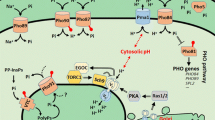
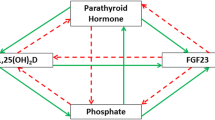
Abstract
Phosphorus is an essential constituent of all living cells. This element is part of fundamental building blocks that constitute nucleic acids, phospholipids and complex carbohydrates. Phosphorus compounds play a central role in anabolic and catabolic pathways and in the energy conversion of the cell via transfer of energy-rich phosphoanhydride bonds, a reaction which is also involved in posttranslational regulation of enzyme activities.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Aiba H, Mizuno T (1994) A novel gene whose expression is regulated by the response- regulator, SphR, in response to phosphate limitation in Synechococcus species PCC7942. Mol Microbiol 13: 25–34
Aiba H, Nagaya M, Mizuno T (1993) Sensor and regulator proteins from the cyanobacterium Synechococcus species PCC7942 that belong to the bacterial signal-transduction protein families: implication in the adaptive response to phosphate limitation. Mol Microbiol 8: 81–91
Allen MM (1968) Photosynthetic membrane system in Anacystis nidulans. J Bacteriol 96: 836–841
Amemura M, Makino K, Shinagawa H, Kobayashi A, Nakata A (1985) Nucleotide sequence of the genes involved in phosphate transport and regulation of the phosphate regulon in Escherichia coli. J Mol Biol 184: 241–259
Ames GF (1986) Bacterial periplasmic transport systems: structure, mechanism, and evolution. Annu Rev Biochem 55: 397–425
Armstrong RA, McGhee R (1980) Competitive exclusion. Am Nat 115: 151–170
Aubriot L, Wagner F, Falkner G (2000) The phosphate uptake behaviour of phytoplankton communities in eutrophic lakes reflects alterations in the phosphate supply. Eur J Phycol 35: 255–262
Ault-Riché D, Fraley CD, Tzeng CM, Kornberg A (1998) Novel assay reveals multiple pathways regulating stress-induced accumulations of inorganic polyphosphate in Escherichia coli. J Bacteriol 180: 1841–1847
Bennett RL, Malamy MH (1970) Arsenate resistant mutants of Escherichia coli and phosphate transport. Biochem Biophys Res Commun 40: 496–503
Bieleski RL, Ferguson IB (1983) Physiology and metabolism of phosphate and its compounds. In: Pirson A, Zimmermann MH (eds) Encyclopedia of plant physiology vol 15A. Laeuchli A. Bieleski RL (eds) Inorganic plant nutrition. Springer, Berlin Heidelberg New York dd 422–449
Boos W, Lucht JM (1996) Periplasmic protein-dependent ABC transporters. In: Neidhardt FC (ed) Escherichia coli and Salmonella. ASM Press, Washington, pp 1175–1209
Borchardt MA, Hoffmann JP, Cook PW (1994) Phosphorus uptake kinetics of Spirogyra fluviatilis ( Charophyceae) in flowing water. J Phycol 30: 403–417
Bornefeld T, Weis U (1981) Adenylate energy charge and phosphorylation potential in blue-green bacterium Anacystis nidulans. Biochem Physiol Pflanz 176: 71–82
Brass JM, Higgins CF, Foley M, Rugman PA, Birmingham J, Garland PB (1986) Lateral diffusion of proteins in the periplasm of Escherichia coli. J Bacteriol 165: 787–794
Bray D (1995) Protein molecules as computational elements in living cells. Nature 376:307–312. Errata: (1995) Nature 378: 419
Brown EJ, Button DK (1979) Phosphate-limited growth kinetics of Selenastrum capricornutum ( Chlorophyceae ). J Phycol 15: 305–311
Brzoska P, Rimmele M, Brzostek K, Boos W (1994) The Ugp paradox: the phenomenon that glycerol-3-phosphate, exclusively transported by the Escherichia coli Ugp system, can serve as a sole source of phosphate but not as a sole source of carbon is due to trans inhibition of Ugp-mediated transport by phosphate. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 30–36
Button DK (1978) On the theory of control of microbial growth kinetics by limiting nutrient concentrations Deep Sea Res 25: 1163–1177
