Abstract
Hybrid imaging is beneficial for improved medical diagnosis and therapy planning today. Hybrid imaging describes the prospective correlation of two or more complementary sets of imaging information, such as functional and anatomical image volumes. Correlation can be performed through physically combined imaging modalities, such as PET/CT, SPECT/CT, or PET/MR. Here we present first results from employing fully integrated PET/MR tomography for intensity-modulated radiotherapy (IMRT) treatment planning in patients with meningioma using [68Ga]-DOTATOC as the biomarker of choice. Combined PET/MR offers higher soft tissue contrast and the ability to add functional information to the plain combination of MR-based anatomy and PET-based metabolic and molecular information. Furthermore, fully integrated PET/MR employs novel PET technology that is neither available in PET-only nor PET/CT systems. Despite the current lack of broad clinical evidence, integrated PET/MR may become particularly important and clinically useful for improved, individualized RT therapy planning for brain lesions. In particular, logistical and diagnostic benefits of integrated PET/MR-based treatment planning over treatment planning based on PET/CT data may be expected in meningioma patients.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Noninvasive medical imaging is an integral part of state-of-the-art patient management. Medical imaging can be categorized broadly into anatomical and functional imaging, making reference, respectively, to the ability to acquire information of the anatomy such as bone and soft tissue structures through the use of computed tomography (CT) or magnetic resonance imaging (MRI), and to acquire information on molecular or biochemical processes such as hypermetabolism as a criterion for potential cancerous tissue infiltration (Warburg 1956) through the use of positron emission tomography (PET).
Historically, medical devices to image either anatomical structure or functional processes have developed along somewhat independent paths, but the usefulness of combining anatomical and functional planar images was evident to physicians as early as the 1960s. The recognition that combining images from different modalities offers significant diagnostic advantages gave rise to sophisticated software techniques to co-register structure and function (Townsend 2008). For relatively rigid objects such as the brain, software can successfully align images from MR, CT, and PET, whereas in regions of the body outside the brain, accurate alignment is difficult owing to the large number of possible degrees of freedom.
Alternatives to software-based fusion have now become available through hardware fusion (Beyer et al. 2011). Combined, or hybrid, tomographs such as SPECT/CT or PET/CT can acquire co-registered structural and functional information sequentially within a single study. The data are complementary, allowing CT to accurately localize functional abnormalities and SPECT or PET to highlight areas of abnormal tracer uptake. This is of particular importance in case PET (or SPECT) images of the distribution of highly specific tracers reveal very little anatomical background information. Figure 1 illustrates this with the example of a highly specific tracer ([68Ga]-labeled somatostatin-receptor ligand: [68Ga]-DOTATOC), which specifically binds to somatostatin-expressing receptors. Several studies are available today demonstrating the utility and efficacy of combined PET/CT imaging using a variety of tracers (Czernin et al. 2007; Nestle et al. 2009). As a consequence, following their recent introduction into the clinic, combined PET/CT and SPECT/CT devices are now playing an increasing role in diagnosis and staging of patients with oncological and inflammatory diseases.
[68Ga]-DOTATOC study of a patient with neuroendocrine tumor. From left to right axial images of: a attenuation-corrected PET, b CT, and c PET/CT. Without any anatomical correlated uptake, highly specific PET tracer uptake cannot be localized sufficiently; here, combined imaging becomes mandatory. Data courtesy of Hofmann (Hannover) and Bockisch (Essen)
However, despite the success and wide distribution of PET/CT, there are some shortcomings in use of CT as the anatomical complement to PET. CT uses a source of ionizing radiation for imaging and, therefore, adds significant radiation dose to the overall examination (Brix et al. 2005), which may raise concern in selected populations such as adolescents, females, and patients with benign diseases. Further, CT provides comparatively low soft tissue contrast unless additional CT contrast agent are used. MR, on the other hand, does not suffer from these two major disadvantages and, in addition, offers more advanced functional imaging information, such as dynamic contrast-enhanced (DCE), diffusion-weighted imaging (DWI), or magnetic resonance spectroscopy (MRS), without adding to the overall radiation exposure burden.
