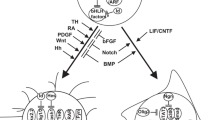
Several congenital syndromes caused by germline mutations in tumor suppressor genes predispose to the development of glial tumors. In the last few decades our knowledge about the molecular functions of these genes and the pathogenesis of hereditary tumor syndromes has greatly increased. The most common syndromes are the neu-rofibromatoses (type 1 and type 2) and the tuberous scleroses complex. There are interesting overlaps in the molecular pathogen-esis. Deregulation of Ras or downstream Ras pathways including MEK/ERK and AKT/ mTOR plays an important role in these three syndromes. Other rare syndromes include Li-Fraumeni, melanoma-astrocytoma, and Turcot syndrome involving cell cycle regulators and DNA repair genes. The genes and pathways involved in the pathogenesis of these syndromes also play an important role in the development of sporadic tumors. Therefore research on hereditary syndromes contributes substantially to our understanding of tumor formation.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Tuberous Sclerosis
- Tuberous Sclerosis Complex
- Neurofibromatosis Type
- Plexiform Neurofibroma
- Subependymal Giant Cell Astrocytoma
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Al-Saleem T, Wessner LL, Scheithauer B W, Patterson K, Roach ES, Dreyer SJ, Fujikawa K, Bjornsson J, Bernstein J, Henske EP (1998) Malignant tumors of the kidney, brain, and soft tissues in children and young adults with the tuberous sclerosis complex. Cancer 83(10):2208–2216
Antinheimo J, Haapasalo H, Haltia M, Tatagiba M, Thomas S, Brandis A, Sainio M, Carpen O, Samii M, Jaaskelainen J (1997) Proliferation potential and histological features in neurofi-bromatosis 2-associated and sporadic meningi-omas. J Neurosurg 87(4):610–614
Aravind L, Neuwald AF, Ponting CP (1999) Sec14p-like domains in NF1 and Dbl-like proteins indicate lipid regulation of Ras and Rho signaling. Curr Biol 9(6):R195–197
Azizi E, Friedman J, Pavlotsky F, Iscovich J, Bornstein A, Shafir R, Trau H, Brenner H, Nass D (1995) Familial cutaneous malignant melanoma and tumors of the nervous system. A hereditary cancer syndrome. Cancer 76(9):1571–1578
Bader JL (1986) Neurofibromatosis and cancer. Ann N Y Acad Sci 486:57–65
Bahuau M, Vidaud D, Jenkins RB, Bieche I, Kimmel DW, Assouline B, Smith JS, Alderete B, Cayuela JM, Harpey J P, Caille B, Vidaud M (1998) Germ-line deletion involving the INK4 locus in familial proneness to melanoma and nervous system tumors. Cancer Res 58(11):2298–2303
Bahuau M, Vidaud D, Kujas M, Palangie A, Assou-line B, Chaignaud-Lebreton M, Prieur M, Vidaud M, Harpey J P, Lafourcade J, Caille B (1997) Familial aggregation of malignant melanoma/dysplastic naevi and tumours of the nervous system: an original syndrome of tumour proneness. Ann Genet 40(2):78–91
Bell DW, Varley JM, Szydlo TE, Kang DH, Wahrer DC, Shannon KE, Lubratovich M, Verselis SJ, Isselbacher KJ, Fraumeni JF, Birch JM, Li F P, Garber JE, Haber DA (1999) Heterozygous germ line hCHK2 mutations in Li-Fraumeni syndrome. Science 286(5449):2528–2531
Birch JM, Heighway J, Teare MD, Kelsey AM, Hartley AL, Tricker KJ, Crowther D, Lane D P, Santibanez-Koref MF (1994) Linkage studies in a Li-Fraumeni family with increased expression of p53 protein but no germline mutation in p53. Br J Cancer 70(6):1176–1181
Blatt J, Jaffe R, Deutsch M, Adkins JC (1986) Neurofibromatosis and childhood tumors. Cancer 57(6):1225–1229
Boguski MS, McCormick F (1993) Proteins regulating Ras and its relatives. Nature 366(6456): 643–654
Bougeard G, Limacher JM, Martin C, Charbonnier F, Killian A, Delattre O, Longy M, Jonveaux P, Fricker J P, Stoppa-Lyonnet D, Flaman JM, Frebourg T (2001) Detection of 11 germline inactivating TP53 mutations and absence of TP63 and HCHK2 mutations in 17 French families with Li-Fraumeni or Li-Fraumeni-like syndrome. J Med Genet 38(4):253–257
Bourn D, Carter SA, Mason S, Gareth D, Evans R, Strachan T (1994) Germline mutations in the neurofibromatosis type 2 tumour suppressor gene. Hum Mol Genet 3(5):813–816
