameter growth-tree diameter curve expected for dominant trees in monocultures as a reference for revealing the competition and species admixture effect. For derivation of the species-specific unimodal potential diameter growth-diameter relationships, see Pretzsch and Biber (2010). Following the approach from Sect. 6.2, we use this potential curve (the curve for dominant trees in monospecific stands) as the 1.0-line to show any mixing effects. Of main interest is how stand density and mixing proportion lead growth away from this baseline.
Tree diameter increment along the tree diameter and site spectrum covered by data from long-term experimental plots in southern Germany according to Pretzsch and Biber (2010). Model curves are given for trees growing under solitary conditions (upper curve triplets) and for dominant trees growing in stands (lower curve triplets). The light grey, grey, and black lines represent the diameter growth for fertile, mean, and poor sites (dominant height of 35 m, 30 m, and 25 m, respectively, at stand age 100 years)
6.4.2 Evidence of Facilitation and Competition Effects at the Tree Level
Figure 6.7 shows exemplarily for even-aged mixtures of Norway spruce/European beech, European beech/sessile oak, and Scots pine/Norway spruce how mixing can modify the individual tree diameter growth under ceteris paribus conditions, i.e. when all effects (tree size, stand density, or site conditions) except for species composition are statistically eliminated. The analysis is based on long-term mixing experimental plots in Bavaria (Pretzsch 2009). Established in the 1930s, they mostly include reference plots in monospecific stands, provide individual tree data (tree position, tree size, growth rate) of several thousand trees in mixed and neighbouring monospecific stands, and thus provide information about mixing effects on tree growth.
Relative tree diameter increment, id, of various tree species in dependence on the mixing portion (monospecific stand, 25%, 50% admixture of a second tree species) and the given local SDI on the x-axis. The 1.0-line serves as a reference and represents the growth of a dominant tree in monospecific stands of the respective species
The underlying model for the reaction patterns shown in Fig. 6.7 considers that growth of trees in mixed and monospecific stands may simply differ because of different size. Given space restrictions it is introduced here only briefly: the size effect is taken into consideration by the unimodal relationship between growth and size in monospecific stands (Fig. 6.6). This unimodal curve expected for dominant trees in monospecific stands is used as a reference to reveal the effects of species mixing and local stand density. Of main interest is how mixing proportion and stand density lead growth away from the baseline (1.0-line). Competition was quantified in the model using the local stand density index, i.e. the number of trees, ni, within a defined radius around each tree scaled up to a hectare (N i = n i /(r i 2 × π)) and converted to the SDI i = N i × (25/d q )−1.605 according to Reineke (1933). This total SDI was split into two components, the part containing the species equal to the species 1 (= species of the centre tree) and the rest. The mixing portion was therefore the ratio between the SDI portion of trees differing from the species of the centre tree and the total SDI. So a mixing portion of mix = 0 signifies a monoculture composed of species equal to the centre tree, whereas mix = 0.25 indicates that within the circle of radius r around the centre tree, 25% of trees are unequal to species 1. We considered two-species stands, so that mix = 0.25 or 0.50 means an admixture of 25 or 50%, respectively, of trees of species 2 in the vicinity of species 1. To show the tree reaction patterns at the tree level, we applied a model which considers the size effect on growth (id–d relationship) and the stand density effect (id-SDI relationship) so that the effect of inter- versus intraspecific environments on growth under ceteris paribus conditions can be eliminated. The model was fitted with individual tree data from N. spruce (n = 10,160), E. beech (n = 13,861), pine (n = 2052), and oak (n = 4960) growing in monospecific and mixed stands.
In all tree associations shown in Fig. 6.7, stand density reduces tree growth; the reduction is very strong in the case of beech and rather small in the cases of oak and pine. Even more variable is the mixing reaction between the species combinations investigated. In mixture of Norway spruce and European beech (Fig. 6.7a, b), both species show a reduction in competition and increase in growth compared with the monospecific stand (black solid line). Norway spruce benefits moderately from beeches in the neighbourhood (Fig. 6.7a), while European beech benefits strongly from Norway spruce (Fig. 6.7b). In the mixture with sessile oak, European beech again benefits considerably (Fig. 6.7c), while tree growth of oak is hardly affected (not shown). Scots pine is the beneficiary in the mixture with Norway spruce (Fig. 6.7d), and the benefit gained by Scots pine is partly at the expense of Norway spruce (not shown). Of the species considered, European beech experiences the strongest growth reduction through increased stand density index (SDI) but also the greatest benefit through admixture of other species like Norway spruce and sessile oak. The results suggest that mixing reactions vary depending on both the particular species investigated and the species which is added in its vicinity.
Important is the finding that the positive mixing effects in terms of growth are modified by stand density. The mixing effects are present to various extents all along the density range. However, they can be at maximum in sparsely stocked stands and decrease with increasing stand density (from left to right in Fig. 6.7). Under low density, facilitation and competition can raise the growth above the potential for dominant trees in monospecific stands (1.0-line), i.e. the net effect of mixing can be positive. Of course, at the latest when density gets very close to zero, there is no longer any facilitation or competition caused by trees. In dense stands, the increase from monoculture to 25% and 50% is still relevant, but the positive mixing effect cannot overcompensate the negative effects of density on tree growth, so the combined effect of mixing and density on tree growth is negative.
6.4.3 Basic Growth Reaction Patterns in Intra- Versus Interspecific Neighbourhoods
The last section showed that—depending on the mixing portion and the degree of stand density—living in association can increase or decrease the growth rate. This means that the unimodal course of individual tree growth is modulated depending on the prevailing local environmental neighbourhood conditions of the tree. Figure 6.8 is a schematic representation showing that facilitation and competition in inter- and intraspecific neighbourhoods can modify the volume growth in manifold ways. In contrast to an open-grown tree (o), dominant trees (d) in forest stands grow permanently under net competition. For the dominant tree (d) in Fig. 6.8a we assume a constant reduction in the solitary tree growth by 35% due to a sequestration of resources by neighbours. The trajectories of dominant trees (d) are more suitable as a reference for indicating mixing effects than are open-grown trees (o) (see Sect. 6.4.1). However, in Fig. 6.8b–e we include both open-grown and dominant trees as references.
Generic patterns of tree volume growth in dependence on age, modified by competition and facilitation. The bold lines represent the response to competition and facilitation. The thin curves for open-grown trees (o) and dominant trees in a stand (d) serve as references. (a) Behaviour of an open-grown tree (o) and a dominant tree (d) in a forest stand. (b) Facilitation can increase tree growth beyond open-growth level. (c) Mixture can increase dominant tree growth through facilitation and competition reduction. (d) Mixture can increase and accelerate or (e) decrease and slow down growth in relation to dominant trees
Figure 6.8b shows how permanent facilitation, e.g. through wide spacing, can increase the growth of an individual tree (bold line) permanently above that of the solitarily open-grown tree (o). In this case, the balance between the respective positive and negative effects on the growth of competing and facilitating neighbours is positive. Trees growing in group structure in the alpine zone compete for light, but their neighbours also protect them against snow and wind so that they frequently benefit from being associated and can grow more than solitary trees. Forest management makes use of such a permanent facilitation by planting trees in groups or clusters known as ‘Rotten’ in the alpine zone (Strobel 1995) or ‘Nester’ in the lowlands (Saha et al. 2012).
The pattern in Fig. 6.8c (bold line) occurs frequently when a dominant tree benefits from neighbours of a different species through competition reduction (e.g. the effect of a rather translucent species) or facilitation (e.g. the effect of an atmospheric nitrogen fixing species or hydraulic water redistribution). In this case, the growth of a dominant tree might be accelerated permanently, and its superiority may extend over the whole tree age.
The net effect of competition and facilitation may vary over time. Figure 6.8d (bold line) shows a frequently observed acceleration in growth through facilitation in the early state, followed by a reduction in the advanced state of plant development. So, tree growth exceeds the reference (d) in the early phase and falls behind in the late development phase. For example, in mixture with beech, larch growth can be accelerated in the juvenile state through competition reduction, whereas it can be slowed down by European beech in the mature phase due to the increasing height, crown extension, and overshading effects. This means an earlier culmination of volume growth due to the rather solitary conditions of an early-successional species when combined with a late-successional species.
Figure 6.8e (bold line) shows the opposite case. Neighbouring trees might reduce the growth level compared with dominant trees and delay the time of culmination. Suppression of Norway spruce by Scots pine is a common example of this growth response pattern in species mixing (Wiedemann 1951; Christmann 1939).
6.5 Crown Size and Morphology in Mixed Compared with Monospecific Stands
6.5.1 Interspecific Variation in Crown Shape and Size
Tree species can differ considerably in crown shape and size (Fig. 6.9). The allometric equation in logarithmic (ln(y) = a + α × ln(x)) or untransformed version (y = e a × x α) can be used to compare different species regarding their crown size and scaling of the crown to tree size (Niklas 2004). Factor a represents a multiplicative effect of species on the crown expansion. Species-specific differences of exponent α signify an exponential difference in the dynamic of the crown expansion with increasing size (Box 6.3).
To show the interspecific variation in tree growing area, we used the dataset compiled by Pretzsch and Dieler (2012) which includes 126 yield tables of 52 species, of which 30 are of angiosperm and 22 of gymnosperm taxonomy. Species included the genera Abies, Acer, Alnus, Betula, Carpinus, Castanea, Cunninghamia, Eucalyptus, Fagus, Fraxinus, Juglans, Larix, Nothofagus, Picea, Pinus, Populus, Prunus, Pseudotsuga, Quercus, Robinia, Shorea, Thuja, and Tilia. From these yield tables, we derived among others the mean tree diameter, \( \overline{d} \), and tree stand area, \( \overline{sa} \). The dataset includes only yield tables with self-thinning conditions or light and moderate thinning regimes where crown cross-section area, \( \overline{cpa} \), is scaled proportionally to tree stand area, \( \overline{sa} \), (\( \overline{cpa}\cong \overline{sa} \)). Note that light and moderate thinning permanently maintain a stand’s canopy in a structure in which the tree crowns touch each other but barely overlap (Verein Deutscher Forstlicher Versuchsanstalten 1873, 1902).
Figure 6.10a shows the considerable variation in the allometry between stand area and tree diameter; the intercepts amount to a = − 1.96, − 3.57, 2.47 (mean, min, max) and the slopes to α = 1.47, 0.14, 2.33 (mean, min, max). According to Fig. 6.10, angiosperms (ln(sa) = − 1.33 + 1.36 × ln(d)) need on average more growing area than gymnosperms (ln(sa) = − 2.03 + 1.52 × ln(d)). Angiosperms with a tree diameter of d = 25 cm, for example, occupy on average sa = 19.51 m2, while gymnosperms occupy just sa = 16.90 m2, i.e. significantly (p < 0.05) less tree stand area. The fact that species can differ in the intercept of their allometric crown relationship is represented, e.g. in yield tables, by species-specific tree numbers per ha at a given stand age (Assmann and Franz 1965) and by species-specific levels of the self-thinning lines (Pretzsch 2006).
Overview of relationship between mean tree stand area, \( \overline{sa} \), and quadratic mean tree diameter, d q, in even-aged stands for 52 tree species of which 30 are of angiosperm (a and grey) and 22 of gymnosperm (g and black) taxonomy according to Pretzsch and Dieler (2012). (a) The mean \( \overline{sa} \)-d q-line of angiosperms differs significantly (p < 0.05) from the mean line of gymnosperms in its intercept, but not in its slope. (b) Mean tree stand area requirement for trees with d = 25 cm calculated on the basis of the species-specific \( \overline{sa} \)-d q-allometries shown in (a). For further statistical characteristics, see Pretzsch (2014)
The species-specific growing space requirement is relevant for calculation of the mixing proportions, stand density, and quantification of mixing effects on growth. If the species-specific growing space requirements are not considered and the calculation of the mixing proportions is simply based on mean tree stand area or tree numbers, this can result in flawed diagnoses of overyielding at the stand level and especially at the species level (see Chap. 4, Box 4).
Note that in this section the relationships between crown width and stem diameter are represented by the allometric relationship between crown projection area, cpa, and stem diameter at breast height, d, in logarithmic (ln(cpa) = a + α × ln(d)) or untransformed (cpa = e a × d α) version. When crown diameter, cd, or crown radius, cr, is required, they can be calculated as follows: \( cd=2\times \sqrt{cpa/\uppi} \) and \( cr=\sqrt{cpa/\uppi} \), respectively.
6.5.2 Intraspecific Variation in Crown Size
The crown size and shape of a tree are strongly determined by the local environment in the stand that has prevailed in the past and is now present. While maximum crown extension is achieved under solitary growing conditions, crown width decreases from open-grown (o) to dominant (d) and suppressed trees (s) (Fig. 6.11a). Stand density and competition can considerably modify the crown allometry, e.g. the cd–d relationship (Fig. 6.11b). The extent to which a tree may vary in crown extension in different environments is a crucial requirement for its success in coping with crowding.