The combination of PET and MR in a single imaging system has the potential to become a multimodality imaging technology of choice combining anatomical, functional, and molecular information into a single multiparametric imaging modality. Nonetheless, it is hard to make hypotheses on the main clinical applications of combined PET/MR at this stage (Beyer and Pichler 2009), where first prototype systems are being validated in clinical and research settings (Pichler et al. 2010). The potential areas of application of combined PET/MR imaging in brain imaging, for example, benefit greatly from the additional morphologic information provided by MRI. Combined amino acid PET and MR imaging is likely to enhance diagnostic sensitivity for gliomas and may allow closer correlation between tracer uptake and metabolic changes (e.g., choline peaks in MR spectroscopy) in neoplastic tissue (Bisdas et al. 2009). Likewise, arterial spin-labeling estimations of perfusion and diffusion changes occurring in low-grade gliomas may be studied in conjunction with different PET tracers to establish reliable disease markers. Boss and colleagues recently evaluated simultaneous PET/MRI for assessing intracranial tumors using [11C]-methionine for glioma or [68Ga]-DOTATOC for meningioma. They demonstrated image quality and quantitative data achieved from PET/MRI to be similar to that of PET/CT (Boss et al. 2010).
Given the availability of combined PET/MR systems, functional MRI and molecular PET image data can be acquired simultaneously, thus offering much improved spatial and temporal co-registration, which may become beneficial for image-guided radiotherapy (RT) treatment planning of various oncological diseases (Werner et al. 2011).
2 Methods
2.1 PET/MR Tomography
MR requires very high field homogeneity. Furthermore, the presence of PET detectors within the field could interfere with MR imaging. In contrast, PET detectors have to withstand not only a high static field level (up to 3 T for clinical MR) but also the rapidly changing field gradients required by the imaging process. PET/MR was destined to remain in the preclinical arena for another decade until, in 2006, the first simultaneous MR and PET images of the human brain were acquired (Schmand et al. 2007).
Existing hardware concepts for clinical PET/MR imaging essentially fall into three categories (Fig. 2): (a) separate gantries operated in different rooms, (b) gantries arranged in the direction of the main scanner axis with a patient-handling system mounted in between, and (c) fully integrated systems. The first design, presented in 2006 and also being the most challenging (Fig. 2c), is based on a PET detector ring designed as an insert that can be placed inside a Siemens 3-T Trio MR scanner (Siemens Healthcare). This prototype system (BrainPET) was anticipated for brain imaging only (Schlemmer et al. 2008). A similar design was replicated and made available for whole-body applications in late 2010 by the same company.
System designs for combined PET/MR tomography: a PET (or PET/CT) and MR are situated in separate rooms and combined via a mobile, docking table platform, b PET and MR are within the same room with a joint table platform, and c integrated design where the PET component fits within the MRI gantry, and data are acquired simultaneously
The coplanar PET/MR concept (Fig. 2b), first presented in 2010, is based on a tandem design of a whole-body time-of-flight (TOF) (Budinger 1983) PET system and a 3-T Philips Achieva MR system (Philips Healthcare, Cleveland, USA) with a rotating table platform in between. Through minor modifications of the PET detector system (e.g., orientation of the photomultiplier tube (PMT), minor shielding) the PET gantry can be operated in close proximity to the 3-T MRI system.
The third design was proposed by GE Healthcare in late 2010 and is available as prototype technology only. This design is based on a combination of a dual-modality PET/CT and a 3-T MR system that are operated side-by-side in separate rooms next to each other; patients are shuttled from one system to the other without getting off the bed. This approach substitutes the challenges of hardware integration for immense logistical challenges in timing access to the two systems while minimizing patient motion in between examinations. However, this approach has been argued as the most cost-effective compared with fully integrated PET/MR based on workflow aspects and machine utilization (Schulthess and Burger 2010).
In addition to the technical challenges of combining PET and MRI, which increase with the amount of PET and MR system integration, the necessary attenuation correction factors for the PET emission data must be derived from PET/MR measurements (Hofmann et al. 2009). While in PET/CT PET attenuation data can be derived from transforming available CT transmission images into maps of attenuation coefficients at 511 keV (Kinahan et al. 2003), no such transmission data are available in PET/MR. This is primarily due to the lack of physical space to host a transmission source. Therefore, PET/MR requires novel approaches to MR-based attenuation correction (MR-AC). While segmentation-based approaches have been proposed to classify tissues on MR images and to assign respective attenuation coefficients, which work well in brain imaging (Zaidi 2007); MR-AC in extracerebral applications is, however, much more challenging (Beyer et al. 2008).