Boyanapalli M, Lahoud OB, Messiaen L, Kim B, Anderle de Sylor MS, Duckett SJ, Somara S, Mikol DD (2006) Neurofibromin binds to cave-olin-1 and regulates ras, FAK, and Akt. Biochem Biophys Res Commun 340(4):1200–1208
Brault E, Gautreau A, Lamarine M, Callebaut I, Thomas G, Goutebroze L (2001) Normal membrane localization and actin association of the NF2 tumor suppressor protein are dependent on folding of its N-terminal domain. J Cell Sci 114(Pt 10):1901–1912
Bretscher A, Edwards K, Fehon RG (2002) ERM proteins and merlin: integrators at the cell cortex. Nat Rev Mol Cell Biol 3(8):586–599
Brown LT, Sexsmith E, Malkin D (2000) Identification of a novel PTEN intronic deletion in Li-Fraumeni syndrome and its effect on RNA processing. Cancer Genet Cytogenet 123(1):65–68
Buday L (1999) Membrane-targeting of signalling molecules by SH2/SH3 domain-containing adaptor proteins. Biochim Biophys Acta 1422(2):187–204
Burt EC, McGown G, Thorncroft M, James LA, Birch JM, Varley JM (1999) Exclusion of the genes CDKN2 and PTEN as causative gene defects in Li-Fraumeni syndrome. Br J Cancer 80(1–2):9–10
Castle B, Baser ME, Huson SM, Cooper DN, Upadhyaya M (2003) Evaluation of genotype-phenotype correlations in neurofibromatosis type 1. J Med Genet 40(10):e109
Cawthon RM, O'Connell P, Buchberg AM, Viskochil D, Weiss RB, Culver M, Stevens J, Jenkins NA, Copeland NG, White R (1990a) Identification and characterization of transcripts from the neurofi-bromatosis 1 region: the sequence and genomic structure of EVI2 and mapping of other transcripts. Genomics 7(4):555–565
Cawthon RM, Weiss R, Xu GF, Viskochil D, Culver M, Stevens J, Robertson M, Dunn D, Gesteland R, O'Connell P, et al. (1990b) A major segment of the neurofibromatosis type 1 gene: cDNA sequence, genomic structure, and point mutations. Cell 62(1):193–201
Cawthon RM, Andersen LB, Buchberg AM, Xu GF, O'Connell P, Viskochil D, Weiss RB, Wallace MR, Marchuk DA, Culver M, et al. (1991) cDNA sequence and genomic structure of EV12B, a gene lying within an intron of the neurofibroma-tosis type 1 gene. Genomics 9(3):446–60
Chan JA, Zhang H, Roberts PS, Jozwiak S, Wieslawa G, Lewin-Kowalik J, Kotulska K, Kwiatkowski DJ (2004) Pathogenesis of tuberous sclerosis sub-ependymal giant cell astrocytomas: biallelic inacti-vation of TSC1 or TSC2 leads to mTOR activation. J Neuropathol Exp Neurol 63(12):1236–42
Cheadle J P, Reeve MP, Sampson JR, Kwiatkowski DJ (2000) Molecular genetic advances in tuberous sclerosis. Hum Genet 107(2):97–114
Cook JA, Oliver K, Mueller RF, Sampson J (1996) A cross sectional study of renal involvement in tuberous sclerosis. J Med Genet 33(6):480–484
Corradetti MN, Inoki K, Bardeesy N, DePinho RA, Guan KL (2004) Regulation of the TSC pathway by LKB1: evidence of a molecular link between tuberous sclerosis complex and Peutz-Jeghers syndrome. Genes Dev 18(13):1533–1538
Corral T, Jimenez M, Hernandez-Munoz I, Perez de Castro I, Pellicer A (2003) NF1 modulates the effects of Ras oncogenes: evidence of other NF1 function besides its GAP activity. J Cell Physiol 197(2):214–224
D'Angelo I, Welti S, Bonneau F, Scheffzek K (2006) A novel bipartite phospholipid-binding module in the neurofibromatosis type 1 protein. EMBO Rep 7(2):174–179
Dan HC, Sun M, Yang L, Feldman RI, Sui XM, Ou CC, Nellist M, Yeung RS, Halley DJ, Nicosia S V, Pledger WJ, Cheng JQ (2002) Phosphatidylinositol 3-kinase/Akt pathway regulates tuberous sclerosis tumor suppressor complex by phosphorylation of tuberin. J Biol Chem 277(38):35364–35370
Danglot G, Regnier V, Fauvet D, Vassal G, Kujas M, Bernheim A (1995) Neurofibromatosis 1 (NF1) mRNAs expressed in the central nervous system are differentially spliced in the 5′ part of the gene. Hum Mol Genet 4(5):915–920
Dasgupta B, Dugan LL, Gutmann DH The neurofi-bromatosis 1 gene product neurofibromin regulates pituitary adenylate cycase-activating polypeptide-mediated signaling in astrocytes. J Neurosci 23(26):8949–8954.