Schematic representation of the effect of stand density on the lateral crown expansion. (a) Crown width of an open-grown, solitary tree (o), a dominant tree growing under medium stand density (d), and in suppressed conditions under self-thinning conditions (s). (b) Allometric relationship between crown diameter, cd, and stem diameter, d, for open-grown, dominant, and suppressed trees
In the following, we use 2346 crown projection area measurements of beech crowns in monospecific stands with spacing and thinning ranging from solitary growing conditions to moderate thinning and self-thinning, to illustrate the intraspecific variation (Fig. 6.12) (see Pretzsch 2014). The stand age ranges from 57 to 207 years; the surveys come from the experiments Arnstein 638, Gerolzhofen 627, Fabrikschleichach 15, Hain 27, Starnberg 91, Waldbrunn 105/106, and Zwiesel 111 in Bavaria in the years 1980–2004. Regressions for the upper 95% quantile (cpa = 3.03 × d 0.92), the group of moderately and heavily thinned stands (cpa = 1.05 × d 1.01) and the slightly and unthinned plots (cpa = 0.21 × d 1.36) describe the broad variation (Fig. 6.12, upper, middle, and lower regression line). According to these allometric equations, a beech with 25 cm stem diameter occupies 58 m2 when growing without lateral restriction, 27 m2 under medium stand density, and 16 m2 when growing in close to self-thinning conditions. This plasticity equips beech with high competitive strength in intra- and interspecific environments.
Allometric relationship between tree crown projection area, cpa, and stem diameter, d, of European beech derived from crown measurements on long-term experimental plots in Germany. As the database includes solitary trees as well as trees in thinned and unthinned stands of all ages, the ln(cpa) − ln(d) relationships can be derived for solitary trees (upper line: 95% quantile regression: ln(cpa) = 1.11 + 0.92 × ln(d)), for trees in moderately heavily (B/C-grade, n = 1685) thinned stands (middle line: ln(cpa) = 0.05 + 1.01 × ln(d)), and for trees in unthinned stands (A-grade, n = 661) (lower line: ln(cpa) = − 1.59 + 1.36 × ln(d))
In terms of a species’ acclimation to intra- and interspecific competition, the maximum and minimum of its crown extension are of special interest as these indicate the range of plasticity. Figure 6.13a shows exemplarily for maple the upper (95%) and lower (5%) quantile of the cpa–d allometry in addition to the mean relationship. The ranking of species in terms of their 95% quantile is different. Species such as lime tree and silver birch lose when ranked in terms of their 95% quantile, whereas rather plastic species such as beech and silver fir win in this ranking. This emphasises that deriving crown behaviour for solitary conditions from average behaviour in closed stands, especially from monocultures, is questionable. For example, beech, elm, maple, and silver fir profit considerably in crown expansion when released from competition in monospecific or mixed stands.
Crown projection area-stem diameter relationship of various tree species. (a) Example derivation of the upper (95% quantile), mean, and lower (5% quantile) ln(cpa)-ln(d) relationship for sycamore maple (Acer pseudoplatanus L.). (b) Interspecific variation in the mean ln(csa) − ln(d) relationships. The model ln(cpa) = a + α × ln(d) was fitted by OLS to the data and yielded the following parameters (a, α) for mountain elm (1.30, 0.67), European beech (0.18, 1.02), lime tree (0.08, 0.98), sycamore maple (–1.96, 1.55), hornbeam (–0.25, 1.24), silver fir (0.37, 0.82), rowanberry tree (1.13, 0.40), black alder (–0.85, 1.22), silver birch (–1.69, 1.48), Scots pine (–1.41, 1.27), Norway spruce (–1,43, 1.20), European larch (–1.69, 1.32), sessile oak/ common oak (–2.48, 1.64), and common ash (–3.02, 1.79). (c) Interspecific variation in the 95% quantile of the ln(cpa) − ln(d) relationships.1mountain elm (Ulmus glabra Huds.), 2European beech (Fagus sylvatica L.), 3lime tree (Tilia cordata Mill.), 4sycamore maple (Acer pseudoplatanus L.), 5hornbeam (Carpinus betulus L.), 6silver fir (Abies alba Mill.), 7rowanberry tree (Sorbus aucuparia L.), 8black alder (Alnus glutinosa (L.) Gaertn.), 9silver birch (Betula pendula Roth), 10Scots pine (Pinus sylvestris L.), 11Norway spruce (Picea abies (L.) Karst.), 12European larch (Larix decidua Mill.), 13sessile oak/common oak (Quercus petraea (Matt.) Liebl./Quercus robur L.), and 14common ash (Fraxinus excelsior L.)
Figure 6.14 illustrates that crown plasticity can differ considerably between tree species. The data comes from crown measurements on long-term experimental plots in Germany and covers a broad range of tree ages and densities, in monospecific and mixed stands (Pretzsch et al. 2015; Pretzsch 2014). Based on the 95% and 5% quantile of the cpa–d allometry (Fig. 6.14, upper and lower lines), the following relative measure, CPL, for tree crown plasticity can be derived. Based on the lower quantile line (\( {cpa}_{5\%}={a}_{5\%}\times {25}^{\alpha_{5\%}} \)) and the upper one (\( {cpa}_{95\%}={a}_{95\%}\times {25}^{\alpha_{95\%}} \)), and using a reference stem diameter of 25 cm (where stand density in monolayered stands is rather high or even at maximum), CPL is formulated as follows: \( CPL={cpa}_{95\%,25}/{cpa}_{5\%,25}=\left({a}_{95\%}/{a}_{5\%}\right)\times {25}^{\alpha_{95\%}-{\alpha}_{5\%}} \). For a tree with a reference diameter of 25 cm, CPL indicates how wide a crown can range in solitary conditions in relation to maximum restriction. By setting the 95% in relation to the 5% width, any species-specific differences in shape and form (e.g. that beech crowns are a priori wider than spruces) are eliminated. CPL only indicates the relative potential for expansion which is of particular importance for competing in mixture.
Allometric relationships between stem diameter, d, and crown projection area, cpa, for European beech (n = 14,898), silver fir (Abies alba Mill.) (n = 1079), sessile/common oak (Quercus petraea (Matt.) Liebl. and Quercus robur L.) (n = 4485), Norway spruce (n = 10,724), sycamore maple (n = 942), and Scots pine (n = 1609) in even-aged and uneven-aged stands. Range of crown dimensions measured on long-term experimental plots which cover dense as well as very sparsely spaced stands. The upper and lower lines represent the 95% and 5% quantile regression ln(cpa) = a + α × ln(d). The width of the scattering and the distance between the 95% and 5% quantile regression represents the crown plasticity. For the statistical characteristics of the quantile regressions, see Pretzsch (2014)
Analyses of CPL values for various tree species in Europe revealed a maximum value for European beech of CPL = 5.1, i.e. upper crown area is more than fivefold compared to its lower crown area under strong competition. With respect to CPL, the species represented in Fig. 6.14 are ranked as follows: beech (CPL = 5.1) > silver fir (4.7) > oak (4.5) > spruce (4.2) > maple (4.0) > pine (3.7). Out of the set of 14 species represented in Fig. 6.13b and c, beech (5.1) has the highest value, whereas alder (2.8) and birch (2.6) have the lowest.
6.5.3 Modification of Tree Allometry in Interspecific Local Environments
6.5.3.1 Shift in Crown Allometry in Interspecific Compared with Intraspecific Environments
Species mixing can considerably reduce a tree’s competition and increase its lateral crown expansion even when the stand density in mixture equals that in monoculture (Dieler and Pretzsch 2013). Of special relevance for stand dynamics, growing area efficiency and stand productivity are the behaviours of the lateral and vertical crown extension in mixed versus monospecific stands. This determinates both a tree’s growth and its space occupation and competition pressure on its neighbours.
We use data from Bavarian long-term experiments in monospecific and mixed stands of Norway spruce and European beech (Pretzsch and Schütze 2009) to scrutinise any shift in morphology caused by intra- versus interspecific competition. Figure 6.15 shows that the lateral and vertical crown extension of (a) Norway spruce and (b) European beech is higher in mixed than in monospecific stands.
Crown allometry of (a) Norway spruce and (b) European beech in mixed stands (grey) compared with monospecific stands (black). Mixing significantly increases lateral and vertical crown extension in terms of the relationship between crown projection area, cpa, and tree diameter, d, and between height to crown base, hcb, and tree diameter, d. It hardly changes the relationship between tree height, h, and tree diameter, d. The respective equations are for spruce monoculture cpa = 0.05 × d 1.61, hcb = 3.04 × d 0.52, h = 2.78 × d 0.68; for spruce in beech cpa = 0.09 × d 1.49, hcb = 2.19 × d 0.57, h = 2.96 × d 0.64; for beech monoculture cpa = 0.24 × d 1.43, hcb = 5.14 × d 0.31, h = 3.00 × d 0.64; and for beech in spruce cpa = 0.46 × d 1.29, hcb = 1.22 × d 0.67, h = 3.09 × d 0.64
Scaling between cpa and d is mostly considerably steeper as predicted by West et al. (2009) for the allometrically ideal plant (α cpa, d = 4/3 in cpa ∝ d 4/3). It is shallower in mixed than in monospecific stands but lies at a higher level in mixture (Fig. 6.15a). With increasing size, the collectives become more similar in this regard, i.e. in young and middle-aged stands, the species profit from interspecific competition but in mature stands, where crowns are less restricted, crown extensions become similar. According to allometric theory (Enquist et al. 2009; West et al. 2009), scaling between hcb and d should be hcb ∝ d 2/3. That applies to neither of the species investigated here; the exponents are mostly lower. The intercepts of trees in mixed stands differ considerably from those in monospecific stands; crowns are significantly longer in mixed compared with monospecific stands. The h–d allometry, in contrast, is only slightly modified by species mixing, and the observed scaling is close to h ∝ d 2/3 as predicted by the allometric theory. Stem taper may be different in mixed versus monospecific stands; however, the h–d relationships are similar. Note that by comparing the species’ behaviour based on their scaling size, differences are eliminated. This contrasts with less indicative and questionable comparisons based on ratios (see Box 6.3).
Box 6.3 Allometric Relationship Between Tree Dimensions, Ratios, and Ontogenetic Drift
As an example for size relationships between tree organs, Box Fig. 6.3-1 shows the allometric relationship between tree diameter, d in (cm), and crown diameter, cd in (m). It is linear at the ln–ln scale (Box Fig. 6.3-1a) and degressive with α cd, d < 1.0 at the logarithmic scale (Box Fig. 6.3-1b).
(a–c) Analysis of the allometric relationship between tree crown diameter, cd in (m), and stem diameter, d in (cm), and the resulting ratio cd/d for Norway spruce. (a) Based on n = 3974 crown records, OLR regression yields ln(cd) = − 1.24(±0, 018) + 0.75(±0.006) × ln(d) (R 2 = 0.82). (b) The same relationship shown in linear grid and regression line cd = 0.289 × d 3/4. (c) Ontogenetic drift in the relationship between cd/d and tree size (cd/d = 0.289 × d −1/4)
The allometric theory assumes cd ∝ d 2/3 (Enquist et al. 1998; West et al. 2009); however, our empirical evaluation based on crown measurement of n = 3974 Norway spruces yielded ln(cd) = − 1.24(±0, 018) + 0.75(±0.006) × ln(d), i.e. cd = 0.289 × d 3/4 with α cd, d = 0.75. The OLR regression was based on the ln-transformed values, according to Niklas (1994, p 331) RMA slope is α RMA = α OLS/r y, x with r y, x = r cd, d = 0.95 as correlation coefficient between cd and d. In this case, α RMA = 0.75/0.95=0.79 deviated even more from the slope for the allometrically ideal plant predicted by allometric theory (α RMA = 2/3 = 0.66).
From cd = 0.289 × d 3/4, it follows that cd/d ∝ d −1/4, i.e. the ratio decreases with tree size. Assmann (1970, p 112) used this ratio to compare crown morphology within and between different species. The non-isometric cd–d relationship shows that the ratio changes simply with size due to the ontogenetic drift of the tree size proportions. That means that comparison can be misleading when trees in mixed and monospecific stands differ in size. As the ratio decreases with size (Box 6.3), any size differences between the compared trees in mixed versus monospecific stands should be eliminated before comparison (see Sect. 6.7.2).
In this case, the observed cd/d ratio of the trees in mixture can be extrapolated to the size of neighbouring monocultures by cd/d ′ = cd/d × (d p /d m )−1/4. After this transformation, the adjusted observed ratio (cd/d ′) can be compared with the observation in neighbouring monocultures.
Analysis of cpa–d allometry of beech in monocultures compared with beech in mixture with Norway spruce, European larch, common ash, and sessile oak shows striking differences (Fig. 6.16). This analysis is based on densely stocked stands with no or only light thinning. Mixing matters even when the stand density is at maximum. Obviously a neighbouring European beech restricts the crown of a beech more than any other of the analysed species. For a beech with stem diameter 25 cm, the allometric equation shown in Fig. 6.16 (be) predicts a crown projection area of cpa = 17 m2. Beeches with the same stem diameter achieve cpa = 25 m2 when mixed with ash, and 27, 37, or even 45 m2 when mixed with spruce, larch, or oak, respectively. The ranking of neighbours regarding the effect on crown restriction is beech > ash > spruce > larch > oak. For European beech, mixing with each of the other species means ‘competition reduction’ in terms of crown extension in the sense of Kelty (1992) and Vandermeer (1992). In other words, a neighbouring Norway spruce, sessile oak, or Scots pine means a relief in crown restriction compared with a neighbouring beech. This is in accordance with findings by Pretzsch and Biber (2005) and Zeide (1985) that self-thinning is the highest in beech monocultures and much lower in monocultures of the other three species, with the ranking European beech > Norway spruce > Scots pine > sessile oak.