Caperon J (1968) Population growth response of Isochrysis galbana to variable nitrate environment. Ecology 49: 866–872
Coleman JE (1992) Structure and mechanism of alkaline phosphatase. Annu Rev Biophys Chem 21: 441–483
Currie DJ, Kalff J (1984) The relative importance of bacterioplankton and phytoplankton in phosphorus uptake in freshwater. Limnol Oceanogr 29: 311–321
De Cock H, Overeem W, Tommassen J (1992) Biogenesis of outer membrane protein PhoE of Escherichia coli. J Mol Biol 224: 369–379
Droop MR (1968) Vitamin B12 and marine ecology. IV: The kinetics of uptake, growth and inhibition in Monochrysis lutheri. J Mar Biol Assoc UK 48: 689–733
Droop MR (1973) Some thoughts on nutrient limitation in algae. J Phycol 9: 264–272
Droop MR (1974) The nutrient status of algal cells in continuous culture. J Mar Biol Assoc UK 54: 825–855
Ducobu H, Huisman J, Jonker RR, Mur LR (1998) Competition between a prochlorophyte and a cyanobacterium under various phosphorus regimes: comparison with the Droop model. J Phycol 34: 467–476
Egge JK (1998) Are diatoms poor competitors at low phosphate concentrations? J Mar Syst 16: 191–198
Einsele W (1941) Die Umsetzung von zugefuehrtem, anorganischem Phosphat im eutrophen See und ihre Rückwirkungen auf seinen Gesamthaushalt. Z Fisch Hilfswissensch 39: 407–488
Falkner G (1984) Die Phosphataufnahme bei Algen unter besonderer Berücksichtigung der Cyanophyceen. Sitzungsber Oesterr Akad Wissensch 193: 33–88
Falkner G, Werdan H, Horner F, Heidt HW (1974) Energieabhaengige Phosphataufnahme der Blaualge Anacystis nidulans. Ber Dtsch Bot Ges 87: 263–266
Falkner G, Strasser P, Graffius D (1984a) Phosphate uptake by blue-green algae in vitro and in a lake during an algal bloom: useful application of a force-flow relationship. Hydrobiologia 108: 265–271
Falkner G, Strasser P, Graffius D (1984b) Phosphate uptake by blue-green algae during an algal bloom. Verh Int Ver Limnol 22: 195–199
Falkner G, Falkner R, Schwab AJ (1989) Bioenergetic characterization of transient state phosphate uptake by the cyanobacterium Anacystis nidulans. Theoretical and experimental basis for a sensory mechanism adapting to varying environmental phosphate levels. Arch Microbiol 152: 353–361
Falkner G, Falkner R, Wagner F (1993) Adaptive phosphate uptake behaviour of the cyanobacterium Anacystis nidulans: analysis by a proportional flow-force relation. C R Acad Sci Paris Life Sci 316: 784–787
Falkner G, Wagner F, Falkner R (1994) On the relation between phosphate uptake and growth of the cyanobacterium Anacystis nidulans. C R Acad Sci Paris Life Sci 317: 535–541
Falkner G, Wagner F, Small JV, Falkner R (1995) Influence of fluctuating phosphate supply on the regulation of phosphate uptake by the blue-green alga Anacystis nidulans. J Phycol 31: 745–753
Falkner G, Wagner F, Falkner R (1996) The bioenergetic coordination of a complex biological system is revealed by its adaptation to changing environmental conditions. Acta Biotheor 44: 283–299
Falkner R, Falkner G (1989) Phosphate uptake by eukaryotic algae in cultures and by a mixed phytoplankton population in a lake: analysis by a force-flow relationship. Bot Acta 102: 283–286
Falkner R, Wagner F, Aiba H, Falkner G (1998) Phosphate-uptake behaviour of a mutant of Synechococcus sp. PCC7942 lacking one protein of the high-affinity phosphate-uptake system. Planta 206: 461–465
Fuhs GW (1969) Phosphorus content and rate of growth in the diatoms Cyclotella nana and Thalassiosira fluviatilis. J Phycol 5: 312–321
Ghilarov AM (1984) The paradox of the plankton reconsidered; or, why do species coexist? Oikos 43: 46–52
Goldstein AH (1994) Involvement of the quinoprotein glucose dehydrogenase in the solubilization of exogenous mineral phosphates by Gram-negative bacteria. In: Torriani- Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 197–203