Dasgupta B, Yi Y, Chen DY, Weber JD, Gutmann DH (2005) Proteomic analysis reveals hyperactivation of the mammalian target of rapamycin pathway in neurofibromatosis 1-associated human and mouse brain tumors. Cancer Res 65(7):2755–2760
Daston MM, Scrable H, Nordlund M, Sturbaum AK, Nissen LM, Ratner N (1992) The protein product of the neurofibromatosis type 1 gene is expressed at highest abundance in neurons, Schwann cells, and oligodendrocytes. Neuron 8(3):415–428
de Andrade M, Barnholtz JS, Amos CI, Adatto P, Spencer C, Bondy ML (2001) Segregation analysis of cancer in families of glioma patients. Genet Epidemiol 20(2):258–270
DeClue JE, Cohen BD, Lowy DR (1991) Identification and characterization of the neurofibro-matosis type 1 protein product. Proc Natl Acad Sci USA 88(22):9914–9918
Dockhorn-Dworniczak B, Wolff J, Poremba C, Schafer KL, Ritter J, Gullotta F, Jurgens H, Bocker W (1996) A new germline TP53 gene mutation in a family with Li-Fraumeni syndrome. Eur J Cancer 32A(8):1359–1365
Downward J (2004) PI 3-kinase, Akt and cell survival. Semin Cell Dev Biol 15(2):177–182
Dugan LL, Kim JS, Zhang Y, Bart RD, Sun Y, Holtzman DM, Gutmann DH (1999) Differential effects of cAMP in neurons and astrocytes. Role of B-raf. J Biol Chem 274(36):25842–25848
Europen Chromosome 16 Tuberous Sclerosis Consortium (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16. The European Chromosome 16 Tuberous Sclerosis Consortium. Cell 75(7):1305–1315
Evans DG, Trueman L, Wallace A, Collins S, Strachan T (1998) Genotype/phenotype correlations in type 2 neurofibromatosis (NF2): evidence for more severe disease associated with truncating mutations. J Med Genet 35(6):450–455
Fahsold R, Hoffmeyer S, Mischung C, Gille C, Ehlers C, Kucukceylan N, Abdel-Nour M, Gewies A, Peters H, Kaufmann D, Buske A, Tinschert S, Nurnberg P (2000) Minor lesion mutational spectrum of the entire NF1 gene does not explain its high mutability but points to a functional domain upstream of the GAP-related domain. Am J Hum Genet 66(3):790–818
Fernandez-Valle C, Tang Y, Ricard J, Rodenas-Ruano A, Taylor A, Hackler E, Biggerstaff J, Iacovelli J (2002) Paxillin binds schwannomin and regulates its density-dependent localization and effect on cell morphology. Nat Genet 31(4):354–362
Frazier MW, He X, Wang J, Gu Z, Cleveland JL, Zambetti GP (1998) Activation of c-myc gene expression by tumor-derived p53 mutants requires a discrete C-terminal domain. Mol Cell Biol 18(7):3735–3743
Friedman JM (1999) Epidemiology of neurofi-bromatosis type 1. Am J Med Genet 89(1):1–6
Garami A, Zwartkruis FJ, Nobukuni T, Joaquin M, Roccio M, Stocker H, Kozma SC, Hafen E, Bos JL, Thomas G (2003) Insulin activation of Rheb, a mediator of mTOR/S6K/4E-BP signaling, is inhibited by TSC1 and 2. Mol Cell 11(6):1457–1466
Gary R, Bretscher A (1995) Ezrin self-association involves binding of an N-terminal domain to a normally masked C-terminal domain that includes the F-actin binding site. Mol Biol Cell 6(8):1061–1075
Gonzalez-Agosti C, Wiederhold T, Herndon ME, Gusella J, Ramesh V (1999) Interdomain interaction of merlin isoforms and its influence on intermolecular binding to NHE-RF. J Biol Chem 274(48):34438–34442
Goutebroze L, Brault E, Muchardt C, Camonis J, Thomas G (2000) Cloning and characterization of SCHIP-1, a novel protein interacting specifically with spliced isoforms and naturally occurring mutant NF2 proteins. Mol Cell Biol 20(5):1699–1712