Allometric relationship between crown projection area, cpa, and the tree diameter, d, for European beech in monocultures (blue) and shift in the allometry when beech is mixed with Norway spruce, European larch, ash, and sessile oak. The respective equations are cpa = 0.12 × d 1.54 for be; csa = 2.17 × d 0.76 for be, (ash); csa = 0.75 × d 1.21 for be, (la); cpa = 0.84 × d 1.08 for be, (sp); and cpa = 1.30 × d 1.10 for be, (oak)
While allometric scaling theory predicts cpa ∝ d 4/3 (i.e. α = 1.33) for the allometrically ideal plant, the allometric exponent is at a maximum α = 1.54 in beech monocultures and ranges between α = 0.76 (be, (ash)) and α = 1.21 depending on the species composition of the neighbours.
6.5.3.2 Root Morphology and Root-Shoot Allometry
According to the optimal partitioning theory (McCarthy and Enquist 2007), the shape of the tree crown, root system, and the relationship between these depend highly on the resource supply of the plant. Part of the large variation in the root-shoot relationship of plants can be explained by this theory. It predicts that the limitation of a resource leads to the promotion of growth of the plant organ responsible for supplying that critical resource (Comeau and Kimmins 1989; Keyes and Grier 1981).
In Fig. 6.17, four examples indicate the behaviour of the root-shoot ratio. In the first example, where light, water, and nutrient conditions are favourable (Fig. 6.17a), the tree develops a root-shoot ratio of 10:90. Under adequate light conditions, but with a water or nutrient deficiency (Fig. 6.17b), the tree invests more in root growth, especially fine roots. Thus, the root-shoot ratio increases in favour of roots to 45:55. With an adequate water and nutrient supply combined with critical light conditions, e.g. on nutrient-rich soils or for understorey trees (Fig. 6.17c), shoot growth is enhanced so that the tree root-shoot ratio may become 30:70. In the case of rich soils, trees in the upperstorey tend to allocate growth resources to extensive crown development, whereas trees in the understorey often invest in height growth to escape the shade (Oliver and Larson 1996). If light, water, and nutrient supply is limited (Fig. 6.17d), the root-shoot ratio may resemble the example in Fig. 6.17a of 10:90.
Partitioning of total plant biomass on shoot and root organs in relation to supply of nutrients and water (x-axis) and supply of light (y-axis). Limitation of nutrient and water supply causes a partitioning in favour of roots. Limitation of energy supply increases the investment of biomass in above-ground organs (By courtesy of Kimmins (1993, p 13))
Changes in the partitioning between root and shoot growth can indicate a modification of environmental conditions through mixing as shown for the rather easily accessible crown growth. Intraspecific variation in root morphology due to species mixing, however, is more difficult to measure. Analysis of root-shoot allometry based on tree-ring analyses at increment cores from stem and coarse roots can reveal how the root-shoot relationships depend on site conditions (Pretzsch et al. 2012a, b) and silvicultural treatment (Pretzsch et al. 2014). For the methodology see Box 6.4. Figure 6.18 shows results of combined increment boring of coarse roots and stem at breast height applied in mixed spruce/beech stands versus monospecific spruce stands.
Development of coarse root diameter, dr, in relation to stem diameter, ds, of Norway spruce in mixture with European beech compared with neighbouring monospecific stands of Norway spruce on the long-term experimental plots (a) SON 814 and (b) ARN 815. The straight lines represent the allometric linear regression lines ln(dr) = a + α × ln(ds) for spruce in mixed stands (grey) and monospecific stands (black)
At both sample sites in the prealpine lowlands in Bavaria (SON 814 and ARN 815), the mixture with beech causes a significantly shallower root-shoot allometry of spruce compared with neighbouring monospecific stands. In the size range of 20–50 cm stem diameter, which is well backed with sample trees (right sections of the regression lines), spruces in mixture have a significantly lower root-stem growth than in monospecific stands. Coarse root growth is more sluggish in growth reaction than the resource-capturing fine roots. However, as in the case of the crown structure, coarse roots indicate the long-term dynamics as they represent the holding fixture and connecting pipes for fine roots. A cause of the repartitioning from root to stem growth in mixed stands may be a better supply of below-ground resources in mixed versus monospecific stands (Rothe 1997; Rothe and Binkley 2001) which triggers crown expansion to remedy above-ground resource limitation. Spatial niche separation between spruce roots in the upper soil horizon and beech roots in the deeper layer may be an additional reason (Rothe 1997; Wiedemann 1942, 1951). Decrease in root diameter growth entails shorter root length and a smaller influence zone of a tree. This may indicate a reduced investment in mechanical stabilisation and a decrease in the tree’s growing area in mixed versus monospecific stands (see Sect. 6.6).
Box 6.4 Analysing the Allometric Relationship Between Coarse Root and Stem Growth of Trees in Mixed Versus Monospecific Stands
To analyse the allometric relationship between coarse root and stem growth increment, cores can be sampled from both stem at breast height and coarse roots in close distance from the stem axis (Pretzsch et al. 2014; Nikolova et al. 2011). For each trunk, two increment cores may be sampled from the cardinal direction north and east (Box Fig. 6.4-1, left). In addition, the trees’ two to three biggest coarse roots can be sampled. Because of their rather elliptical cross section, two increment cores may be taken from each root at a 45° angle from the vertical line. To enable the sampling, the selected roots need to be partly excavated (Box Fig. 6.4-1, right).
Based on tree-ring analysis, the diameter development of coarse roots may be plotted over the stem diameter development for each tree in a double-logarithmic scale. The grey trajectories in the background of Box Fig. 6.4-2 show such root-shoot allometries exemplarily for European beech and Douglas-fir grown in monospecific and mixed-species stands (Thurm et al. 2016).
Compared with monospecific stands (a and d), in mixed-species stands the growth of coarse roots in European beech (b and c) and Douglas-fir (e and f) in relation to stem growth is reduced. In monocultures of European beech (a) and Douglas-fir (d), the allometric relationship between coarse root growth and stem diameter growth is steepest. The graphs show the results of increment boring at coarse roots and stem of European beech (n = 85) and Douglas-fir (n = 90) in 50–100-year-old monospecific and mixed-species stands on moist and nutrient rich sites in southern Germany. For the double-logarithmic allometric model, coarse root diameter = f (stem diameter, mixing proportion, tree species), see Thurm et al. (2016)
The regression analysis of the individual coarse root diameter-stem diameter trajectories revealed that both European beech and Douglas-fir have reduced coarse root growth in mixed compared with monospecific stands (Box Fig. 6.4-2c versus a and f versus d). In both monocultures (Box Fig. 6.4-2a and d), the allometric relationship between coarse root growth and stem diameter growth is the steepest, i.e. the investment in roots compared with stem is maximal.
Certainly, the selected coarse roots represent just a small portion of the trees’ whole root system, and their development is more sluggish and persistent than that of the ephemeral fine roots. But analogously to the stem which indicates the development of the crown and leaf area, the coarse root diameter reflects the activity of the whole root, since the coarse roots ultimately provide the basic structure and pipe system for the fine roots. So, the coarse root growth might be used as an integrative and non-specific indicator for the root system as a whole.
6.5.4 From Pattern to Process of Crown Expansion in Interspecific Neighbourhoods
6.5.4.1 Dynamics of Lateral Crown Expansion in Inter- Versus Intraspecific Environments
The change in intraspecific crown morphology in mixed versus monospecific stands becomes even more obvious by analysing successive crown maps of monospecific and mixed stands (Pretzsch 1992, 2009). Based on long-term plots with first crown measurements in the 1950s and successive measurements every 10–20 years, a 3D model delivers distances between neighbouring crowns (Fig. 6.19). The successive eight-radius crown measurements (Box 6.5) provide lateral crown growth for all eight directions. This enables derivation of functions for predicting lateral crown growth in dependence on the distance to neighbouring crowns in the respective directions (Fig. 6.20).
Determination of the lateral crown restriction of individual trees: distances from the crown perimeter of tree A to the neighbouring crowns of the trees B, C, and D are determined in eight directions d1 … d8. When crowns intersect, the distance can become negative (see Fig. 6.20)
Influence of the distance to the neighbouring crown on the relative increment of the crown radius (Pretzsch 2009). (a) Radial crown increment of Norway spruce in a Norway spruce (N. sp → N. sp.) and a European beech (N. sp. → E. be.) neighbourhood. Functions: icrsp→sp = 1.0−e−3.84 (dist+1.2) and icrsp→be = 1.0−e−1.71 (dist+2.7). (b) Radial crown growth of European beech in a European beech (E. be. → E. be.) and a Norway spruce (E. be. → N. sp.) neighbourhood: icrbe→be = 1.0-e−2.56 (dist+1.8) and icrbe→sp = 1.0−e−1.49 (dist+3.1)
The functions describe the relative changes in crown radii of Norway spruce and European beech in relation to the crown distance to neighbours. A relative increment of crown radius of 1.0 means that a tree can expand its crown radius without lateral restriction and achieve the potential crown radial increment. A relative increment of 0.0 or less, by contrast, means stagnation and degeneration of the crown.
The curves in Fig. 6.20 show the decline in crown radial increment as the central tree crown becomes increasingly interlocked with neighbouring crowns as well as in the crown distance at which a crown dieback commences (below the dotted 0-line). Based on these distance-dependent crown growth functions, the crown cross-sectional development at a trial plot can be simulated for each individual tree in relation to its neighbourhood. Figure 6.21 shows the crown map for a 20 m × 20 m section of the experimental trial plot ZWI 111/3 in 1954, the year the simulation run starts based on the crown measurements in this year. The crown development of Norway spruce and European beech, at this time 60 and 80 years old, respectively, was simulated for a 50-year period at 5-year intervals. The interim results at times t = 10 years, t = 30 years, and t = 50 years (Fig. 6.21b, c, and d) show how the crowns expand and respond to increasing restriction by developing oval and eccentric crown shapes. To develop the crown perimeter, the eight crown radii are connected by a cubic spline (Pretzsch 2009).
Crown cross-sectional area development on the experimental plot Zwiesel 111/3 based on simulation runs with the individual tree growth simulator SILVA 2.0 (Pretzsch 2009) starting with the crown measurement in the year 1954. The crown map for a 20 m × 20 m section of the experimental trial plot Zwiesel 111/3 is presented in a 50-year simulation run at the times (a) t = 0, (b) t = 10 years, (c) t = 30 years, and (d) t = 50 years
The simulations and underlying functions (Fig. 6.20) reveal the following species-specific competition behaviour: when a Norway spruce grows towards another Norway spruce, the decrease in radial increment after an overlap of 1.0 m is almost linear. Yet, if it grows towards a European beech, a decrease in radial increment of the crown occurs much later. Effectively, this leads to a tighter interlocking of a Norway spruce crown with neighbouring European beech than with neighbouring Norway spruce (Pretzsch 2014). Similarly, the crown interlocking for a European beech growing towards a Norway spruce is also tighter than for European beech in monocultures. This finding confirms the species-specific behaviour of beech shown in Fig. 6.16. For Norway spruce and European beech, a reduction in lateral crown expansion of more than 5% only occurs when crowns interlock by 1.0–2.0 m, and the crowns recede only after the crowns overlap by 2.0–3.0 m. Thus, the crown growth dynamics of Norway spruce and European beech differ considerably from, e.g. the response of the light-demanding species European larch, which shows a clear reduction in the growth of side branches once the distances from branches to the neighbouring crown fall below 40 cm (Schütz 1989).
6.5.4.2 Variability of Crown Projection Area in Inter- Versus Intraspecific Environments
Solitary trees achieve wide and, apart from a slight tendency towards ovality due to one-sided solar irradiation in northern or southern latitudes, rather circular crowns. The symmetry of their crowns indicates unimpeded lateral expansion or at least all-round homogeneous restriction by, for example, water, light, or nutrients (Møller and Swaddle 1997). When coping with crowding, crowns may adapt their lateral extension to their prevailing neighbourhood conditions and increasingly lose the symmetry typical for solitary growth. Species with higher crown plasticity can to some extent overcome their restriction by occupying emerging niches, penetrating neighbouring crowns, or even edging out neighbours. The eccentric crown expansion and crown asymmetry which enables occupation of additional space by directional lateral crown expansion should be distinguished from degeneration-caused asymmetry through die-off and mechanical abrasion of branches and crown parts due to overwhelming competition.
Box 6.5 Measures for the Variability of Lateral Crown Expansion of Individual Trees in Intra- and Interspecific Environments
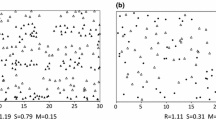
The following measures of crown plasticity are based on common crown maps with eight-radius crown projection areas as shown in Box Fig. 6.5-1. The graph shows exemplarily a 400 m2 sized section of the 80-year-old triplet Freising 813/2 of Norway spruce, European beech, and a mixture of both species close to Freising in southern Bavaria. The crown projection areas (in m2 per crown) and their shape (divergence from circle) can be compared between mixed and neighbouring monocultures to reveal inter- versus intraspecific crown variability.