Grover JP (1989) Phosphorus-dependent growth kinetics of 11 species of freshwater algae. Limnol Oceanogr 34: 341–348
Grover JP (1991a) Non-steady state dynamics of algal population growth: experiments with two chlorophytes. J Phycol 27: 70–79
Grover JP (1991b) Resource competition in a variable environment: phytoplankton growing according to the variable-internal-stores model. Am Nat 138: 811–835
Hameed HA, Kilinc S, McGowan S, Moss B (1999) Physiological tests and bioassays: aids or superfluities to the diagnosis of phytoplankton nutrient limitation? A comparative study in the Broads and the Meres of England. Eur J Phycol 34: 253–269
Healey FP (1980) Slope of the Monod equation as an indicator of advantage in nutrient competition. Microb Ecol 5: 281–286
Healey FP (1982) Phosphate. In: Carr NG, Whitton BA (eds) The biology of cyanobacteria. Blackwell, Oxford, pp 105–124
Hernandez I (1996) Analysis of the expression of alkaline phosphatase activity as a measure of phosphorus status in the red alga Porphyra umbilicalis (L) Klitzing. Bot Mar 39: 255–262
Higgins CF (1992) ABC transporters: from microorganisms to man. Annu Rev Cell Biol 8: 67–113
Huang BQ, Hong HS (1999) Alkaline phosphatase activity and utilization of dissolved organic phosphorus by algae in subtropical coastal waters. Mar Pollut Bull 39: 205–211
Hutchinson GE (1961) The paradox of the plankton. Am Nat 95: 137–145
Ikeya T, Ohki K, Takahashi M, Fujita Y (1997) Study on phosphate uptake of the marine cyanophyte Synechococcus sp. NIBBI071 in relation to oligotrophy environments in the open ocean. Mar Biol 129: 195–202
Istvanovics V, Herodek S (1995) Estimation of net uptake and leakage rates of orthophosphate from P-uptake kinetics by a linear force-flow model. Limnol Oceanogr 40: 17–32
Istvanovics V, Padisak J, Pettersson K, Pierson DC (1994) Growth and phosphorus uptake of summer phytoplankton in Lake Erken ( Sweden ). J Plankton Res 16: 1167–1196
Jenssen M, Olsson H, Pettersson K (1988) Phosphatases: origin, characterization, and function in lakes. Hydrobiologia 170: 157–175
Kallas T, Dahlquist FW (1981) Phosphorus-31 nuclear magnetic resonance analysis of internal pH during photosynthesis in the cyanobacterium Synechococcus. Biochemistry 20: 5900–5907
Kantrowitz ER (1994) Introduction: sructure and function of Escherichia coli alkaline
phosphatase. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 319–328
Kedem O, Caplan SR (1965) Degree of coupling and its relation to efficiency of energy conversion. Trans Faraday Soc 61: 1897–1911
Kilham SS (1978) Nutrient kinetics of freshwater planktonic algae using batch and semicontinuous methods. Mitt Int Ver Limnol 21: 147–157
Kornberg A (1994) Inorganic polyphosphate: a molecular fossil comes to life. In: Torriani- Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 204–208
Kornberg A, Rao NN, Ault-Riche D (1999) Inorganic polyphosphate: a molecule of many functions. Annu Rev Biochem 68: 89–125
Korteland J, Tommassen J, Lugtenberg B (1982) PhoE protein pore of the outer membrane of Escherichia coli K12 is a particularly efficient channel for organic and inorganic phosphate Biochim Biophys Acta 690: 282–289
Kubena BD, Luecke H, Rosenberg H, Quiocho FA (1986) Crystallization and X-ray diffraction studies of a phosphate-binding protein involved in active transport in Escherichia coli J Biol Chem 261: 7995–7996
Kuenzler EJ, Ketchum BH (1962) Rate of phosphorus uptake by Phaeodactylum tricornutum Biol Bull Woods Hole 123: 134–145
Kulaev IS, Vagabov VM (1983) Polyphosphate metabolism in micro-organisms. Adv Microb Physiol 24: 83–171