Grand RJ, Owen D (1991) The biochemistry of ras p21. Biochem J 279 (Pt 3):609–631
Gronholm M, Vossebein L, Carlson CR, Kuja-Panula J, Teesalu T, Alfthan K, Vaheri A, Rauvala H, Herberg F W, Tasken K, Carpen O (2003) Merlin links to the cAMP neuronal signaling pathway by anchoring the RIbeta subunit of protein kinase A. J Biol Chem 278(42):41167–41172
Grossman SA, Osman M, Hruban R, Piantadosi S (1999) Central nervous system cancers in first-degree relatives and spouses. Cancer Invest 17(5):299–308
Guo HF, The I, Hannan F, Bernards A, Zhong Y (1997) Requirement of Drosophila NF1 for activation of adenylyl cyclase by PACAP38-like neuropeptides. Science 276(5313):795–798
Guo HF, Tong J, Hannan F, Luo L, Zhong Y (2000) A neurofibromatosis-1-regulated pathway is required for learning in Drosophila. Nature 403(6772): 895–898
Gutman DH, Andersen LB, Cole JL, Swaroop M, Collins FS (1993) An alternatively-spliced mRNA in the carboxy terminus of the neurofibromatosis type 1 (NF1) gene is expressed in muscle. Hum Mol Genet 2(7):989–992
Gutmann DH, Donahoe J, Brown T, James CD, Perry A (2000) Loss of neurofibromatosis 1 (NF1) gene expression in NF1-associated pilocytic astrocyto-mas. Neuropathol Appl Neurobiol 26(4):361–367
Gutmann DH, Wood DL, Collins FS (1991) Identification of the neurofibromatosis type 1 gene product. Proc Natl Acad Sci USA 88(21): 9658–9662
Habib AA, Gulcher JR, Hognason T, Zheng L, Stefansson K (1998) The OMgp gene, a second growth suppressor within the NF1 gene. Oncogene 16(12):1525–1531
Hamilton SR, Liu B, Parsons RE, Papadopoulos N, Jen J, Powell SM, Krush AJ, Berk T, Cohen Z, Tetu B, et al. (1995) The molecular basis of Turcot's syndrome. N Engl J Med 332(13):839–847
Han S, Santos TM, Puga A, Roy J, Thiele EA, McCollin M, Stemmer-Rachamimov A, Ramesh V (2004) Phosphorylation of tuberin as a novel mechanism for somatic inactivation of the tuberous sclerosis complex proteins in brain lesions. Cancer Res 64(3):812–816
Hannan F, Ho I, Tong JJ, Zhu Y, Nurnberg P, Zhong Y (2006) Effect of neurofibromatosis type I mutations on a novel pathway for adenylyl cyclase activation requiring neurofibromin and Ras. Hum Mol Genet 15(7):1087–1098
Huson SM, Hughes RAC (1994) The Neurofibro-matoses: a pathogenetic and clinical overview. Chapman & Hall Medical, London/New York
Inoki K, Li Y, Xu T, Guan KL (2003a) Rheb GTPase is a direct target of TSC2 GAP activity and regulates mTOR signaling. Genes Dev 17(15): 1829–1834
Inoki K, Zhu T, Guan KL (2003b) TSC2 mediates cellular energy response to control cell growth and survival. Cell 115(5):577–590
Jacks T, Shih TS, Schmitt EM, Bronson RT, Bernards A, Weinberg RA (1994) Tumour predisposition in mice heterozygous for a targeted mutation in Nf1. Nat Genet 7(3):353–361
James MF, Manchanda N, Gonzalez-Agosti C, Hartwig JH, Ramesh V (2001) The neurofi-bromatosis 2 protein product merlin selectively binds F-actin but not G-actin, and stabilizes the filaments through a lateral association. Biochem J 356(Pt 2):377–386
Jannatipour M, Dion P, Khan S, Jindal H, Fan X, Laganiere J, Chishti AH, Rouleau GA (2001) Schwannomin isoform-1 interacts with syntenin via PDZ domains. J Biol Chem 276(35): 33093–33100
Johannessen CM, Reczek EE, James MF, Brems H, Legius E, Cichowski K (2005) The NF1 tumor suppressor critically regulates TSC2 and mTOR. Proc Natl Acad Sci USA 102(24):8573–8578