Crown maps with eight crown radii as basis for quantifying the variability of lateral crown expansion of individual trees. 400 m2 sized sections of the 80-year-old triplet Freising 813/2 of Norway spruce (a), a mixed stand of Norway spruce and European beech (b), and European beech (c) close to Freising in southern Bavaria
The following three measures address different aspects of the variability of lateral crown expansion (see Box Fig. 6.5-2):
The ratio cpa/sa between crown projection area, cpa, and stand area of an individual tree, sa, indicates the degree of crown engagement. A value of cpa/sa = 1 would indicate identity of cpa and sa. High values (cpa/sa > 1) indicate wide crown expansion reaching beyond the trees’ sa. Small cpa/sa values indicate a tree’s suppression and crown recession (see Box Fig. 6.5-2 from left to right).
The ratio between the shortest and longest of the eight crown radii, r min/r max, indicates the crown rotundity. In Box Fig. 6.5-2 the rotundity decreases (from left to right) with stand density due to growing competitive pressure.
Crown projection area (grey area) and growing area (black line) of trees in (left) low density, (centre) medium density, and (right) high density with decreasing crown symmetry and measures for quantifying crown variability. cpa/sa, cpa-sa ratio represents crown extension in relation to stand area. r min/r max, crown rotundity in terms of the ratio between the largest to shortest radius. ecc, Crown eccentricity indicated by the standardised distance between the gravity centre of the crown projection area and the tree position
Crown eccentricity, \( ecc=\sqrt{{\left({x}_s-{x}_g\right)}^2+{\left({y}_s-{y}_g\right)}^2}/{d}_{1.3} \), is based on the Cartesian coordinates of the stem position, x s and y s , and on the coordinates of the centre of gravity, x g and y g , of the crown, calculated on the basis of the coordinates of the corner points of the crown projection area (\( {x}_g={\sum}_{i=1}^8{x}_i/8 \), \( {y}_g={\sum}_{i=1}^8{y}_i/8 \)). Division of the distance \( \sqrt{{\left({x}_s-{x}_g\right)}^2+{\left({y}_s-{y}_g\right)}^2} \) by the stem diameter, d 1.3, eliminates size effects and makes different trees and species comparable. A measure of, for example, ecc = 0, 2.0, and 10.2 means that the gravity centre of the crown is perpendicular above the tree position (ecc = 0), twice (ecc = 2), or even more than ten times (ecc = 10) the stem diameter deviating from the tree position (see Box Fig. 6.5-2 left to right). Note that in Box Fig. 6.5-2 the trees’ centres of gravity are represented by circles and the trees’ stem foot positions by the origin of the coordinate system.
In the following, crown maps of monocultures and mixed stands of Norway spruce and European beech and sessile oak and European beech on long-term plots in Germany with eight-radius crown measurements are used to illustrate the differences between crown dynamics in intra- and interspecific environments. In order to quantify the variability of crown structures in those stands, the measures introduced in Box 6.5 will be applied.
Using the ratios, the crown engagement, rotundity, and eccentricity can be compared between mixed and neighbouring pure stands. The cpa/sa ratios in Table 6.1 reflect that, except for beech when mixed with oak, all three species can significantly expand their crowns laterally far beyond their stand area when growing in mixed compared with pure stands.
The ratio r min/r max reveals that, compared with the other species, beech crown projection areas are mostly less circular, i.e. more jagged. Mixing significantly increases the rotundity in the case of spruce (p < 0.001), decreases the rotundity of beech (p < 0.001), and leaves the rotundity of the crowns of sessile oak unaffected.
The ecc values are the highest for beech, especially when growing in mixed stands. Values of ecc = 5.7 to 7.4 indicate that beeches have plastic crowns for resource capture even distant from their stem position. The measures show that crown morphology can significantly shift from intra- to interspecific competition and trigger the space occupation of the combined species in a species-specific way. Mixing increases the crown eccentricity of both beech (p < 0.001) and oak (p < 0.001) but fosters the centricity in the case of Norway spruce (p < 0.001).
6.5.4.3 Species Mixing and Inner Crown Properties
Species mixing can obviously modify the fractal dimension of the crown surface area and leaf area from a lower space filling (Euclidian scaling) towards higher space filling of the crown volume (fractal scaling) (Pretzsch and Dieler 2012). Tree crowns as described by Oldemann (1990), Purves et al. (2007) and Roloff (2001) lie somewhere along the continuum between the borderline cases of an umbrella-like crown with the majority of the leaf surface area allocated close to the convex hull (surface dimension n = 2) and a broom-like crown with leaf surface area distributed all over the crown space (surface dimension n = 3) (Zeide 1998).
Terrestrial laser scanning and subsequent skeletonisation of tree crowns based on the voxel patterns revealed significantly different inner crown structures in mixed versus monospecific stands of Norway spruce and European beech (Bayer et al. 2013). An example of the procedure from compilation of point clouds to derivation of inner crown properties is shown in Fig. 6.22a–d.
(a–d) TLidar scans in mixed-species stands of Norway spruce and European beech, skeletonisation, and measurement of crown periphery and inner structure. (a) Original voxel cloud compiled from various scans, (b) skeletonisation, (c) extracted branch pattern, (d) measurement of outer and inner crown characteristics (according to Bayer et al. 2013)
Although similar in stem diameter and tree height, beeches growing in mixture with spruce can have significantly more branches of first order and—because of stronger ramification—nearly twice the number of second and third order branches compared with those in monocultures (Bayer et al. 2013). The branch bending can be higher and the branch angle flatter. Also, the sum of branch length and the crown volumes can be higher in mixed compared with monospecific stands. The schematic representation in Fig. 6.23 accentuates the broom-like crown shape of beeches when growing in intraspecific interaction (left) with most of the leaf area allocated close to the top of the convex hull. In interspecific environments, the crowns are rather bouquet-like (right) with the leaves reaching deeper into the crown volume (Arz 2013; Bayer et al. 2013).
Schematic representation of (left) broom-like crown structure of adult European beech in even-aged monocultures and (right) bouquet-like crown structures in mixed stands with spruce (according to Arz (2013)). While individuals growing in monocultures develop steeper branches, those growing in mixture with Norway spruce have more branches, flatter branches, and nearly twice the number of second and third order branches (Bayer et al. 2013)
6.6 From Tree Morphology to Space Occupation
All individuals in monocultures—particularly when they are relatively homogeneous in genetics and size—use similar strategies to compete for the growing space in a stand and for the same resources when these are not sufficient for all. When their demands and abilities are relatively similar (Fig. 6.24a), the function and structure of all competitors vary within a small range. Theoretically, this leads to a hexagonal pattern of tree distribution. Given that all are equipped with rather similar tricks and traits, neighbouring trees are in a stalemate rather than outwitting each other. That is why many species, when growing in monocultures, have tree crown projection areas that are rather similar to growing area. Their crowns barely interlock and 5–10% of the stand area is not covered by crowns at all (see Chap. 5, Fig. 5.19 and Table 5.6).
Crown and root shapes of tree species can complement or prevent each other from filling the canopy and soil space. Schematic representation of beech in monoculture (a), mixing of spruce and beech (b), and mixing of beech and oak (c). Packing density can be increased compared with monoculture of beech through the complementary structure of spruce and beech (b) but not through the rather obstructive structure of beech and oak (c)
Compared with monocultures, the trees’ elbowroom in mixed stands can be wider. If the crown shapes of the combined species complement each other (Fig. 6.24b), the trees might simply have more space to occupy without mechanical abrasion or penetration of the neighbours’ crowns, while mixtures with similar crown shapes (Fig. 6.24c) cause less competition reduction. Beyond these differences in species-specific structure, the species-specific strategies for space occupation through, e.g. crown penetration due to different light compensation points and lateral expansion due to change in branch angle may result in competition release and wider crown expansion than in monoculture of one species and crown restriction and regression compared with the monospecific stands for the other species.
6.6.1 Crown Projection Area, Tree Stand Area, and Degree of Crown Engagement
Regarding the crown as a tool for resource sequestration, any crown and root plasticity may be relevant for tree and stand productivity. Suppose that if species combined in mixture retained the same morphology as they have in monoculture, the mixing effect may simply be additive. Any crown structures emerging in mixture, e.g. a plastic acclimation or complementarity of spatial niches, may cause higher resource capture and multiplicative mixing effects such as overyielding or underyielding in mixed versus monospecific stands.
The crown projection area, cpa, of a tree of a given size indicates its reach for resource acquisition, light interception, and shading of neighbours. In contrast, the tree stand area, sa, represents its available growing area. So the ratio between cpa and sa, introduced as the degree of crown engagement in Box 6.5 indicates how wide crowns reach beyond their growing area, fill the canopy space, and forage for light in their local environment.
Table 6.1 shows exemplarily for monospecific and mixed stands of spruce, beech, and oak that the cpa/sa ratio is mostly close to cpa/sa = 1.0 in monocultures, i.e. the tree crown area is similar to its growing area. In mixture, trees can overcome the lateral crown restriction by expanding and interlocking more closely. In total, the cpa/sa value increases by 24 and 39%, respectively, when spruce and beech are mixed, and by 12 and 54%, respectively, when beech and oak grow in mixture (see Table 6.1, cpa/sa values). This increase in the crown expansion beyond growing area can be coupled with a reduction in the mean growing area per tree in mixed versus monospecific stands (Sect. 6.4).
Figure 6.25a is a schematic representation showing that in monocultures crown projection area is rather similar to stand area, whereas in mixed stands the crowns often exceed the tree stand area. Due to the higher stand density in mixed compared with monospecific stands, the mean tree stand area decreases (see Chap. 4, Sect. 4.3.2). So, in mixed stands the cpa-d allometry can shift upwards and the sa-d allometry downwards, and the cpa/sa relationship, i.e. the crown engagement, often rises in mixed compared with monospecific stands (see cpam/sam > cpap/sap in Fig. 6.25b).
Schematic representation of the crown shape of beech monocultures (p) compared with mixed stands (m). While crown projection area is frequently rather similar to or just slightly lower than growing area in monocultures, mixing can considerably increase crown projection area, cpa, and slightly reduce tree stand area, sa. As a result, the cpa/sa ratio which indicates the degree of crown engagement can rise significantly when trees grow in interspecific environments
Tree size is an ambiguous trait. As trees become larger, they tend to gain more privileged access to resources, occupy more space, and have a greater repression effect on neighbours. However, size can also mean higher maintenance costs and susceptibility to windthrow, drought, or bark beetle attacks. When trees expand far beyond their stand area by spreading their crowns without much additional investment in tree diameter or tree mass, this can mean a higher resource acquisition without the drawbacks of higher resource investment. Section 6.6 will show that this can increase the growth per stand area and thereby the tree and stand productivity.
6.6.2 Dynamics of Canopy Gap Occupation
Crown plasticity is species specific. We quantified it by CPL in Sect. 6.5.2 and saw that, for instance, CPL of beech >> CPL of Scots pine. A species’ crown plasticity is relevant for its persistence and continuous extension in closed stands; it enables crown competition through penetration or even edging out of neighbouring crowns in the course of stand development. But plasticity also means a strong capacity for first access to unoccupied space in the early stand phase or spontaneous occupation of gaps caused by mortality or thinning. During stand development, various kinds of disturbances (e.g. tree mortality due to competition, storm damage, bark beetle attacks, and thinnings) may cause openings in the canopy and gaps in the root space. The quicker and wider a species’ crown extension, the greater the benefit it gains from erratic gaps. At the stand level, plastic crowns which can quickly occupy gaps can mean continuous cover of the ground and high resilience of productivity in the case of disturbances.
The following rough estimation underlines the relevance of gap occupation for interspecific competition and stand productivity. The roughly 3% annual rate of tree loss due to mortality or thinning means a gap area of 300 m2 per hectare and year. A continuation of the drop-out process of trees over a 100-year rotation period can cause a transient gap area of about 30,000 m2, i.e. 3.0 ha. In other words, a stand’s given growing area is repeatedly contested by neighbours, occupied by trees, released after mortality or thinning, and finally captured by the most competitive neighbours. Starting with some thousand (when planted) or million trees (when naturally regenerated), capture and recapture of growing area due to competition ultimately leave just a few hundred trees per hectare in the mature stand. Again and again trees drop out and release space for the remaining neighbours, which can occupy it faster and more completely the more plastic their crowns are.
Particularly in more or less monolayered stands, this ongoing mortality and subsequent closure through crown extension causes a continuous opening up of the canopy. Crown size and accordingly gap size increases with tree age. As larger gaps need more time to be recaptured and closed by neighbours and because older trees’ lateral shoot growth is rather slow, the crown re-coverage becomes more sluggish with increasing stand age. The ongoing gap dynamics are probably one additional reason why even in unthinned monolayered monocultures, 5–15% of the stand area remains uncovered by crowns (Chap. 5, Table 5.6). In temperate forests, where individual tree growth is mainly light limited, gaps are closed more rapidly through crown extension above ground than through tree root extension below ground (Bauhus 2009). Species with higher crown plasticity can achieve more additional canopy space through faster and more extensive gap occupation equivalent with additional resource capture and competitive strength. Particularly gaps caused abruptly by thinning favour those species capable of wide and fast crown extension. Additional growing space caused by mortality emerges more continuously and is more gradually occupied by neighbours already in advance of a tree’s death.