Lawerence BA, Suarez C, Depina A, Click E, Kolodny NH, Allen MM (1998) Two internal pools of soluble polyphosphate in the cyanobacterium Synechocystis sp. Strain PCC6308 — an in vivo P-31 NMR spectroscopic study. Arch Microbiol 169: 195–200
Lawry N, Jensen TH. (1979) Deposition of condensed phosphate as an effect of varying sulfur deficiency in the cyanobacterium Synechococcus sp. ( Anacystis nidulans ). Arch Microbiol 120: 1–7
Liss E, Langen P (1962) Versuche zur Polyphosphat-Überkompensation in Hefezellen nach Phosphatverarmung. Arch Mikrobiol 41: 383–392
Luecke H, Quiocho FA (1990) High specificity of a phosphate transport protein determined by hydrogen bonds. Nature 347: 402–406
Makino K, Shinagawa H, Amemura M, Nakata A (1986a) Nucleotide sequence of the phoB gene, the positive regulatory gene for the phosphate regulon of Escherichia coli K 12. J Mol Biol 190: 37–44
Makino K, Shinagawa H, Amemura M, Nakata A (1986b) Nucleotide sequence of the phoR gene, a regulatory gene for the phosphate regulon of Escherichia coli. J Mol Biol 192: 549–556
Makino K, Amemura M, Kim S-K, Nakata A, Shinagawa H (1994) Mechanism of
transcriptional activation of the phosphate regulon in Escherichia coli. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 5–12
Mann NH, Scanlan DJ (1994) The SphX protein of Synechococcus species PCC7942 belongs to a family of phosphate-binding proteins. Mol Microbiol 14: 595–596
Medveczky N, Rosenberg H (1970) The phosphate-binding protein of Escherichia coli. Biochim Biophys Acta 211: 158–168
Medveczky N, Rosenberg H (1971) Phosphate transport in Escherichia coli. Biochim Biophys Acta 241: 404–506
Monod J (1942) Recherches sur la croissance des cultures bactériennes. Hermann, Paris, 211 pp
Monod J (1950) La technique de la culture continue: theorie et applications. Ann Inst Pasteur Lille 79: 390–410
Morel FMM (1987) Kinetics of nutrient uptake and growth in phytoplankton. J Phycol 23: 137–150
Mueller H (1970) Das Wachstum von Nitzschia actinastroides ( Lemm.) v. Goor im
Chemostaten bei limitierender Phosphatkonzentration. Ber Dtsch Bot Ges 83:537–544
Mueller H (1972) Growth and phosphate requirements of Nitzschia actinastroides (Lemm.) v. Goor in batch and chemostat cultures under phosphate limitation. Arch Hydrobiol Suppl 38: 399–484
Nalewajko C, Lean DRS (1980) Phosphorus. In: Morris I (ed) The physiological ecology of phytoplankton. Blackwell, Oxford, pp 325–358
Nicklisch A (1999) Competition between the cyanobacterium Limnothrix redekei and some spring species of diatoms under P limitation. Int Rev Hydrobiol 84: 233–241
Olsen Y (1989) Evaluation of competitive ability of Staurastrum luetkemuellerii (Chlorophyceae) and Microcytis aeruginosa (Cyanophyceae) under P limitation. J Phycol 25: 486–499
Olsen Y, Vadstein O, Andersen T, Jensen A (1989) Competition between Staurastrum luetkemuellerii (Chlorophyceae) and Microcytis aeruginosa ( Cyanophyceae) under varying modes of phosphate supply. J Phycol 25: 499–508
Overbeeke N, Lugtenberg B (1980) Expression of outer membrane protein e of Escherichia coli K12 by phosphate limitation. FEBS Lett 112: 229–232
Overbeeke N, Bergmans H, Mansfeld FV, Lugtenberg B (1983) Complete nucleotide sequence of phoE, the structural gene for the phosphate limitation inducible outer membrane pore protein of Escherichia coli K12. J Mol Biol 163: 513–532
Perez-Llorens JL, Niell FX (1995) Short-term phosphate uptake kinetics in Zostera noltii Hornem: a comparison between excised leaves and sediment-rooted plants. Hydrobiologia 297: 17–27
Pietrobon D, Zoratti M, Azzone GF, Stucki JW, Walz D (1982) Non-equilibrium thermodynamic assessment of redox-driven H+ pumps in mitochondria. Eur J Biochem 127: 483–494
Pietrobon D, Zoratti M, Azzone GF (1983) Molecular slipping in redox and ATPase H+ pumps. Biochim Biophys Acta 723: 317–321