2.2 Diagnosis and RT Treatment Planning of Meningioma
Meningiomas are mesodermal tumors originating from the arachnoid membrane and represent one of the most frequently diagnosed types of brain tumor. Incidence is about 25 per million people, peaking in patients between 40 and 70 years and increasing with age. Henze et al. (2001) demonstrated for the first time that PET imaging using [68Ga]-DOTATOC [1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA)-d-Phe1-Tyr3-octreotide (TOC)] is a promising diagnostic test for meningioma patients. This is due to the fact that meningiomas are known to express somatostatin receptor subtype 2 (SSTR2) for which, consequently, [68Ga]-DOTATOC can be used as a biomarker to visualize the extension of meningiomas in PET or PET/CT studies. Henze et al. (2001) showed good correlation of tracer boundaries with anatomical delineation obtained from (co-registered) CT and MR image volumes, even for small meningiomas of 7 mm.
Treatment of meningiomas can be difficult due to their proximity to organs at risk (e.g., brainstem, cranial nerves). Although surgical resection is the main treatment approach, RT has been reported to be a highly effective treatment modality (Milker-Zabel et al. 2007). Until now, RT treatment planning has been based mainly on CT and contrast-enhanced MRI (Debus et al. 2001). However, in the case of quasiplanar growth, these imaging techniques have limitations concerning visualization of the exact tumor extension. Thus, PET or PET/CT is highly valuable as an additional imaging modality for more accurate target volume (TV) delineation (Milker-Zabel et al. 2006).
Intensity-modulated radiation therapy (IMRT) is an effective technique for treating meningiomas with high levels of conformality. A combination of CT, MR, and [68Ga]-DOTATOC-PET imaging has been reported to improve TV delineation for IMRT treatment planning (Gehler et al. 2009; Thorwarth et al. 2011).
2.3 68Ga-DOTATOC PET/MR Protocol
Here we present three cases of patients with meningiomas undergoing [68Ga]-DOTATOC PET/MR imaging. All patients had a clinical indication for a PET/CT study, which was performed prior to the PET/MR study.
The PET/CT examination was performed with a Biograph HiRez 16 unit (Siemens Healthcare, Knoxville, USA), which is equipped with 4 × 4 × 16 mm3 lutetium oxyorthosilicate (LSO) scintillation crystals in combination with a 16-slice CT. Patients were injected intravenously with an average activity of 150 MBq of [68Ga]-DOTATOC. Static emission scans of two overlapping bed positions in the tumor region were acquired 40 min post injection (p.i.), with acquisition time of 4 min per bed position. PET images were reconstructed iteratively using a two-dimensional (2D) ordered-subset expectation-maximization (OSEM) algorithm with 4 iterations and 8 subsets. CT-based attenuation and scatter correction was performed. Reconstructed PET data had in-plane resolution of 6 mm and slice thickness of 5 mm.
PET/MR imaging commenced immediately after the completion of the PET/CT acquisition in a different part of the hospital. A prototype PET/MR system dedicated to brain imaging (BrainPET; Siemens Healthcare, Knoxville, USA) was used (Schmand et al. 2007). Briefly, a clinical 3-T MR system (Magnetom Trio; Siemens Healthcare, Erlangen, Germany) is equipped with an MR-compatible brain PET insert. The PET detector ring consists of 2.5 × 2.5 × 20 mm3 LSO scintillation crystals in combination with avalanche photodiodes (APD). The PET resolution is less than 3 mm in the center of the field of view. PET data acquisition started approximately 100 min p.i. List-mode data were acquired over a period of 30 min to compensate for the longer uptake time. PET reconstruction was performed using a 3D ordinary Poisson OSEM resolution-modeling algorithm with 6 iterations and 16 subsets, 3D scatter correction, and MRI-based attenuation correction (Hofmann et al. 2008).