Kamb A, Gruis NA, Weaver-Feldhaus J, Liu Q, Harshman K, Tavtigian S V, Stockert E, Day RS, 3rd, Johnson BE, Skolnick MH (1994a) A cell cycle regulator potentially involved in genesis of many tumor types. Science 264(5157): 436–440
Kamb A, Shattuck-Eidens D, Eeles R, Liu Q, Gruis NA, Ding W, Hussey C, Tran T, Miki Y, Weaver-Feldhaus J, et al. (1994b) Analysis of the p16 gene (CDKN2) as a candidate for the chromosome 9p melanoma susceptibility locus. Nat Genet 8(1):23–26
Kaufman DK, Kimmel DW, Parisi JE, Michels VV (1993) A familial syndrome with cutaneous malignant melanoma and cerebral astrocytoma. Neurology 43(9):1728–1731
Kaufmann D, Muller R, Kenner O, Leistner W, Hein C, Vogel W, Bartelt B (2002) The N-terminal splice product NF1–10a-2 of the NF1 gene codes for a transmembrane segment. Biochem Biophys Res Commun 294(2):496–503
Kim H, Lim JY, Kim YH, Park SH, Lee KH, Han H, Jeun SS, Lee JH, Rha HK (2002) Inhibition of ras-mediated activator protein 1 activity and cell growth by merlin. Mol Cells 14(1):108–114
Kim HA, Ratner N, Roberts TM, Stiles CD (2001) Schwann cell proliferative responses to cAMP and Nf1 are mediated by cyclin D1. J Neurosci 21(4):1110–1116
Kissil JL, Wilker E W, Johnson KC, Eckman MS, Yaffe MB, Jacks T (2003) Merlin, the product of the Nf2 tumor suppressor gene, is an inhibitor of the p21-activated kinase. Pak1. Mol Cell 12(4):841–849
Kleihues P, Schauble B, zur Hausen A, Esteve J, Ohgaki H (1997) Tumors associated with p53 germline mutations: a synopsis of 91 families. Am J Pathol 150(1):1–13
Kluwe L, Hagel C, Tatagiba M, Thomas S, Stavrou D, Ostertag H, von Deimling A, Mautner VF (2001) Loss of NF1 alleles distinguish sporadic from NF1-associated pilocytic astrocytomas. J Neuropathol Exp Neurol 60(9):917–920
Kwiatkowski DJ, Short MP (1994) Tuberous sclerosis. Arch Dermatol 130(3):348–354
Lau N, Feldkamp MM, Roncari L, Loehr AH, Shannon P, Gutmann DH, Guha A (2000) Loss of neurofibromin is associated with activation of RAS/MAPK and PI3-K/AKT signaling in a neu-rofibromatosis 1 astrocytoma. J Neuropathol Exp Neurol 59(9):759–767
Lim J Y, Kim H, Jeun SS, Kang SG, Lee KJ (2006) Merlin inhibits growth hormone-regulated Raf-ERKs pathways by binding to Grb2 protein. Biochem Biophys Res Commun 340(4):1151–1157
Lim JY, Kim H, Kim YH, Kim S W, Huh P W, Lee KH, Jeun SS, Rha HK, Kang JK (2003) Merlin suppresses the SRE-dependent transcription by inhibiting the activation of Ras-ERK pathway. Biochem Biophys Res Commun 302(2):238–245
Lopez-Correa C, Zucman-Rossi J, Brems H, Thomas G, Legius E (2000) NF2 gene deletion in a family with a mild phenotype. J Med Genet 37(1):75–77
Lucci-Cordisco E, Zito I, Gensini F, Genuardi M (2003) Hereditary nonpolyposis colorectal cancer and related conditions. Am J Med Genet A 122(4):325–334
Lynch HT, McComb RD, Osborn NK, Wolpert PA, Lynch JF, Wszolek ZK, Sidransky D, Steg RE (2000) Predominance of brain tumors in an extended Li-Fraumeni (SBLA) kindred, including a case of Sturge-Weber syndrome. Cancer 88(2):433–439
Ma L, Chen Z, Erdjument-Bromage H, Tempst P, Pandolfi PP (2005) Phosphorylation and functional inactivation of TSC2 by Erk implications for tuberous sclerosis and cancer pathogenesis. Cell 121(2):179–193
Macara IG, Lounsbury KM, Richards SA, McKie-rnan C, Bar-Sagi D (1996) The Ras superfamily of GTPases. Faseb J 10(5):625–630
Maeda M, Matsui T, Imamura M, Tsukita S (1999) Expression level, subcellular distribution and rho-GDI binding affinity of merlin in comparison with Ezrin/Radixin/Moesin proteins. Oncogene 18(34):4788–4797