Rao NN, Roberts MF, Torriani A, Yashphe J (1993) Effect of glpT and glpD mutations on expression of the phoA gene in Escherichia coli. J Bacteriol 175: 74–79
Rao NN, Kar A, Roberts MF, Yashphe J, Torriani-Gorini A (1994) Phosphate, phosphorylated metabolites, and the Pho regulon of Escherichia coli. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington DC, pp 22–29
Ray JM, Bhaya D, Block MA, Grossman AR (1991) Isolation, transcription, and inactivation of the gene for an atypical alkaline phosphatase of Synechococcus sp. strain PCC7942. J Bacteriol 173: 4297–4309
Reynolds CS (1993) The ecology of freshwater phytoplankton. Cambridge University Press, Cambridge, 384 pp
Reynolds CS (1998) The state of freshwater ecology. Freshwater Biol 39: 741–753
Rhee GY (1973) A continuous culture study of phosphate uptake, growth rate and polyphosphate in Scenedesmus sp.. J Phycol 9: 495–506
Rhee GY (1974) Phosphate uptake under nitrate limitation by Scenedesmus sp. and its ecological implications. J Phycol 10: 470–475
Rhee GY (1982) Effects of environmental factors and their interactions on phytoplankton growth. In: Marshall KC (ed) Advances in microbial ecology, vol 6. Plenum Press New York, pp 33–74
Rigler FH (1956) A tracer study of phosphorus cycle in lake water. Ecology 37: 550–562
Rigler FH (1968) Further observations inconsistent with the hypothesis that the molybdenium blue method measures orthophosphate in lake water. Limnol Oceanogr 13: 7–13
Ritchie RJ, Trautman DA, Larkum A WD (1997) Phosphate uptake in the cyanobacterium Synechococcus R-2 PCC7942. Plant Cell Physiol 38: 1232–1241
Rosenberg H (1987) Phosphate transport in prokaryotes. In: Rosen BP, Silver S (eds) Ion transport in prokaryotes. Academic Press, New York, pp 205–248
Rosenberg H, Gerdes RG, Chegwidden K (1977) Two systems for the uptake of phosphate in Escherichia coli. J Bacteriol 131: 505–511
Rosenberg H, Gerdes RG, Harold FM (1979) Energy coupling to the transport of inorganic phosphate in Escherichia coli K-12. Biochem J 178: 133–137
Ruiz RG, Hernandez I, Lucena J, Niell FX (1997) Preliminary studies on the significance of alkaline phosphatase activity in the diatom Phaeodactylum tricornutum Bohlin. Sci Mar 61: 517–525
Russell LM, Rosenberg H (1979) Linked transport of phosphate, potassium ions and protons in Escherichia coli. Biochem J 184: 13–21
Russell LM, Rosenberg H (1980) The nature of the link between potassium transport and phosphate transport in Escherichia coli. Biochem J 188: 715–723
Salonen K, Jones RI, de Haan H, James M (1994) Radiotracer study of phosphorus uptake by plankton and redistribution in the water column of a small humic lake. Limnol Oceanogr 39: 69–83
Scanlan DJ, Wilson WH (1999) Application of molecular techniques to addressing the role of P as a key effector in marine ecosystems. Hydrobiologia 401: 149–175
Scanlan DJ, Mann NH, Carr NG (1993) The response of the picoplanktonic marine cyanobacterium Synechococcus species WH7803 to phosphate starvation involves a protein homologous to the periplasmic phosphate-binding protein of Escherichia coli. Mol Microbiol 10: 181–191
Scanlan DJ, Bourne JA, Mann NH (1997) A putative transcriptional activator of the Crp/Fnr family from the marine cyanobacterium Synechococcus sp. WH7803. J Appi Phycol 8: 565–567
Schindler DW (1977) Evolution of phosphorus limitation in lakes. Science 195: 260–262
Schlegel HG (1985) Allgemeine Mikrobiologie. Thieme, Stuttgart, 571 pp
Schwoerbel J (1984) Einfuehrung in die Limnologie. Fischer, Stuttgart, 233 pp
Segel I (1975) Enzyme kinetics. Behavior and analysis of rapid equilibrium and steady-state enzyme systems. Wiley, New York, 957 pp
Shapiro JA (1998) Thinking about bacterial populations as multicellular organisms. Annu Rev Microbiol 52: 81–104