Time series of TLidar measurements before and after gap cuttings (2006, 2008, 2010, and 2012) in monospecific and mixed stands of Norway spruce and European beech reveal the species-specific strategies for occupying crown gaps caused by thinning (Bayer et al. unpublished). Figure 6.26 illustrates the initial gap size (non-black areas in the centre), the gap filling till 2008 (dark grey) and 2012 (light grey), and the still uncovered area in 2012 (white). The progressing occupation of gap area (in m2) is much slower in the monospecific spruce stand (left) compared with beech (middle). This finding corresponds with the much lower growth resilience to stand density reduction through thinning of spruce compared with beech (Pretzsch 2005b). The gap in the mixed stand of spruce and beech is closed relatively quickly; again beech contributes much more than spruce. On average, the area occupation in the 6-year period of spring 2006 to spring 2012 amounts to 0.8 m2 year−1 for spruce and 2.8 m2 year−1 for beech.
Progressing canopy closure in monocultures of Norway spruce (left), European beech (centre), and a mixed stand of Norway spruce and European beech (right) after cutting gaps in 2006 (experimental area FRE 813/1). TLidar scans show the initial state of the canopy in 2006 (black), the progressing area occupation until 2008 (dark grey) and 2012 (light grey), and the still open part in 2013 (white)
Figure 6.27a quantifies the dynamic of gap closure for the canopy cover of the ground. While spruce in the monospecific stand (sp) closes about 5 m2 during the 6-year period (2006–2012), beech (be) and the mixed stand (sp, be) close more than 15 m2, i.e. about three times this area. Figure 6.27b reveals the contribution of both species to closing the gap in the mixed stand in terms of their sum of crown area which has grown into the gap since 2006. Again, beech (17.5 m2) is superior to spruce (10 m2), and large portions of the canopy covered area are multilayered (hatched area). The high multiple crown coverage in mixed stands indicates the complementary use of light by spruce and beech.
Progressing gap area occupation on the experimental plot FRE 813/1 from 2006 (closed gap area = 0 m2) to 2012 for monospecific and mixed stands of Norway spruce and European beech. (a) Progressing gap closure in monospecific spruce (sp), monospecific beech (be), and mixed stand of both species (sp, be) in terms of canopy cover of the ground. (b) Canopy cover for the mixed plot of spruce and beech (bold line), sum of crown projection area for N. spruce (sp, (be)) and E. beech ((sp), be), and multilayering on the mixed plot (hatched area)
TLidar reveals that beech is superior in terms of both the absolute area occupation and the velocity of gap occupation. This continuous contest for space occupation is rather unspectacular, but in view of the above-stated total area of about 3 ha during a rotation, it is highly relevant for a species’ survival and spread. Efficient space occupation does not necessarily mean superior productivity but signifies an increase in dominance and persistence through restraining, penetrating, and suppressing the other species in a kind of permanent guerrilla warfare. Even in stands without active silvicultural interference, this contest for space and resources can gradually change the mixing proportion in favour of those species which are best equipped for continuous gap dynamics.
A superior crown plasticity and gap occupation of species such as European beech, maritime pine (Pinus pinaster Ait.), or lenga (Nothofagus pumilio [Poepp. U. Endl.] Krasser), respectively, is probably one essential reason for their dominance in Central European, Mediterranean, and South American forest ecosystems.
6.7 Efficiency of Tree Crown Area and Tree Growing Area
Productivity means growth per time and unit area and indicates how efficiently a tree uses a given growing area for volume or biomass growth. So far we have compared tree morphology and growth in mixed stands versus monocultures without any relation to the required growing area. The following transmission to growth per tree stand area yields tree and stand productivity.
Dividing the annual volume increment of a tree (m3 year−1) by its crown projection area or stand area (m2) (see Box 6.6) yields a measure of the tree’s area use efficiency in terms of volume growth. In this case, area is used as a surrogate variable for the tree’s resource supply.
Note that indices which set the growth in relation to the consumption of light, water, or nitrogen and water follow the same concept and are much more informative because they are directly related to the resources. Although they reflect the efficiency of light, water, and nutrient use, they are still rarely available for trees in mixed and monospecific stands.
Efficiency parameters based on growing area or growing space were frequently used to compare the performance of trees of different sizes or social classes in the stand, at different ages, or in different environments (Gspaltl et al. 2012; Pretzsch and Schütze 2009; Sterba et al. 1993; Sterba and Amateis 1998). When calculated for trees in mixed stands and neighbouring monospecific stands, efficiency parameters can contribute to quantifying and understanding the emergence of mixing effects.
Box 6.6 Crown Projection Area, cpa, and Stand Area, sa, of Trees
The crown projection area of a tree, cpa, results from projection of four or eight crowns. In Box Fig. 6.6-1a, the crown projection area is represented by a circle with the mean crown radius circle with a radius \( {\overline{r}}_q \), which corresponds to the quadratic mean of four or eight radial measurements r 1, … r n . The growing area—also called stand area—of a tree, sa, is a substitute for its resource supply (Box Fig. 6.6-1a). It results from dividing the whole-stand area (e.g. the plot area of 1 ha) by the number of living stocking trees. The sum of the tree stand areas of all trees is the stand area (\( \mathrm{AA}={\sum}_{i=1}^n{sa}_i \)). The simplest method for obtaining the mean stand area, \( \overline{sa}=\mathrm{AA}/n \), is to divide the stand area by the number of trees, n. Other methods such as the circle segment method by Alemdag (1978), the rastering methods by Faber (1981, 1983) and Nagel (1985), and the growing area polygons by Brown (1965), Jack (1968), Fraser (1977), and Pelz (1978) make additional assumptions about the trees such as their diameter, crown size, or growth rate in order to partition the total stand area between them (see Pretzsch (2009)).
The cpa/sa relationship can become greater in mixed stands and the degree of crown closure and therefore the crown closure and the sum of crown projection areas higher than in monocultures (Box Fig. 6.6-2). Wider crown extension may result in denser canopy space filling and higher tree growth per unit area.
Relationship between tree growing area, sa, and crown projection area, cpa. (a) Crown projection area, cpa, is often equal to or slightly smaller than tree stand area, sa, in monocultures. (b) Because of structural or functional complementarity, crown projection area is often larger than stand area in mixed stands. This results in multiple crown coverage (black intersections of crowns)
6.7.1 Ratios for Comparing Tree Growth in Mixed Versus Monospecific Stands
The ratios presented in Table 6.2 are useful for analysing efficiency of growth at the tree level and for the transition from growth per tree to growth per stand unit area (Box 6.6). The ratios can be quantified for trees in monospecific and mixed stands. Comparing these can reveal, for example, whether overyielding results from higher stand density, higher growth rate per tree, or from a combination of both.
The ratios E cpa/v and E iv/cpa indicate the efficiency of the crown in terms of crown space occupation and exploitation without considering the required tree stand area, sa, i.e. they reflect aspects of plant ecology. E cpa/v (m2 m−3) indicates how much crown area a tree occupies per unit of tree volume. E iv/cpa (cdm year−1 m−2) reflects the growth efficiency of the tree’s crown projection area. The ratios E cpa/sa, E iv/sa, and E v/sa relate the structure and growth of the tree to its required growing space. They thereby enable the transition from tree growth to tree productivity. E cpa/sa (m2 m−2) reflects the degree of crown extension beyond the tree stand area and is equivalent to the degree of crown engagement. E iv/sa (cdm year−1 m−2) represents the efficiency of stand area in terms of tree volume growth (m3 year−1) per tree stand area (m2) and is equivalent to the tree productivity. E v/sa (m3 m−2) indicates the stand density in terms of tree volume per tree growing area (Pretzsch and Schütze 2005). We use the above ratios to compare tree development in mixed versus monospecific stands. The ratio rE iv/csa = E iv/cpa, m /E iv/cpa, p represents how trees perform with regard to crown growth efficiency in mixed versus monospecific stands. Most of the ratios change with tree size and require allometric adjustment before being applied to compare trees in mixed versus monospecific stands when there are differences in tree size, e.g. in tree diameter or tree volume (Dirnberger and Sterba 2014).
6.7.2 Elimination of the Size Effect from Efficiency Parameters
Efficiency ratios such as E iv/cpa or E iv/sa are useful for comparing tree growth in mixed versus monospecific stands. However, these ratios change with tree size. If the size of trees in mixed and monospecific stands differs, such size-dependent changes in the efficiency parameters should be eliminated. Allometric theory (Enquist et al. 1998; West et al. 2009) and empirically derived allometric exponents (Pretzsch and Dieler 2012) are available for such corrections. If these are not undertaken, then mixing effects might be confounded with size effects as shown in the following example.
Supposing that Norway spruce volume increment, iv, scales with an exponent of α iv, v = 0.42 and cpa with α csa, v = 0.62 to total plant volume, then the efficiency of crown area, E iv, csa = iv/csa scales with α iv/cpa, v = − 0.20 to plant volume (as iv ∝ v 0.42 and cpa ∝ v 0.62 E iv, cpa = iv/cpa ∝ v 0.42 − 0.62 ∝ v −0.20) as shown in Fig. 6.28 at (a) linear and (b) double-logarithmic scale. Suppose a tree of volume v m = 1.0 m3 growing in the mixed stand has a crown area efficiency of E iv/cpa, m = 1.25 cdm year−1 m−2 and should be compared with a tree of volume v p = 3.0 m3 and crown area efficiency of E iv/cpa, p = 1.20 cdm year−1 m−2 in the neighbouring monocultures. The relative efficiency of the tree in the mixed stand is then rE iv/cpa = E iv/cpa, m /E iv/cpa, p = 1.25/1.20 = 1.04, which is equivalent to a 4% increase through mixing. In this case it is obvious that the size growth of the tree (or a mean tree) in the mixed stand is behind the monoculture (v m < < v p ), and part of its superiority is related to its younger allometric state. If we apply α iv/cpa, v = − 0.20 and estimate which E ′ iv, cpa, m the tree in the mixed stand would have if it was the same size as the reference tree in the monoculture (v p = 3.0 m3), we get E ′ iv/cpa, m = (iv m /cpa m ) × (v p /v m )−0.20 in the example E ′ iv/cpa, m = (1.25) × (3/1)−0.20 = 1.00 and the relative efficiency calculated on the basis of \( r{E}_{iv/cpa}^{\prime }={E}_{iv/cpa,m}^{\prime }/{E}_{iv/cpa,p}=1.00/1.20=0.83 \). In other words, the real mixing effect is −17% as opposed to +4%; the different sizes create the illusion of a positive mixing effect.
Elimination of size effects from efficiency parameters such as E iv/cpa prior to their application for comparing tree growth in mixed versus monospecific stands, illustrated at (a) linear and (b) logarithmic scale. Allometric correction is recommendable when the compared trees in mixed and monospecific stands differ in size, as represented by their stem volume, v m and v p , respectively. The adjustment starts with the observed E iv/cpa value for the tree with volume v m in mixture (E iv/cpa, m ) and uses the allometric slope α iv/csa, v to extrapolate up to the volume v p (solid arrow from v m to v p ). The read off value is the adjusted efficiency E ′ iv/cpa, m which can be compared with the reference value E iv/cpa, r for the monoculture in order to calculate the real mixing effect rE = E ′ iv/cpa, m /E iv/cpa, p
In such cases, one should either refrain from comparison at the tree level or eliminate the size effects as shown. Based on the uncorrected mixing effect rE iv, cpa = 1.04 and the ‘real’ mixing effect rE ′ iv, cpa = 0.83, we can calculate the size effect S = rE iv, cpa/rE ′ iv, cpa = 1.04/0.83 = 1.25. The observed uncorrected mixing effect rE iv, csa = 0.83 × 1.25 = 1.04 can be split into a real negative mixing effect (rE ′ iv, cpa = 0.83) and a positive size effect (S = 1.25). Part of the size effect might have been caused in the past by mixing but should not be confounded with present mixing effects.
For the allometric adjustment in this chapter, the following exponents for Norway spruce and European beech, αiv/v, v = − 0.58, − 0.41, αcpa/v, v = − 0.38, − 0.31, αiv/cpa, v = − 0.20, − 0.10, and αsa/v, v = − 0.38, − 0.31, were specially derived from trees on long-term experiments. See Pretzsch and Dieler (2012) for empirically derived allometric scaling relationships of other tree species or Enquist et al. (1998, 2009), and West et al. (2009) for general scaling exponents of allometrically ideal plants.
6.7.3 Transition from Tree Growth to Tree Productivity in Mixed Versus Monospecific Stands: An Example
The transition from growth of the individual tree to the productivity of the mean tree and the stand productivity of mixed versus monospecific stands is illustrated for the age series NOR 811, FRE 813, and SON 814 of monospecific and mixed forest stands of Norway spruce and European beech in southern Germany. The age series provide triplets of neighbouring plots which are different in terms of species composition (spruce monoculture, mixed spruce/beech, beech monoculture) and relatively similar in all other characteristics such as site conditions, stand age, silvicultural treatment, etc. The 12 triplets of plots cover 36–161-year-old stands with dominant height of 25–44 m at age 100, standing volume stock of 55–1.041 m3 ha−1, and current annual volume growth of 2–32 m3 ha−1 year−1. The relatively even-aged and monolayered monospecific and mixed stands are fully stocked, unthinned, or only slightly thinned and represent good to optimal site conditions for both spruce and beech. For more details about the plots, see Pretzsch and Schütze (2009) and Pretzsch et al. (2010).