Malmer B, Iselius L, Holmberg E, Collins A, Henriksson R, Gronberg H (2001) Genetic epidemiology of glioma. Br J Cancer 84(3):429–434
Manning BD, Cantley LC (2003) United at last: the tuberous sclerosis complex gene products connect the phosphoinositide 3-kinase/Akt pathway to mammalian target of rapamycin (mTOR) signalling. Biochem Soc Trans 31(Pt 3):573–578
Marshall CJ (1996) Ras effectors. Curr Opin Cell Biol 8(2):197–204
Matsui T, Maeda M, Doi Y, Yonemura S, Amano M, Kaibuchi K, Tsukita S, Tsukita S (1998) Rho-kinase phosphorylates COOH-terminal thre-onines of ezrin/radixin/moesin (ERM) proteins and regulates their head-to-tail association. J Cell Biol 140(3):647–657
McClatchey AI, Saotome I, Ramesh V, Gusella JF, Jacks T (1997) The Nf2 tumor suppressor gene product is essential for extraembryonic development immediately prior to gastrulation. Genes Dev 11(10):1253–1265
McLaughlin MR, Gollin SM, Lese CM, Albright AL (1998) Medulloblastoma and glioblastoma mul-tiforme in a patient with Turcot syndrome: a case report. Surg Neurol 49(3):295–301
Morrison H, Sherman LS, Legg J, Banine F, Isacke C, Haipek CA, Gutmann DH, Ponta H, Herrlich P (2001) The NF2 tumor suppressor gene product, merlin, mediates contact inhibition of growth through interactions with CD44. Genes Dev 15(8):968–980
Morrison H, Sperka T, Manent J, Giovannini M, Ponta H, Herrlich P (2007) Merlin/neurofi-bromatosis type 2 suppresses growth by inhibiting the activation of Ras and Rac. Cancer Res 67(2):520–527
Murthy A, Gonzalez-Agosti C, Cordero E, Pinney D, Candia C, Solomon F, Gusella J, Ramesh V (1998) NHE-RF, a regulatory cofactor for Na(+)-H + exchange, is a common interactor for merlin and
Neill GW, Crompton MR (2001) Binding of the merlin-I product of the neurofibromatosis type 2 tumour suppressor gene to a novel site in beta-fodrin is regulated by association between merlin domains. Biochem J 358(Pt 3):727–735
Nishi T, Lee PS, Oka K, Levin VA, Tanase S, Morino Y, Saya H (1991) Differential expression of two types of the neurofibromatosis type 1 (NF1) gene transcripts related to neuronal differentiation. Oncogene 6(9):1555–1559
Obremski VJ, Hall AM, Fernandez-Valle C (1998) Merlin, the neurofibromatosis type 2 gene product, and beta1 integrin associate in isolated and differentiating Schwann cells. J Neurobiol 37(4): 487–501
Oliner JD, Kinzler KW, Meltzer PS, George DL, Vogelstein B (1992) Amplification of a gene encoding a p53-associated protein in human sarcomas. Nature 358(6381):80–83
Pan D, Dong J, Zhang Y, Gao X (2004) Tuberous sclerosis complex: from Drosophila to human disease. Trends Cell Biol 14(2):78–85
Parry DM, MacCollin MM, Kaiser-Kupfer MI, Pulaski K, Nicholson HS, Bolesta M, Eldridge R, Gusella JF (1996) Germ-line mutations in the neurofibromatosis 2 gene: correlations with disease severity and retinal abnormalities. Am J Hum Genet 59(3):529–539
Paunu N, Syrjakoski K, Sankila R, Simola KO, Helen P, Niemela M, Matikainen M, Isola J, Haapasalo H (2001) Analysis of p53 tumor suppressor gene in families with multiple glioma patients. J Neurooncol 55(3):159–165
Petronzelli F, Sollima D, Coppola G, Martini-Neri ME, Neri G, Genuardi M (2001) CDKN2A germ-line splicing mutation affecting both p16(ink4) and p14(arf) RNA processing in a melanoma/ neurofibroma kindred. Genes Chromosomes Cancer 31(4):398–401