Silver S, Walderhaug M (1992) Gene regulation of plasmid- and chromosome-determined inorganic ion transport in bacteria. Microbiol Rev 56: 195–228
Simonis W, Urbach W (1973) Photophosphorylation in vivo. Annu Rev Plant Physiol 24: 89–114
Simonis W, Bornefeld T, Lee-Kaden J, Majumdar K (1974) Phosphate uptake and photophosphorylation in the blue-green alga Anacystis nidulans. In: Zimmermann U, Dainty J (eds) International workshop on membrane transport in plants and plant organelles. Springer, Berlin Heidelberg New York, pp 220–225
Sommer U (1983) Nutrient competition between phytoplankton species in multispecies
chemostat experiments. Arch Hydrobiol 96:399–416
Sommer U (1985) Comparison between steady state and non-steady state competition: experiments with natural phytoplankton. Limnol Oceanogr 30: 335–346
Sommer U (1989) Nutrient status and nutrient competition of phytoplankton in a shallow, hypertrophic lake. Limnol Oceanogr 34: 1162–1173
Sommer U (1993) Phytoplankton competition in Plußsee: a field test of the resource-ratio hypothesis. Limnol Oceanogr 38: 838–845
Sommer U (1994) Planktologie. Springer, Berlin Heidelberg New York, 274 pp
Spijkerman E, Coesel PFM (1998) Different response mechanisms of two planktonic desmid species ( Chlorophyceae) to a single saturating addition of phosphate. J Phycol 34: 438–445
Stein WD (1981) Concepts of mediated transport. In: Bonting SL, De Pont JJ (eds) Membrane transport. Elsevier, Amsterdam, pp 123–157
Stucki JW (1980) The optimal efficiency and the economic degrees of coupling of oxidative phosphorylation. Eur J Biochem 109: 269–283
Stucki JW, Compiani M, Caplan SR (1983) Efficiency of energy conversion in model biological pumps. Optimization by linear nonequilibrium thermodynamic relations. Biophys Chem 18: 101–109
Surin BP, Rosenberg H, Cox GB (1985) Phosphate-specific transport system of Escherichia coli: nucleotide sequence and gene-polypeptide relationships. J Bacteriol 161: 189–198
Thellier M (1970) An electrokinetic interpretation of the functioning of biological systems and its application to the study of mineral salts absorption. Ann Bot 34: 983–1009
Tilman D (1977) Resource competition between planktonic algae: an experimental and theoretical approach. Ecology 58: 338–348
Tilman D, Sterner RW (1984) Invasions of equilibria: tests of resource competition using two species of algae. Oecologia 61: 197–200
Tilman D, Kilham SS, Kilham P (1982) Phytoplankton community ecology: the role of limiting nutrients. Annu Rev Ecol Syst 13: 349–372
Titman D (1976) Ecological competition between algae: experimental confirmation of resource-based competition theory. Science 192: 463–465
Torriani A (1990) From cell membrane to nucleotides: the phosphate regulon in Escherichia coli. Bioessays 12: 371–376
Vadstein O (1998) Evaluation of competitive ability of two heterotrophic planktonic bacteria under phosphorus limitation. Aquat Microb Ecol 14: 119–127
Vadstein O, Olsen Y, Reinertsen H, Jensen A (1993) The role of planktonic bacteria in phosphorus cycling in lakes — sink and link. Limnol Oceanogr 38: 1539–1544
Van Veen HW (1997) Phosphate transport in prokaryotes: molecules, mediators and mechanisms. Antonie van Leeuwenhoek 72: 299–315
Van Veen HW, Abee T, Kortstee GJJ, Konings WN, Zehnder AJB (1994a) Translocation of metal phosphate via the phosphate inorganic transport (Pit) system of Escherichia coli. Biochemistry 33: 1766–1770
Van Veen HW, Abee T, Kortstee GJJ, Konings WN, Zehnder AJB (1994b) Phosphate inorganic transport (Pit) system in Escherichia coli and Acinetobacter johnsonii. In: Torriani-Gonni A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 43–49