As the considered stands are more or less even aged and the size characteristics are normally distributed, this is the easiest way to explain the transition from the tree level to the stand level. In these stands, the mean tree is a good representative of the respective cohort. In order to demonstrate how mixing can modify the growth and growth efficiency at the individual tree level, we calculated the mean ratios cpa/v, iv/cpa, cpa/sa, iv/sa, and v/sa introduced in Table 6.2 for all plots of the 12 triplets. The mean ratios between the mean tree volume in mixed versus monospecific stands of v m /v r = 0.85 for beech and v m /v r = 1.04 for spruce indicate that beech is 15% smaller and spruce 4% larger in mixed stands versus the neighbouring monoculture of the same triplet. The effects of these size differences were eliminated through allometric adjustment; the adjusted ratios are marked by a stroke (e.g. E ′ mixed).
E cpa/v in Table 6.3 reflects that with a given tree volume, v, trees in mixed stands develop a wider crown than those in monospecific stands. The table shows an increase of 18% for spruce and 45% for beech. Beech is already superior to spruce in terms of crown extension in monospecific stands (26.44/11.83 = 2.2) but even more so in mixed stands (38.33/13.96 = 2.8). On average, both species benefit from mixture in E cpa/v , which indicates that the benefit of one species is not at the expense of the other but rather that their strategies are complementary. The E iv/cpavalues indicate that mixing reduces the crown area efficiency. This reduction amounts to 4% for the rather vertically expanding spruce and 20% for laterally expanding beech. In contrast to E cpa/v , the crown efficiency E iv/cpais about twofold for spruce compared with beech. Mixing increases the crown engagement E cpa/sa in spruce by just 6% (E ′ cpa/sa, m /E cpa/sa, p = 1.06) but in beech by 69% (E ′ cpa/sa, m /E cpa/sa, p = 1.69). In both mixed and monospecific stands of beech, the crown engagement E csa/sa is superior to spruce. Mixing hardly changes the stand area efficiency E iv/sa (= productivity) of spruce (E ′ iv/sa, m /E iv/sa, p = 1.01) but increases that of beech by 15% (E ′ iv/sa, m /E iv/sa, p = 1.15). So, productivity of spruce is generally higher in both monospecific and mixed stands, but beech benefits more from mixing than spruce. Note that especially for beech, efficiency measures based on crown area (E iv/cpa) may indicate a negative mixing effect. However, due to the multiple crown coverage of beech as indicated by its high E cpa/sa values, it can achieve an overyielding in inter- as opposed to intraspecific environments. The wider extension and denser interlocking of beech crowns enables a 13% higher stand density of beech in mixed stands as indicated by E v/sa ratios of E ′ v/sa, m = 0.06 compared with E ′ v/sa, p = 0.05 in monoculture.
6.7.4 Crown Engagement cpa/sa as Link Between Tree and Stand Level Evaluation
For linking tree to stand level evaluation, the overlap and interlocking of crowns is crucial. From E iv/sa = iv/sa, E iv/csa = iv/csa, and E sa/csa = sa/csa, it follows that E iv/sa = E iv/csa × E csa/sa. The latter equation conceptualises individual tree productivity as a product of individual tree crown efficiency and crown engagement. Consider further that the mixing effects in terms of crown efficiency and crown engagement can be expressed as rE iv/csa = E iv/csa, m /E iv/csa, p and rE cpa/sa = E cpa/sa, m /E cpa/sa, p , respectively. The relative productivity of trees in mixed versus monospecific stands is then rE iv/sa = rE iv/cpa × rE cpa/sa, i.e. the relative effect of mixing on productivity is the product of the effect of mixing on crown efficiency and crown engagement.
Figure 6.29 illustrates the efficiency ratios for the 12 triplets in monospecific and mixed stands of Norway spruce and European beech introduced in the previous section (see also Table 6.3). In Fig. 6.29a the mean crown efficiency of beech in mixture is plotted over the efficiency in neighbouring monocultures. Observations close to the 1.0-line would indicate equality of efficiency. Points above or below this bisectoral line indicate a plus or minus, respectively, as compared with monospecific stands. In the case of beech, crown efficiency is reduced through mixing, but crown expansion increased to such an extent that productivity is 10–20% higher in mixed compared with neighbouring monospecific stands (Fig. 6.29a, from left to right). For Norway spruce, the reactions to mixing are similar but less pronounced (Fig. 6.29b, from left to right). When coping with interspecific competition, both species benefit to a different extent from a release of lateral crown restriction. Their wider crown spread and overlap can overcompensate the reduction of crown efficiency and result in a higher tree and stand productivity. Fully stocked mixed stands can differ slightly from monocultures in terms of the ground cover ratio, but the main difference is their higher sum of crown area due to two- to fourfold crown overlaps (see also Sect. 5.7.2). As crown maps are rare and crown areas difficult to measure, the multiple coverage was often neglected and counted only once in analyses (e.g. Assmann 1970, p. 105), but this seems to be crucial for over-/underyielding and should be considered more thoroughly in future.
Ratios for refined analysis of tree growing space efficiency and productivity of (a) European beech and (b) Norway spruce growing in mixed versus monospecific stands. The following efficiency parameters are shown for each species E ′ iv/csa, m vs. E iv/cpa, p (left), E ′ cpa/sa, m vs. E cpa/sa, p (centre), E ′ iv/sa, m vs. E iv/sa, p (right). Small symbols represent means for the included mixed versus monospecific stand plots; large symbols represent mean values over all 12 triplets. In the case that the behaviours of mixed and monospecific stands were equal, the observations would lie on the bisectoral lines
Figure 6.30 illustrates the complementarity of Norway spruce and European beech in terms of space occupation with a section of the mixed stand SON 814/8 (age 80, survey 2004) which is one of the 12 triplets (spruce monoculture, spruce/beech mixed, beech monoculture) used for this example analysis. While spruce tends to expand vertically, beech fills the canopy space through lateral expansion and can increase the stand density.
6.8 Overview of Some Main Causes and Effects of Tree Species Mixing on Individual Tree Growth
Many of the beneficial tree mixing reactions introduced in this chapter result from complementary crown and root shape, spatially or temporally complementary resource exploitation, redistribution of resources, or modification of growth allocation and allometry. This section summarises the main causes and effects of tree species mixing on individual tree growth. There are many more causes of beneficial mixing effects, such as mechanical stabilisation, reduction of crown shyness, mechanical abrasion of parts of the crown by wind and storm, barrier building against biotic and abiotic disturbances, holobiontic interactions via mycorrhizal networks, or common use of root channels for exploitation of deeper soil layers. As the effects of these are so difficult to disentangle, the following overview of cause and effect is still rather phenomenological, qualitative, and mostly restricted to two-species mixtures. Many ongoing research projects strive to generalise and better quantify the effects and to extend the research to mixtures with more than two species.
6.8.1 Complementary Exploitation of Crown and Root Space
Any changes in tree crown morphology and canopy structure in interspecific versus intraspecific environments suggest a modification of the trees’ space occupation, resource capture, and productivity. Of special interest is a wider crown extension as this indicates a competition reduction and an increase in light interception and may also contribute to overyielding of mixed compared with monospecific stands (Chap. 4). Changes in crown structure and allometry underline that the mixing effect goes beyond a merely additive effect where the species’ behaviour in terms of structure and growth remains similar to that in the monoculture (Fig. 6.31a–c).
Potential effects of species mixing on the canopy and root space filling when tree crowns have complementary shapes. When two species with complementary crown shapes (a and b) retain the same shape and space occupation in mixed-species stands—as is common for monospecific stands—the space filling in mixture may be simply additive (c). However, in many cases, species mixing enables a wider crown expansion (d), higher stand density (e), or a combination of both (f). The cases (d)–(f) indicate a multiplicative mixing effect through denser canopy space filling (Pretzsch 2014)
In temperate forests where tree growth is primarily limited by light, canopy space filling may be denser and light interception higher in mixed than in monospecific stands. Especially when their crown shapes are complementary, the crown expansion may be wider (Fig. 6.31d), the tree packing density higher (Fig. 6.31e), or both reactions may be combined (Fig. 6.31f). Similar complementarity with regard to their root morphology may increase the benefit of inter- compared with intraspecific neighbourhoods.
Because of the complementarity in crown shape (e.g. a combination of ▼-shaped with ▲-shaped crowns) and resource demand (e.g. combination of light-demanding and shade-tolerant species), tree crowns may expand more widely in inter- compared with intraspecific environments. On top of that, mixing can change inner crown properties such as branch length, branch angle, number of branches, and their straightness. The cpa/sa relationship can become greater in mixed stands (Fig. 6.32b) and the degree of crown closure—and therefore the crown closure and the sum of crown projection areas—may be higher than in monocultures (Fig. 6.32a). Larger crowns and denser canopy space filling can increase the tree and stand growth.
Schematic representation of canopy pattern in monospecific versus mixed stands. (a) Rather circular crowns in monospecific stand with about 5–10% uncovered space in between, a crown cover of 90–95%, and sum of crown areas below 100%. (b) More irregular crown shapes in mixed stand with scarce uncovered space in between, crown cover close to 100%, and multiple crown overlap resulting in a sum of crown areas of greater than 100%
Analogously, a complementary root morphology may benefit inter- compared with intraspecific neighbourhoods in the root space.
6.8.2 Hydraulic Lift and Hydraulic Redistribution
Especially in dry years and on dry sites where water is the limiting factor, processes in the root space become decisive for tree growth and survival. However, outcomes under drought conditions may turn into benefits for some of the associates. One such phenomenon is the well-described ‘hydraulic lift’ or ‘hydraulic redistribution’ of water in woody plants, particularly observable in arid conditions, through deeply extending root systems (Fig. 6.33a), which release water at night into surface-near soil horizons of low water potential (Fig. 6.33b) as described in detail by Caldwell and Richards (1989) and Prieto et al. (2012). Once released, such water represents an external storage capacitance for the deep-rooting plant, but concomitantly enables survival of neighbouring shallow-rooting plants as the profiteers. The resulting benefit is not an active support provided by the deep-rooting plant, but occurs indirectly as an emergent feature of the specific species association under a specific site scenario. In such a case, competition turns into facilitation, which denotes an indirectly mediated, functional emergence between neighbouring plants. One or several of the associates may benefit with regards to water availability and growth.
If shallow-rooting species are mixed with deep-rooting species, they can benefit from hydraulic redistribution. (a) During the day when stomata are opened, both species take up water for transpiration. (b) Dry soil conditions may extract water from the deep-reaching roots, especially during the night when water flow is reduced. Shallower rooting trees may benefit from this redistribution in terms of water supply and growth (Caldwell and Richards 1989)
6.8.3 Increased Availability of Mineral Nutrient Supply Through Deep-Rooting or Atmospheric N2 Fixation
In mixed stands one tree species may benefit from another species which can exploit mineral nutrients in deeper root layers (Fig. 6.34a) or can fix atmospheric N2 (Fig. 6.34a). In both cases, mineral nutrients are procured by a benefactor species and partly transferred to the other through litter fall. Thus, the additional nutrients are socialised among the neighbouring trees of the benefactor independently of their species. An additional benefit may come from root channels abandoned by one species and used by the other for penetrating compacted soil layers and reaching resources in deeper and otherwise inaccessible soil layers (Bauhus and Messier 1999; Gaiser 1952; Puhe 2003; Stone and Kalisz 1991).
In assemblages of tree species with different behaviours of mineral nutrient exploitation, one species may facilitate the other through socialisation of nutrient supply via leaf and needle turnover. (a) Shallower rooting species may benefit from deep-rooting neighbours because they act as a ‘mineral nutrient pump’ (Rothe 1997; Rothe and Binkley 2001). (b) Species with the potential for atmospheric N2 fixation may facilitate neighbouring species that lack this ability, especially when growing on N-limited sites (Forrester et al. 2006, 2007)
6.8.4 Temporal Complementarity of Niches, Resource Use, and Growth
Many studies show clear differences in the course of growth between the species across their lifetimes (Assmann 1970, p 45) but also within a single year (Pretzsch 2005a; Rötzer et al. under review; Schober 1950/51). This asynchrony in growth entails a temporal diversification in resource uptake which can release stress.
For instance, Norway spruce may profit from a released competition for water in early spring when European beech is still leafless and lives from the reserves of the previous year (Fig. 6.35). In this period, photosynthesis and growth of spruce may profit from higher temperatures and light supply till beech sprouts in April/May. Afterwards, European beech may profit from the mixture in relation to the monospecific stand as the admixed Norway spruces have shallower roots. During the main growing season, scattered European beeches in the mixture with Norway spruce may have greater water availability than in monocultures of beech as the interspecific competition with neighbouring spruces is lower than the intraspecific competition with beech (Rötzer et al. under review). This may explain why beeches in interspecific environments grow much better than those beside beeches, especially in drought years (Goisser et al. 2016; Rötzer et al. under review).