MRI was performed using the birdcage transmit/receive coil, which is suitable for all routine clinical examinations. The MR sequences used for examinations of the brain with combined PET/MR are described in detail elsewhere (Boss et al. 2010). The combined PET/MR machine allows PET and MR acquisition of data not only in the same geometrical localization but also simultaneously.
2.4 Radiotherapy Treatment Planning
For all patients, IMRT treatment planning was performed using 6 MV photons from seven different beam angles. IMRT was planned with a dynamic delivery technique using a linear accelerator equipped with a multileaf collimator with leaf widths of 4 mm in the isocenter plane (Elekta Synergy S, Crawley, UK). For accurate patient positioning, a thermoplastic mask system was used in addition to onboard position verification with the cone-beam CT system.
First, IMRT TVs were delineated manually based on a combination of CT, [68Ga]-DOTATOC PET, and separately acquired MR data (Gehler et al. 2009). A second set of contours was created using only combined PET/MR imaging data in addition to the planning CT required for RT dose calculation. IMRT treatment planning was performed using the PET/CT-based TVs as reference, which was then cross-evaluated for the PET/MR-based TVs to evaluate the dosimetric differences imposed by PET/MR-guided IMRT planning.
2.5 Clinical Examples
Case 1 A 73-year-old female presented with recurrence of an atypical meningioma (WHO II) in the right anterior central region 15 months after complete resection. Clinical findings before first resection were left-sided gait abnormality and disease progression on MR imaging. Postoperative imaging consisting of MRI and [68Ga]-DOTATOC-PET/CT did not determine any residual tumor. Due to aggravation of the pre-existing gait abnormality 15 months after surgery, another set of MRI and [68Ga]-DOTATOC-PET/CT examinations was performed, and recurrent disease was detected. This case was discussed in an interdisciplinary neurooncology tumor board, and radiation treatment was recommended.
First, TV delineation was performed separately on PET/CT and MR image volumes. Fusion of these two TVs resulted in a total gross target volume (GTVPET/CT+MR) of 12.7 mL. In a second step, a corresponding GTVPET/MR was defined exclusively based on simultaneous PET/MR, which had size of 13.5 mL. Subsequently, both GTVs were expanded isotropically by 4 mm to create the respective planning target volumes (PTVs). Figure 3 shows the TVs defined on the basis of PET/CT and MR (PTVPET/CT+MR and GTVPET/CT+MR) (Fig. 3b) and PET/MR (PTVPET/MR and GTVPET/MR) (Fig. 3d) superimposed on the planning CT. PET/CT- and PET/MR-based TV delineation resulted in PTVs of 28.0 and 31.5 mL, respectively.
Patient 1: 73-year-old female with meningioma WHO grade II following [68Ga]-DOTATOC PET/CT and PET/MR: axial PET/CT (a) and PET/MR image (c), GTVs based on PET/CT (yellow) and additional MR (green) data in addition to the resulting PTV (red) (b), and after TV delineation based solely on hybrid PET/MR (PET: green, simultaneously acquired MR: orange, PTV: purple) (d). IMRT plan created for TVs based on PET/CT only (red) and cross-evaluated for TVs delineated on simultaneous PET/MR image data (purple) shown in (d). The arrow shows an infiltrated tumor region only visualized on hybrid PET/MR, which would have been significantly underdosed after TV delineated based solely on combined PET/CT
When using PET/MR for TV delineation, the TV was slightly enlarged compared with that derived from PET/CT images. Tumor parts infiltrating into critical regions were visible more clearly on PET/MR, thanks to the higher signal-to-noise ratio (SNR) on the simultaneously acquired PET/MR data. As a consequence, the IMRT plan designed on the basis of PET/CT imaging would have significantly underdosed these specific regions of the tumor only visible on the hybrid PET/MR data (Fig. 3d, arrow) (Thorwarth et al. 2011).