Wagner F, Falkner G (1992) Concomitant changes in phosphate uptake and photophosphorylation in the blue-green alga Anacystis nidulans during adaptation to phosphate deficiency. J Plant Physiol 140: 163–167
Wagner F, Gimona M, Ahorn H, Peschek GA, Falkner G (1994) Isolation and functional reconstitution of a phosphate binding protein of the cyanobacterium Anacystis nidulans induced during phosphate-limited growth. J Biol Chem 269: 5509–5511
Wagner F, Falkner R, Falkner G (1995) Information about previous phosphate fluctuations is stored via an adaptive response of the high-affinity phosphate uptake system of the cyanobacterium Anacystis nidulans. Planta 197: 147–155
Wagner F, Sahan E, Falkner G (1999) The complex relation between phosphate uptake and photosynthetic CO2 fixation in the cyanobacterium Anacystis nidulans. In: Peschek GA Loefffelhardt W, Schmetterer G (eds) The phototrophic prokaryotes. Kluwer Academic/Plenum Publishers, New York, pp 739–744
Wagner F, Sahan E, Falkner G (2000) The establishment of coherent phosphate uptake behaviour by the cyanobacterium Anacystis nidulans. Eur J Phycol 35: 243–253
Wagner KU, Masepohl B, Pistorius EK (1995) The cyanobacterium Synechococcus sp. strain PCC7942 contains a second alkaline phosphatase encoded by phoV. Microbioloev 141: 3049–3058
Wang Z, Choudhary A, Ledvina PS, Quiocho FA (1994) Fine tuning the specificity of the periplasmic phosphate transport receptor. Site-directed mutagenesis, ligand binding, and crystallographic studies. J Biol Chem 269: 25091–25094
Wanner BL (1993) Gene regulation by phosphate in enteric bacteria. J Cell Biochem 51: 47–54
Wanner BL (1994) Multiple controls of the Escherichia coli Pho regulon by the P, sensor PhoR, the catabolite regulatory sensor CreC, and acetyl phosphate. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology Washington, DC, pp 13–21
Wanner BL (1996) Phosphorus assimilation and control of the phosphate regulon. In: Neidhardt FC (ed) Escherichia coli and Salmonella. ASM Press, Washington, pp 1357–1381
Watson GMF, Scanlan DJ, Mann NH (1996) Characterization of the genes encoding a phosphate-regulated two-component sensory system in the marine cyanobacterium Synechococcus sp. WH7803. FEMS Microbiol Lett 142: 105–109
Webb DC, Cox GB (1994) Proposed mechanism for phosphate translocation by the phosphate- specific transport (Pst) system and role of the Pst system in phosphate regulation. In: Torriani-Gorini A, Yagil E, Silver S (eds) Phosphate in microorganisms. American Society for Microbiology, Washington, DC, pp 37–42
Webb DC, Rosenberg H, Cox GB (1992) Mutational analysis of the Escherichia coli phosphate-specific transport system, a member of the traffic ATPase (or ABC) family of membrane transporters. J Biol Chem 267: 24661–24668
Welch GR, Easterby JS (1994) Metabolic channeling versus free diffusion: transition-time analysis. Trends Biochem Sci 19: 193–197
Willsky GR, Malamy MH (1980a) Characterization of two genetically separable inorganic phosphate transport systems in Escherichia coli. J Bacteriol 144: 356–365
Willsky GR, Malamy MH (1980b) Effect of arsenate on inorganic phosphate transport in Escherichia coli. J Bacteriol 144: 366–374
Yoshida A (1955) Studies on metaphosphate. II. Heat of hydrolysis of metaphosphate extracted from yeast cells. J Biochem Tokyo 42: 163–168
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2001 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Wagner, F., Falkner, G. (2001). Phosphate Limitation. In: Rai, L.C., Gaur, J.P. (eds) Algal Adaptation to Environmental Stresses. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-59491-5_4
Download citation
DOI: https://doi.org/10.1007/978-3-642-59491-5_4
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-63996-8
Online ISBN: 978-3-642-59491-5
eBook Packages: Springer Book Archive