Temporal complementarity of resource use and growth when mixing evergreen species (a) with deciduous tree species (c). In mixtures of both species (b), conifers can start growing before the sprouting of their deciduous neighbours. This asynchrony may promote smaller conifers in particular and enrich stand structure and stand productivity (Schober 1950/51)
Analogously, temporal diversification of space occupation during stand development may relax resource competition. It occurs, for example, in species assemblages of shade-tolerant and shade-intolerant, shallow- and deep-rooting, and early- and late-successional tree species. For given site conditions, species 1 and 2 in Fig. 6.36 may access resources in different spaces or at different times.
Complementary occupation and packing density in the canopy and root space found in one stand development phase (a) may change over time (b). In the mixture shown on the right (b), conifer is initially ahead but later lags behind in height growth. However, due to the species-specific crown development, complementary space occupation may result in all stand development phases
6.8.5 Modification of Growth Partitioning and Allometry of Trees in Inter- Versus Intraspecific Neighbourhoods
In order to acclimatise to an interspecific neighbourhood, trees species may change their growth partitioning in mixed-species stands compared with monospecific stands. Most of the comparisons between the productivity of mixed and pure stands are based on stem volume production (Liang et al. 2016; Pretzsch et al. 2015). Comparison based on total biomass production may produce different results, as tree species mixing can change stem-crown allometry (Bayer et al. 2013), root-shoot relationship (Thurm et al. 2016), and also tree-ring width and wood density (Zeller 2016) as illustrated in Fig. 6.37.
Tree species mixing may modify the growth allocation and allometry in mixed (b) compared with monospecific stands (a and c). Mixing may modify the crown size, the root-shoot relationship, the tree-ring width, and wood density. So, comparison between mixed and monospecific stands based on the stem volume growth may be misleading (Zeller 2016)
An increase in crown in relation to stem size in mixed-species stands as reported by Dieler and Pretzsch (2013) and Pretzsch (2014) would mean that the overyielding would be even higher when calculated for the total above-ground volume of mixed versus pure stands. However, the decrease of root in relation to shoot growth as reported by Thurm et al. (2016) and reported in Sect. 6.5.3 as well as the decrease of wood density revealed by Zeller (2016) would lower the overyielding of mixed versus pure stands when calculated for the total biomass growth. Ongoing works on within-tree growth partitioning in mixed versus pure stands will clarify how mixed stand productivity compares with that in pure stands when calculated for the stem volume productivity, which is of primary interest for forestry. They will also clarify how mixed stand productivity compares with pure stands based on their total biomass production, which is relevant for ecosystem understanding and carbon balance.
The presented tree properties emerging when growing in interspecific in contrast to intraspecific neighbourhoods go beyond an acceleration or reduction of growth velocity, i.e. a quicker tree and stand development. Rather, they can change the trees’ structure and space requirement. The presented tree properties emerging in interspecific in contrast to intraspecific competition indicate complementarity in use of above- and below-ground resources.
Beyond the increased productivity, the wider extension of tree crowns and multiple crown overlap in mixture can increase the growth resilience after disturbances, as gaps are smaller or can be closed more quickly. Properties emerging in mixed but not in monospecific stands such as increase in crown size, change in the crowns’ space filling pattern, reduction in growing area requirement, and increase in tree productivity are crucial for appropriate estimation of maximum density, silvicultural treatment, and dynamic modelling of mixed-species stands. Because of their size, firm position, and longevity, trees are the founder species in ecosystems and determine the living conditions of many ecosystem characteristics, functions, and services. So, the effects of mixing on tree and furthermore on stand structure have far-reaching effects on most ecosystem properties.
6. Take-Home Messages
-
1.
Interspecific difference in tree size causes well-known additive mixing effects such as higher or lower tree numbers per unit area in mixed versus monospecific stands due to different growing space requirements. Interspecific functional and structural complementarity (light-demanding vs. shade-tolerant, early- vs. late-successional, shallow vs. deep-rooting, deciduous vs. evergreen species) may cause multiplicative mixing effects such as multiple crown layering, denser canopy space filling, and eventually overyielding.
-
2.
The feedback between environment, growth, and structure enables a structural acclimation of trees to their local environment. Their size and longevity improve the capacity to increasingly acclimate morphologically with progressing stand development. Mixing can foster the variability of both the local environment and the tree structure and growth.
-
3.
Species mixing can trigger emergent properties of individual tree structure and growth which may result in an increase in resource capture and overyielding of mixed compared with monospecific stands.
-
4.
Depending on the net effect of competition and facilitation by neighbours, the unimodal increment curve of individual trees can be modified in different ways; e.g. its level can be raised or lowered and its rhythm can be accelerated or slowed down.
-
5.
Due to the complementarity of species and a reduction of competition, crowns may expand more widely in length and width in interspecific compared with intraspecific environments.
-
6.
Mixing can generate allometric relationships, crown projection areas, and inner crown properties which differ significantly from respective properties in monospecific stands.
-
7.
Wider crown extension and multiple crown overlap in mixed stands may indicate a more complete canopy space filling and light capture. Wider reach of the crowns combined with lower growing space requirements can increase the tree productivity in mixed versus monospecific stands by 10–20%.
-
8.
By relating the achieved tree growth to growing area instead of plant or crown size, we proceed from plant biology to production ecology.
-
9.
Tree characteristics change non-linearly with tree size. Therefore, comparison of structure or growth of trees in mixed versus monospecific stands should consider and eliminate any size differences between trees in mixed and monospecific stands.
-
10.
The beneficial tree mixing reactions introduced in this chapter result mainly from complementary crown and root shape, spatially or temporally complementary resource exploitation, redistribution of resources, or modification of growth allocation and allometry.
References
Alemdag IS (1978) Evaluation of some competition indexes for the prediction of diameter increment in planted white spruce. Can For Serv, Ottawa, Canada
Arz MAO (2013) Strukturelle Kronenanalyse von Fichte (Picea abies [L.] Karst.) und Buche (Fagus sylvativa L.) im Rein- und Mischbestand. Kombination von terrestrischen Laserscan- und Zuwachsdaten
Assmann E (1970) The principles of forest yield study. Pergamon Press, Oxford, New York
Assmann E, Franz F (1965) Vorläufige Fichten-Ertragstafel für Bayern. Forstwissenschaftliches Centralblatt 84, 1/2, Sonderdruck, Verlag Paul Parey, Hamburg/Berlin
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann For Sci 59:233–253
Bauhus J (2009) Rooting patterns of old-growth forests: is aboveground structural and functional diversity mirrored belowground? In: Cea W (ed) Old-growth forests, ecological studies, vol 207. Springer, Berlin, pp 211–229. doi:10.1007/978-3-540-92706-8_10
Bauhus J, Messier C (1999) Soil exploitation strategies of fine roots in different tree species of the southern boreal forest of eastern Canada. Can J For Res 29(2):260–273
Bayer D, Seifert S, Pretzsch H (2013) Structural crown properties of Norway spruce and European beech in mixed versus pure stands revealed by terrestrial laser scanning. Trees 27(4):1035–1047
Belsky AJ, Canham CD (1994) Forest gaps and isolated savanna trees. Bioscience 44(2):77–84
Binkley D, Stape JL, Ryan MG (2004) Thinking about efficiency of resource use in forests. For Ecol Manag 193:5–16
Brooker RW, Maestre FT, Callaway RM, Lortie CL, Cavieres LA, Kunstler G, Michalet R (2008) Facilitation in plant communities: the past, the present, and the future. J Ecol 96(1):18–34
Brown GS (1965) Point density in stems per acre, vol 38. New Zealand Forest Research Note, Wellington, New Zealand
Caldwell MM, Richards JH (1989) Hydraulic lift: water efflux from upper roots improves effectiveness of water uptake by deep roots. Oecologia 79(1):1–5
Callaway RM, Walker LR (1997) Competition and facilitation: a synthetic approach to interactions in plant communities. Ecology 78(7):1958–1965
Canham CD, Finzi AC, Pacala SW, Burbank DH (1994) Causes and consequences of resource heterogeneity in forests: interspecific variation in light transmission by canopy trees. Can J For Res 24(2):337–349
Canham CD, LePage PT, Coates KD (2004) A neighborhood analysis of canopy tree competition: effects of shading versus crowding. Can J For Res 34(4):778–787
Christmann (1939) Ertragstafel für den Kiefern-Fichten-Mischbestand. In: Wiedemann E (ed) Ertragstafeln wichtiger Holzarten bei verschiedener Durchforstung sowie einiger Mischbestandsformen. Schaper Verlag, Hannover, p 100
Comeau PG, Kimmins JP (1989) Above- and below-ground biomass and production of Lodgepole pine on sites with differing soil moisture regimes. Can J For Res 19:447–454
Connell JH (1990) Apparent versus “real” competition in plants. In: Grace JB, Tilman D (eds) Perspectives on plant competition. Academic Press, San Diego, pp 9–26
Dieler J, Pretzsch H (2013) Morphological plasticity of European beech (Fagus sylvatica L.) in pure and mixed-species stands. For Ecol Manag 295:97–108
Dirnberger GF, Sterba H (2014) A comparison of different methods to estimate species proportions by area in mixed stands. For Syst 23(3):534–546
Enquist BJ, Brown JH, West GB (1998) Allometric scaling of plant energetics and population density. Nature 395:163–165
Enquist BJ, West GB, Brown JH (2009) Extension and evaluations of a general quantitative theory of forest structure and dynamics. Proc Natl Acad Sci U S A 106(17):7046–7051
Faber PJ (1981) Die Standflächenschätzung über den Distanzfaktor. In: Proc Dt Verb Forstl Forschungsanst. Sek Ertragskd, Soest, pp 87–95
Faber PJ (1983) Concurrentie en groei van de bomen binnen een opstand (Konkurrenz und Wachstum der Bäume in einem Waldbestand). Pijksinstituut voor onderzoek in de bos- en landschapsbouw “De Dorschkamp”, vol 18(1). Uitvoerig verslag, Wageningen
Forrester DI (2013) The spatial and temporal dynamics of species interactions in mixed-species forests: from pattern to process. For Ecol Manag. doi:10.1016/j.foreco.2013.10.003
Forrester DI (2014) A stand-level light interception model for horizontally and vertically heterogeneous canopies. Ecol Model 276:14–22
Forrester DI, Bauhus J, Cowie AL, Vanclay JK (2006) Mixed-species plantations of Eucalyptus with nitrogen-fixing trees: a review. For Ecol Manag 233:211–230
Forrester DI, Bauhus J, Cowie AL, Mitchell PA, Brockwell J (2007) Productivity of three young mixed-species plantations containing N2-fixing Acacia and non-N2-fixing Eucalyptus and Pinus trees in Southeastern Australia. For Sci 53(3):426–434
Fraser AR (1977) Triangle based probability polygons for forest sampling. For Sci 23(1):111–121
Gaiser RN (1952) Root channels and roots in forest soils. Soil Sci Soc Am J 16(1):62
Goisser M, Geppert U, Rötzer T, Paya A, Huber A, Kerner R, Bauerle T, Pretzsch H, Pritsch K, Häberle K-H, Matyssek R, Grams TEE (2016) Does belowground interaction with Fagus sylvatica increase drought susceptibility of photosynthesis and stem growth in Picea abies? For Ecol Manag 375:268–278
Griess VC, Knoke T (2011) Growth performance, windthrow, and insects: meta-analyses of parameters influencing performance of mixed-species stands in boreal and northern temperate biomes. Can J For Res 41:1141–1158
Gspaltl M, Sterba H, O’Hara KL (2012) The relationship between available area efficiency and area exploitation index in an even-aged coast redwood (Sequoia sempervirens) stand. Forestry 85(5):567–577
Hari P (1985) Theoretical aspects of eco-physiological research. In: Tigerstedt PMA, Puttonen P, Koski V (eds) Crop physiology of forest trees. Helsinki University Press, Helsinki, pp 21–30. (336 p)