Case 2 A 72-year-old female patient presented 17 years ago with dizziness and focal signs of a parasagittal cortex syndrome including sensory disturbance of the right side and headache. After complete resection of a left-sided parasagittal meningioma (WHO I) the symptoms resolved. In 2010, increasing paraesthesia of the right hand and right side of face were observed. Two left-sided subdural recurrences were detected by CT and MRI, extending from frontal to parietal parts of trepanation. Within 3 months, one of the two manifestations progressed from 23 × 18 to 29 × 25 mm. The other lesion did not change with 19 × 15 mm. Hybrid [68Ga]-DOTATOC-PET/CT and PET/MR confirmed both manifestations. Radiation treatment options were recommended, in particular in view of the proximity of the lesion to the sinus sagittalis superior.
PET/CT-based TV delineation yielded a GTV of 18.0 mL, whereas the PET/MR-based TV was slightly larger (19.0 mL). Nevertheless, both GTVs were located within the same geographical area and did not show any significant difference. IMRT treatment planning did not differ significantly (Fig. 4).
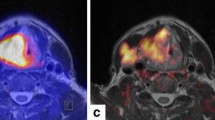
Case 3 A 69-year-old female treated with surgery for a 46 × 47 mm2 atypical meningioma (WHO II) in the right frontal region. The initial signs were weakness of the left hand and sensory disturbance of the seventh cranial nerve. One year later, a 9 × 14 × 13 mm3-sized recurrence was diagnosed on MRI in the right frontal region. On additional MRI 3 months later, a smaller lesion of 8 mm diameter was detected close to the main tumor mass in the right frontal area, which had progressed in size to 16 × 25 × 14 mm3 (Fig. 6a, b). An additional satellite lesion close to the sinus frontalis showed only very moderate tracer uptake on the [68Ga]-DOTATOC PET/CT 60 min p.i. (Fig. 5), but was clearly visible on the combined [68Ga]-DOTATOC PET/MR (Fig. 6c, d). Taking prior complications after the first surgery into account (bony infection followed by repeated surgery with duraplasty), the interdisciplinary recommendation for treatment of recurrence was RT.
[68Ga]-DOTATOC PET/CT image data of patient 3. Axial CT (a) and PET/CT (b) image show the main tumor mass. Offset axial CT (c) and PET/CT (d) show the small satellite lesion in the frontal region. Comparatively low tracer retention can be seen in this lesion (arrow). PET maximum-intensity projection is shown in (e)
Patient 3: 69-year-old female with meningioma WHO II in right frontal region. In addition to the two lesions detected on PET/CT (Fig. 5), PET/MR reveals third satellite lesion in the frontal sinus: transverse (a) and coronal (b) view of the main tumor mass, transverse (c) and sagittal (d) view of a small satellite lesion in the dorsal region of the frontal sinus (arrow)
At the time, the hybrid PET/MR data originating from this system could not be used for diagnostic and therapeutic decisions. Therefore, this patient was treated based on PET/CT information only. However, due to the very moderate tracer uptake in the small frontal satellite lesion, which was not taken into account during PET/CT-based target contouring, PET/MR-based TVs were significantly larger. In contrast, delayed PET/MR images reveal higher resolution (Fig. 6). The IMRT treatment plan for this patient, taking into account the frontal satellite lesion detected on PET/MR only, is shown in Fig. 7.
Patient 3: 69-year-old female with meningioma WHO II in right frontal region. IMRT plan taking into account the small tumor satellite in the frontal region which had been visualized on [68Ga]-DOTATOC PET/MR only (a). Dose distribution in the main tumor region (b), PTV is outlined in red. Seven beam angles for IMRT delivery are schematically represented by yellow lines
3 Discussion
The three cases presented here illustrate the practical application of [68Ga]-DOTATOC PET/MR in a clinical setting. In two cases (patients 1 and 3) the tumor identified on PET/MR was not completely covered with the prescribed radiation dose derived from prior PET/CT imaging. As a consequence, PET/MR imaging with improved PET resolution might be highly beneficial for improved TV delineation for RT treatment of meningioma, although further studies with histology verification or long-time outcome are required.
Somatostatin receptor (SSTR) imaging was shown to provide improved characterization of tumors near the skull base in case of unclear diagnosis and anatomical information on MR imaging data. Also for patients being considered for stereotactic RT, [68Ga]-DOTATOC PET imaging is beneficial to differentiate between meningiomas/neurinomas and optical nerve gliomas (Henze et al. 2001). Furthermore, SSTR imaging using [68Ga]-DOTATOC results in extremely high tumor-to-blood ratios (TBR) even in very small regions (Henze et al. 2005). On the one hand such high TBRs yield high diagnostic quality, but on the other hand they require anatomical correlates and—potentially—simultaneous PET/MR imaging for improved target localization.