Platten M, Giordano MJ, Dirven CM, Gutmann DH, Louis DN (1996) Up-regulation of specific NF 1 gene transcripts in sporadic pilocytic astrocyto-mas. Am J Pathol 149(2):621–627
Portwine C, Lees J, Verselis S, Li F P, Malkin D (2000) Absence of germline p16(INK4a) alterations in p53 wild type Li-Fraumeni syndrome families. J Med Genet 37(8):E13
Prowse AH, Schultz DC, Guo S, Vanderveer L, Dangel J, Bove B, Cairns P, Daly M, Godwin AK (2003) Identification of a splice acceptor site mutation in p16INK4A/p14ARF within a breast cancer, melanoma, neurofibroma prone kindred. J Med Genet 40(8):e102
Randerson-Moor JA, Harland M, Williams S, Cuthbert-Heavens D, Sheridan E, Aveyard J, Sibley K, Whitaker L, Knowles M, Bishop JN, Bishop DT (2001) A germline deletion of p14(ARF) but not CDKN2A in a melanoma-neural system tumour syndrome family. Hum Mol Genet 10(1):55–62
Rasmussen SA, Yang Q, Friedman JM (2001) Mortality in neurofibromatosis 1: an analysis using U.S. death certificates. Am J Hum Genet 68(5):1110–1118
Raught B, Peiretti F, Gingras AC, Livingstone M, Shahbazian D, Mayeur GL, Polakiewicz RD, Sonenberg N, Hershey JW (2004) Phosphorylation of eucaryotic translation initiation factor 4B Ser422 is modulated by S6 kinases. Embo J 23(8):1761–1769
Richardson CJ, Broenstrup M, Fingar DC, Julich K, Ballif BA, Gygi S, Blenis J (2004) SKAR is a specific target of S6 kinase 1 in cell growth control. Curr Biol 14(17):1540–1549
Roach ES, Gomez MR, Northrup H (1998) Tuberous sclerosis complex consensus conference: revised clinical diagnostic criteria. J Child Neurol 13(12):624–628
Rong R, Tang X, Gutmann DH, Ye K (2004) Neurofibromatosis 2 (NF2) tumor suppressor merlin inhibits phosphatidylinositol 3-kinase through binding to PIKE-L. Proc Natl Acad Sci USA 101(52):18200–18205
Rouleau GA, Merel P, Lutchman M, Sanson M, Zucman J, Marineau C, Hoang-Xuan K, Demczuk S, Desmaze C, Plougastel B, et al. (1993) Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature 363(6429):515–521
Roussel MF (1999) The INK4 family of cell cycle inhibitors in cancer. Oncogene 18(38): 5311–5317
Roux P P, Ballif BA, Anjum R, Gygi S P, Blenis J (2004) Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase. Proc Natl Acad Sci USA 101(37): 13489–13494
Ruttledge MH, Andermann AA, Phelan CM, Claudio JO, Han FY, Chretien N, Rangaratnam S, MacCollin M, Short P, Parry D, Michels V, Riccardi VM, Weksberg R, Kitamura K, Bradburn JM, Hall BD, Propping P, Rouleau GA (1996) Type of mutation in the neurofibromatosis type 2 gene (NF2) frequently determines severity of disease. Am J Hum Genet 59(2):331–342
Ryu CH, Kim SW, Lee KH, Lee JY, Kim H, Lee WK, Choi BH, Lim Y, Kim YH, Hwang TK, Jun TY, Rha HK (2005) The merlin tumor suppressor interacts with Ral guanine nucleotide dissociation stimulator and inhibits its activity. Oncogene 24(34):5355–5364
Sainio M, Zhao F, Heiska L, Turunen O, den Bakker M, Zwarthoff E, Lutchman M, Rouleau GA, Jaas-kelainen J, Vaheri A, Carpen O (1997) Neuro-fibromatosis 2 tumor suppressor protein colocalizes with ezrin and CD44 and associates with actin-containing cytoskeleton. J Cell Sci 110 (Pt 18):2249–2260
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/ PKB by the rictor-mTOR complex. Science 307(5712):1098–1101
Scoles DR, Huynh D P, Chen MS, Burke S P, Gutmann DH, Pulst SM (2000) The neurofibromatosis 2 tumor suppressor protein interacts with hepatocyte growth factor-regulated tyrosine kinase substrate. Hum Mol Genet 9(11):1567–1574