Heinsdorf D (1999) Das Revier Sauen – Ein Beispiel für erfolgreichen Waldumbau, Schriftenreihe des Ministeriums für Ernährung, Landwirtschaft und Forsten. vol Band VI. Ministerium für Ernährung, Landwirtschaft und Forsten, Brandenburg, Landesforstanstalt Eberswalde
Helms JA (1998) The dictionary of forestry. The Society of American Foresters, Bethesda, MD
Jack WH (1968) Single trees sampling in evenaged plantations for survey and experimentation. In: 14th IUFRO Congress, München, pp 379–403
Kelty MJ (1992) Comparative productivity of monocultures and mixed stands. In: Kelty MJ, Larson BC, Oliver CD (eds) The ecology and silviculture of mixed-species forests. Kluwer Academic, Dordrecht, pp 125–141
Keyes MR, Grier CC (1981) Above-and below-ground net production in 40-years-old Douglas-fir stands on low and high productivity sites. Can J For Res 11:599–605
Kimmins JP (1993) Scientific foundations for the simulation of ecosystem function and management in FORCYTE-11. Forestry Canada, Northern Forestry Centre, Edmonton, Alberta
Kleiber M (1947) Body size and metabolic rate. Physiol Rev 27(4):511–541
Knoke T, Hahn A (2007) Baumartenvielfalt und Produktionsrisiken: Ein Forschungseinblick und-ausblick – Diversity of tree species and risk: A research insight and outlook. Schweizerische Zeitschrift fur Forstwesen 158(10):312–322
Kuoch R (1972) Zur Struktur und Behandlung von subalpinen Fichtenwäldern. Schweiz Z Forstwes 123:77–89
Larocque GR, Luckai N, Adhikary SN, Groot A, Bell FW, Sharma M (2013) Competition theory-science and application in mixed forest stands: review of experimental and medelling methods and suggestions for future research. Environ Rev 21:71–84
Liang J, Crowther TW, Picard N, Wiser S, Zhou M, Alberti G, Schulze E-D, McGuire AD, Bozzato F, Pretzsch H, de-Miguel S, Paquette A, Hérault B, Scherer-Lorenzen M, Barrett CB, Glick HB, Hengeveld GM, Nabuurs G-J, Pfautsch S, Viana H, Vibrans AC, Ammer C, Schall P, Verbyla D, Tchebakova N, Fischer M, Watson JV, HYH C, Lei X, Schelhaas M-J, Lu H, Gianelle D, Parfenova EI, Salas C, Lee E, Lee B, Kim HS, Bruelheide H, Coomes DA, Piotto D, Sunderland T, Schmid B, Gourlet-Fleury S, Sonké B, Tavani R, Zhu J, Brandl S, Vayreda J, Kitahara F, Searle EB, Neldner VJ, Ngugi MR, Baraloto C, Frizzera L, Bałazy R, Oleksyn J, Zawiła-Niedźwiecki T, Bouriaud O, Bussotti F, Finér L, Jaroszewicz B, Jucker T, Valladares F, Jagodzinski AM, Peri PL, Gonmadje C, Marthy W, O’Brien T, Martin EH, Marshall AR, Rovero F, Bitariho R, Niklaus PA, Alvarez-Loayza P, Chamuya N, Valencia R, Mortier F, Wortel V, Engone-Obiang NL, Ferreira LV, Odeke DE, Vasquez RM, Lewis SL, Reich PB (2016) Positive biodiversity-productivity relationship predominant in global forests. Science 354(6309). doi:10.1126/science.aaf8957
Matyssek R, Lüttge U (2012) Gaia. The planet holobiont. Nova Acta Leopoldina NF 114(391):325–344
Mayer H, Ott E (1991) Gebirgswaldbau Schutzwaldpflege. Gustav Fischer Verlag, Stuttgart, New York
McCarthy MC, Enquist BJ (2007) Consistency between an allometric approach and optimal partitioning theory in global patterns of plant biomass allocation. Funct Ecol 21:713–720
Møller AP, Swaddle JP (1997) Asymmetry, developmental stability, and evolution. Oxford Serie in ecology and evolution. Oxford University Press, Oxford
Nagel J (1985) Wachstumsmodell für Bergahorn in Schleswig-Holstein. Universität Göttingen, 124p
Nelder JA (1962) New kinds of systematic designs for spacing experiments. Biometrics 18(3):283–307
Niklas KJ (1994) Plant allometry. Univ Chicago Press, Chicago, IL
Niklas KJ (2004) Plant allometry: is there a grand unifying theory? Biol Rev 79:871–889
Nikolova PS, Zang C, Pretzsch H (2011) Combining tree-ring analyses on stems and coarse roots to study the growth dynamics of forest trees: a case study on Norway spruce (Picea abies [L.] H. Karst). Trees 5:859–872
Oldemann RAA (1990) Forests: elements of silvology. Springer, Berlin
Oliver CD, Larson BC (1996) Forest stand dynamics: Update edition. Wiley, New York
Pelz DR (1978) Estimating individual tree growth with tree polygons. Blacksburg, VA
Pretzsch H (2005a) Diversity and productivity in forests. In: Scherer-Lorenzen M, Körner C, Schulze E-D (eds) Forest diversity and function, Ecological studies 176. Springer, Berlin, pp 41–64
Pretzsch H (2005b) Stand density and growth of Norway spruce (Picea abies (L.) Karst.) and European beech (Fagus sylvatica L.). Evidence from long-term experimental plots. Eur J For Res 124(3):193–205
Pretzsch H (2006) Von der Standfächeneffizienz der Bäume zur Dichte-Zuwachs-Beziehung des Bestandes. Beitrag zur Integration von Baum- und Bestandesebene. Allgemeine Forst- und Jagdzeitung 177:188–199
Pretzsch H (2009) Forest dynamics, growth and yield. Springer, Berlin
Pretzsch H (2014) Canopy space filling and tree crown morphology in mixed-species stands compared with monocultures. For Ecol Manag 327:251–264
Pretzsch H, Biber P (2005) A re-evaluation of Reinekes rule and stand density index. For Sci 51(4):304–320
Pretzsch H, Biber P (2010) Size-symmetric versus size-asymmetric competition and growth partitioning among trees in forest stands along an ecological gradient in central Europe. Can J For Res 40:370–384
Pretzsch H, Dieler J (2012) Evidence of variant intra- and interspecific scaling of tree crown structure and relevance for allometric theory. Oecologia 169(3):637–649. doi:10.1007/s00442-011-2240-5
Pretzsch H, Schütze G (2005) Crown allometry and growing space efficiency of Norway Spruce (Picea abies [L.] Karst) and European Beech (Fagus sylvatica [L.]) in pure and mixed stands. Plant Biol 7(6):628–640
Pretzsch H, Schütze G (2009) Transgressive overyielding in mixed compared with pure stands of Norway spruce and European beech in Central Europe: evidence on stand level and explanation on individual tree level. Eur J For Res 128:183–204
Pretzsch H, Block J, Dieler J, Dong PH, Kohnle U, Nagel J, Spellmann H, Zingg A (2010) Comparison between the productivity of pure and mixed stands of Norway spruce and European beech along an ecological gradient. Ann For Sci 67:712
Pretzsch H, Biber P, Uhl E (2012a) Coarse root-shoot allometry of Pinus radiata modified by site conditions in the Western Cape province of South Africa. Southern Forests 74(4):237–246
Pretzsch H, Schütze G, Uhl E (2012b) Resistance of European tree species to drought stress in mixed versus pure forests: evidence of stress release by inter-specific facilitation. Plant Biol 15:483–495
Pretzsch H, Heym M, Pinna S, Schneider R (2014) Effect of variable retention cutting on the relationship between growth of coarse roots and stem of black spruce (Picea mariana (Mill.) Britton). Scand J For Res 29(3):222–233
Pretzsch H, Forrester DI, Rötzer T (2015) Representation of species mixing in forest growth models. A review and perspective. Ecol Model 313:276–292
Prieto I, Armas C, Pugnaire FI (2012) Water release through plant roots: new insights into its consequences at the plant and ecosystem level. New Phytol 193(4):830–841
Puhe J (2003) Growth and development of the root system of Norway spruce (Picea abies) in forest stands—a review. For Ecol Manag 175(1):253–273
Purves DW, Lichstein JW, Pacala SW (2007) Crown plasticity and competition for canopy space: a new spatially implicit model parameterized for 250 North American tree species. PLoS One 2(e870). doi:10.1371/journal.pone.0000870
Reineke LH (1933) Perfecting a stand-density index for even-aged forest. J Agric Res 46:627–638
Richards AE, Forrester DI, Bauhus J, Scherer-Lorenzen M (2010) The influence of mixed tree plantations on the nutrition of individual species: a review. Tree Physiol 30(9):1192–1208
Roloff A (2001) Baumkronen. Verständnis und praktische Bedeutung eines komplexen Naturphänomens. Ulmer, Stuttgart
Rothe A (1997) Einfluß des Baumartenanteils auf Durchwurzelung, Wasserhaushalt, Stoffhaushalt und Zuwachsleistung eines Fichten-Buchen-Mischbestandes am Standort Höglwald. Forstl Forschungsber München 163:174
Rothe A, Binkley D (2001) Nutritional interactions in mixed species forests: a synthesis. Can J For Res 31:1855–1870
Rubner M (1931) Die Gesetze des Energieverbrauchs bei der Ernährung, vol 16/18. Proc preuß Akad Wiss Physik-Math Kl, Berlin, Wien
Ryan MG, Waring RH (1992) Maintenance respiration and stand development in a subalpine lodgepole pine forest. Ecology 73(6):2100–2108
Ryan MG, Yoder BJ (1997) Hydraulic limits to tree height and tree growth. Bioscience 47(4):235–242
Saha S, Kuehne C, Kohnle U, Brang P, Ehring A, Geisel J, Leder B, Muth M, Petersen R, Peter J, Ruhm W, Bauhus J (2012) Growth and quality of young oaks (Quercus robur and Quercus petraea) grown in cluster plantings in central Europe: a weighted meta-analysis. For Ecol Manag 283:106–118
Schober R (1950/51) Zum jahreszeitlichen Ablauf des sekundären Dickenwachstums. Allgemeine Forst- und Jagdzeitung 122:81–96
Schütz JP (1989) Zum Problem der Konkurrenz in Mischbeständen. Schweiz Z Forstwes 140(12):1069–1083
Smith TM, Smith RL (2009) Elements of ecology. Pearson International Edition, 7th edn. Benjamin Cummings, San Francisco
Sterba H, Amateis RL (1998) Crown efficiency in a loblolly pine (Pinus taeda) spacing experiment. Can J For Res 28(9):1344–1351
Sterba H, Andrae F, Pambudhi F (1993) Crown efficiency of oak standards as affected by mistletoe and coppice removal. For Ecol Manag 62(1):39–49
Stone EL, Kalisz PJ (1991) On the maximum extent of tree roots. For Ecol Manag 46(1–2):59–102
Strobel GW (1995) Rottenstruktur und Konkurrenz im subalpinen Fichtenwald
Thurm EA, Biber P, Pretzsch H (2016) Tree growth is partitioned in favor of the stem on the expense of the roots for Douglas-fir (Pseudotsuga menziesii (Mirb.) Franco) and European beech (Fagus sylvatica L.) if growing in mixture and under humid conditions. Trees. doi:10.1007/s00468-016-1512-4
Uhl E, Biber P, Ulbricht M, Heym M, Horváth T, Lakatos F, Gál J, Steinacker L, Tonon G, Ventura M, Pretzsch H (2015) Analysing the effect of stand density and site conditions on structure and growth of oak species using Nelder trials along an environmental gradient: experimental design, evaluation methods, and results. For Ecosyst 2(1):17
Vandermeer J (1992) The ecology of intercropping. Cambridge University Press, Cambridge
Verein Deutscher Forstlicher Versuchsanstalten (1873) Anleitung für Durchforstungsversuche. In: von Ganghofer A (ed) Das Forstliche Versuchswesen, vol 2. Schmid’sche Buchhandlung, Augsburg, pp 247–253
Verein Deutscher Forstlicher Versuchsanstalten (1902) Beratungen der vom Vereine Deutscher Forstlicher Versuchsanstalten eingesetzten Kommission zur Feststellung des neuen Arbeitsplanes für Durchforstungs- und Lichtungsversuche. Allgemeine Forst- und Jagdzeitung 78:180–184
von Bertalanffy L (1951) Theoretische Biologie: II. Band, Stoffwechsel, Wachstum, 2nd edn. A Francke AG, Bern
von Lüpke B, Spellmann H (1997) Aspekte der Stabilität und des Wachstums von Mischbeständen aus Fichte und Buche als Grundlage für waldbauliche Entscheidungen. Forstarchiv 68:167–179
von Lüpke B, Spellmann H (1999) Aspects of stability, growth and natural regeneration in mixed Norway spruce-beech stands as a basis of silvicultural decisions. In: Olsthoorn AFM, Bartelink HH, Gardiner JJ, Pretzsch H, Hekhuis HJ, Franc A (eds) Management of mixed-species forest: silviculture and economics, vol 15. IBN Scientific Contributions, pp 245–267
West GB, Enquist BJ, Brown JH (2009) A general quantitative theory of forest structure and dynamics. Proc Natl Acad Sci U S A 106(17):7040–7045
Wiedemann E (1942) Der gleichaltrige Fichten-Buchen-Mischbestand. Mitt Forstwirtsch u Forstwiss 13:1–88
Wiedemann E (1951) Ertragskundliche und waldbauliche Grundlagen der Forstwirtschaft. Frankfurt am Main
Yoder BJ, Ryan MG, Waring RH, Schoettle AW, Kaufmann MR (1994) Evidence of reduced photosynthetic rates in old trees. For Sci 40(3):513–527
Zeide B (1985) Tolerance and self-tolerance of trees. For Ecol Manag 13:149–166
Zeide B (1998) Fractal analysis of foliage distribution in loblolly pine crowns. Can J For Res 28:106–114
Zeller L (2016) Tree ring width and wood density in mixed versus pure stands of Scots pine and European beech. TUM, 39 p
Zilber-Rosenberg I, Rosenberg E (2008) Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol Rev 32(5):723–735
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer-Verlag GmbH Germany
About this chapter
Cite this chapter
Pretzsch, H. (2017). Individual Tree Structure and Growth in Mixed Compared with Monospecific Stands. In: Pretzsch, H., Forrester, D., Bauhus, J. (eds) Mixed-Species Forests. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-54553-9_6
Download citation
DOI: https://doi.org/10.1007/978-3-662-54553-9_6
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-54551-5
Online ISBN: 978-3-662-54553-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)