Alternatively, a number of different PET tracers can be used to accurately visualize meningiomas for improved TV definition, such as [18F]-fluorodeoxyglucose ([18F]-FDG (Lee et al. 2009), O-(2-[18F]-fluoroethyl)-l-tyrosine ([18F]-FET) (Rutten et al. 2007), [11C]-acetate (Liu et al. 2010), [11C]-choline (Fallanca et al. 2009), or [11C]-methionine ([11C]-MET) (Astner et al. 2008; Grosu et al. 2006). More specifically, Lee et al. demonstrate that FDG uptake in meningiomas was predictive of tumor recurrence (Lee et al. 2009). Rutten et al. (2007) showed that meningiomas are visualized clearly after RT using [18F]-FET but report discrepancies between PET- and MR-based TV. Grosu et al. (2006) showed that [11C]-MET helps to reduce interobserver variability in TV delineation of meningiomas.
Improved TV definition using [68Ga]-DOTATOC PET/CT was demonstrated by several studies, but nevertheless PET information as obtained from PET/CT imaging was reported to be of limited value in certain tumor regions such as the pituitary gland and lesions next to the cavernous sinus (Milker-Zabel et al. 2006; Hyun et al. 2011). Here, PET/MR might be advantageous, as simultaneously acquired functional MR data may help to further differentiate tumor diagnosis based on PET.
Another advantage for PET/MR is intrinsic co-registration that dismisses retroregistration, for which more background information would be required. Therefore, [68Ga]-DOTATOC can be used with PET/MR as well as MET and FET, which are reported to contain more background information useful for mutual information (MI)-based registration, which is no longer required in PET/MR (Astner et al. 2008).
Co-registered MR information can be used to define microscopic tumor growth (Nyuyki et al. 2010) that is known to be a problem for PET/CT-guided RT in meningioma (Gehler et al. 2009). Here, accurate image matching is absolutely necessary to accurately locate infiltrative regions of the tumor by means of high-precision RT.
4 Conclusion
Simultaneous [68Ga]-DOTATOC-PET/MR imaging is clinically feasible in brain applications. It may be beneficial for more accurate TV delineation of meningioma, thus benefiting IMRT treatment planning and other highly conformal RT techniques. Simultaneous brain PET/MR imaging may allow more detailed visualization and functional–anatomical co-registration of meningiomas, especially in small infiltrated regions that may not be detected with larger PET detector systems. In addition, combined PET/MR imaging helps expedite clinical workflow.
In general, within the next years, we may expect to see PET/MR gaining momentum and find a place in clinical routine. This advance can be fostered by the adoption of highly specific PET tracers that contain less anatomical background information on PET only, for example. Hybrid imaging by means of PET/CT and PET/MR will be mandatory when using [68Ga]-labeled tracers.
Abbreviations
- APD:
-
Avalanche photodiode
- CT:
-
Computed tomography
- DCE:
-
Dynamic contrast enhanced
- DOTATOC:
-
1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA)-d-Phe1-Tyr3-octreotide (TOC)
- DWI:
-
Diffusion-weighted imaging
- FDG:
-
[18F]-Fluorodeoxyglucose
- FET:
-
O-(2-[18F]-Fluoroethyl)-l-tyrosine
- GTV:
-
Gross target volume
- IMRT:
-
Intensity-modulated radiation therapy
- MET:
-
Methionine
- MI:
-
Mutual information
- mL:
-
Milliliter
- MRI:
-
Magnetic resonance imaging
- MRS:
-
Magnetic resonance spectroscopy
- OSEM:
-
Ordered-subsets expectation maximization
- p.i.:
-
Post injection
- PET:
-
Positron emission tomography
- PTV:
-
Planning target volume
- RT:
-
Radiation therapy
- SNR:
-
Signal-to-noise ratio
- SPECT:
-
Single-photon emission computed tomography
- TBR:
-
Tumor-to-blood ratio
- TV:
-
Target volume
References
Astner ST, Dobrei-Ciuchendea M, Essler M et al (2008) Effect of 11C-methionine-positron emission tomography on gross tumor volume delineation in stereotactic radiotherapy of skull base meningiomas. Int J Radiat Oncol Biol Phys 72(4):1161–1167
Beyer T, Pichler B (2009) A decade of combined imaging: from a PET attached to a CT to a PET inside an MR. Eur J Nucl Med Mol Imaging 36(Suppl 1):S1–S2
Beyer T, Weigert M, Quick HH et al (2008) MR-based attenuation correction for torso-PET/MR imaging: pitfalls in mapping MR to CT data. Eur J Nucl Med Mol Imaging 35(6):1142–1146
Beyer T, Freudenberg LS, Townsend DW et al (2011) The future of hybrid imaging—part 3: combined PET/MR and small animal imaging, other imaging combinations and additional concerns. Insights Imaging 2(3):235–246
Bisdas S, Nägele T, Schlemmer HP et al (2009) Switching on the lights for real-time multimodality tumor neuroimaging: the integrated positron-emission tomography/MR imaging system. AJNR Am J Neuroradiol 31(4):610–614
Boss A, Bisdas S, Kolb A et al (2010) Hybrid PET/MRI of intracranial masses: initial experiences and comparison to PET/CT. J Nucl Med 51(8):1198–1205
Brix G, Lechel U, Glatting G et al (2005) Radiation exposure of patients undergoing whole-body dual-modality FDG-PET/CT examinations. J Nucl Med 46(4):608–613
Budinger T (1983) Time-of-flight positron emission tomography: status relative to conventional PET. J Nucl Med 24(1):73–78
Czernin J, Allen-Auerbach M, Schelbert HR (2007) Improvements in cancer staging with PET/CT: literature-based evidence as of September 2006. J Nucl Med 48(Suppl 1):78S–88S
Debus J, Wuendrich M, Pirzkall A et al (2001) High efficacy of fractionated stereotactic radiotherapy of large base-of-skull meningiomas: long-term result. J Clin Oncol 19(15):3547–3553
Fallanca F, Giovacchini G, Picchio M et al (2009) Incidental detection by [11C]choline PET/CT of meningiomas in prostate cancer patients. Q J Nucl Med Mol Imaging 53(4):417–421
Gehler B, Paulsen F, Öksüz MÖ et al (2009) [68Ga]-DOTATOC-PET/CT for meningioma IMRT treatment planning. Radiat Oncol 4:56
Grosu AL, Weber WA, Astner ST et al (2006) 11C-methionine PET improves the target volume delineation of meningiomas treated with stereotactic fractionated radiotherapy. Int J Radiat Oncol Biol Phys 66(2):339–344
Henze M, Schuhmacher J, Hipp P et al (2001) PET imaging of somatostatin receptors using [68GA]DOTA-D-Phe1-Tyr3-octreotide: first results in patients with meningiomas. J Nucl Med 42(7):1053–1056
Henze M, Dimitrakopoulou-Strauss A, Milker-Zabel S et al (2005) Characterization of 68Ga-DOTA-D-Phe1-Tyr3-octreotide kinetics in patients with meningiomas. J Nucl Med 46(5):763–769
Hofmann M, Steinke F, Scheel V et al (2008) MRI-based attenuation correction for PET/MRI: a novel approach combining pattern recognition and atlas registration. J Nucl Med 49(11):1875–1883
Hofmann M, Pichler B, Schölkopf B et al (2009) Towards quantitative PET/MRI: a review of MR-based attenuation correction techniques. Eur J Nucl Med Mol Imaging 36(Suppl 1):S93–S103
Hyun SH, Choi JY, Lee KH et al (2011) Incidental focal 18F-FDG uptake in the pituitary gland: clinical significance and differential diagnostic criteria. J Nucl Med 52(4):547–550
Kinahan PE, Hasegawa BH, Beyer T (2003) X-ray-based attenuation correction for positron emission tomography/computed tomography scanners. Semin Nucl Med 33(3):166–179
Lee JW, Kang KW, Park SH et al (2009) 18F-FDG PET in the assessment of tumor grade and prediction of tumor recurrence in intracranial meningioma. Eur J Nucl Med Mol Imaging 36(10):1574–1582
Liu RS, Chang CP, Guo WY et al (2010) 1–11C-acetate versus 18F-FDG PET in detection of meningioma and monitoring the effect of gamma-knife radiosurgery. J Nucl Med 51(6):883–891
Milker-Zabel S, Zabel-du Bois A, Henze M et al (2006) Improved target volume definition for fractionated stereotactic radiotherapy in patients with intracranial meningiomas by correlation of CT, MRI, and [68Ga]-DOTATOC-PET. Int J Radiat Oncol Biol Phys 65(1):222–227
Milker-Zabel S, Zabel-du Bois A, Huber P et al (2007) Intensity-modulated radiotherapy for complex-shaped meningioma of the skull base: long-term experience of a single institution. Int J Radiat Oncol Biol Phys 68(3):858–863
Nestle U, Weber W, Hentschel M, Grosu AL (2009) Biological imaging in radiation therapy: role of positron emission tomography. Phys Med Biol 54(1):R1–R25
Nyuyki F, Plotkin M, Graf R et al (2010) Potential impact of (68)Ga-DOTATOC PET/CT on stereotactic radiotherapy planning of meningiomas. Eur J Nucl Med Mol Imaging 37(2):310–318
Pichler B, Kolb A, Nägele T et al (2010) PET/MRI: paving the way for the next generation of clinical multimodality imaging applications. J Nucl Med 51(3):333–336
Rutten I, Cabay JE, Withofs N et al (2007) PET/CT of skull base meningiomas using 2-18F-fluoro-l-tyrosine: initial report. J Nucl Med 48(5):720–725
Schlemmer HP, Pichler BJ, Schmand M et al (2008) Simultaneous MR/PET imaging of the human brain: feasibility study. Radiology 248(3):1028–1035
Schmand M, Burbar Z, Corbeil JL et al (2007) BrainPET: first human tomograph for simultaneous (functional) PET and MR imaging. J Nucl Med 48(6):45P
Schulthess G, Burger C (2010) Integrating imaging modalities: what makes sense from a workflow perspective? Eur J Nucl Med Mol Imaging 37(5):980–990
Thorwarth D, Henke G, Müller AC et al (2011) Simultaneous [68Ga]DOTATOC PET/MR for IMRT treatment planning of meningeoma: first experience. Int J Radiat Oncol Biol Phys 81(1):277–283
Townsend DW (2008) Multimodality imaging of structure and function. Phys Med Biol 53(4):R1–R39
Warburg O (1956) On the origin of cancer cells. Science 123:306–314
Werner MK, Schwenzer NF, Thorwarth D (2011) Hybrid MR/PET imaging: principles, problems and potential for radiotherapy. Der Nuklearmediziner 34:108–113
Zaidi H (2007) Is MR-guided attenuation correction a viable option for dual-modality PET/MR imaging? Radiology 244(3):639–642
Acknowledgments
NS, SW, GH, MR, AK, BP for help with patient management, data acquisition, image analysis and data interpretation. DT is supported by the European Social Fund and the Ministry of Science, Research, and the Arts Baden-Württemberg. This work was supported by the Imaging Science Institute (ISI) Tübingen. Mirada Imaging, UK (w3.mirada-medical.com): Software fusion and image display.
Duality-of-Interest
T.B. is president and founder of Switzerland-based cmi-experts GmbH. The Imaging Science Institute of Tübingen is supported by Siemens Healthcare.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Thorwarth, D., Müller, AC., Pfannenberg, C., Beyer, T. (2013). Combined PET/MR Imaging Using 68Ga-DOTATOC for Radiotherapy Treatment Planning in Meningioma Patients. In: Baum, R., Rösch, F. (eds) Theranostics, Gallium-68, and Other Radionuclides. Recent Results in Cancer Research, vol 194. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-27994-2_23
Download citation
DOI: https://doi.org/10.1007/978-3-642-27994-2_23
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-27993-5
Online ISBN: 978-3-642-27994-2
eBook Packages: MedicineMedicine (R0)