Scoles DR, Huynh D P, Morcos PA, Coulsell ER, Robinson NG, Tamanoi F, Pulst SM (1998) Neurofibromatosis 2 tumour suppressor schwan-nomin interacts with betaII-spectrin. Nat Genet 18(4):354–359
Scoles DR, Yong WH, Qin Y, Wawrowsky K, Pulst SM (2006) Schwannomin inhibits tumorigenesis through direct interaction with the eukaryotic initiation factor subunit c (eIF3c). Hum Mol Genet 15(7):1059–1070
Sharma MK, Mansur DB, Reifenberger G, Perry A, Leonard JR, Aldape KD, Albin MG, Emnett RJ, Loeser S, Watson MA, Nagarajan R, Gutmann DH (2007) Distinct genetic signatures among pilocytic astrocytomas relate to their brain region origin. Cancer Res 67(3):890–900
Sharma MK, Zehnbauer BA, Watson MA, Gutmann DH (2005) RAS pathway activation and an oncogenic RAS mutation in sporadic pilocytic astrocytoma. Neurology 65(8):1335–1336
Shaw RJ, Bardeesy N, Manning BD, Lopez L, Kosmatka M, DePinho RA, Cantley LC (2004) The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell 6(1):91–99
Shen MH, Harper PS, Upadhyaya M (1996) Molecular genetics of neurofibromatosis type 1 (NF1). J Med Genet 33(1):2–17
Sherman L, Xu HM, Geist RT, Saporito-Irwin S, Howells N, Ponta H, Herrlich P, Gutmann DH (1997) Interdomain binding mediates tumor growth suppression by the NF2 gene product. Oncogene 15(20):2505–2509
Sigal A, Rotter V (2000) Oncogenic mutations of the p53 tumor suppressor: the demons of the guardian of the genome. Cancer Res 60(24):6788–6793
Stone JG, Eeles RA, Sodha N, Murday V, Sheriden E, Houlston RS (1999) Analysis of Li-Fraumeni syndrome and Li-Fraumeni-like families for germ-line mutations in Bcl10. Cancer Lett 147(1–2): 181–185
Stone S, Jiang P, Dayananth P, Tavtigian S V, Katcher H, Parry D, Peters G, Kamb A (1995) Complex structure and regulation of the P16 (MTS1) locus. Cancer Res 55(14):2988–2994
Tachibana I, Smith JS, Sato K, Hosek SM, Kimmel DW, Jenkins RB (2000) Investigation of germ-line PTEN, p53, p16(INK4A)/p14(ARF), and CDK4 alterations in familial glioma. Am J Med Genet 92(2):136–141
Tang X, Jang SW, Wang X, Liu Z, Bahr SM, Sun SY, Brat D, Gutmann DH, Ye K (2007) Akt phospho-rylation regulates the tumour-suppressor merlin through ubiquitination and degradation. Nat Cell Biol 9(10):1199–1207
The I, Hannigan GE, Cowley GS, Reginald S, Zhong Y, Gusella J F, Hariharan IK, Bernards A (1997) Rescue of a Drosophila NF1 mutant phenotype by protein kinase A. Science 276(5313):791–794
Tikoo A, Varga M, Ramesh V, Gusella J, Maruta H (1994) An anti-Ras function of neurofibromato-sis type 2 gene product (NF2/Merlin). J Biol Chem 269(38):23387–23390
Tong J, Hannan F, Zhu Y, Bernards A, Zhong Y (2002) Neurofibromin regulates G protein-stimulated adenylyl cyclase activity. Nat Neurosci 5(2):95–96
Trofatter JA, MacCollin MM, Rutter JL, Murrell JR, Duyao M P, Parry DM, Eldridge R, Kley N, Menon AG, Pulaski K, et al. (1993) A novel moesin-, ezrin-, radixin-like gene is a candidate for the neurofibromatosis 2 tumor suppressor. Cell 72(5):791–800
Upadhyaya M, Huson SM, Davies M, Thomas N, Chuzhanova N, Giovannini S, Evans DG, Howard E, Kerr B, Griffiths S, Consoli C, Side L, Adams D, Pierpont M, Hachen R, Barnicoat A, Li H, Wallace P, Van Biervliet J P, Stevenson D, Viskochil D, Baralle D, Haan E, Riccardi V, Turnpenny P, Lazaro C, Messiaen L (2007) An absence of cutaneous neurofibromas associated with a 3-bp inframe deletion in exon 17 of the NF1 gene (c.2970–2972 delAAT): evidence of a clinically significant NF1 genotype-phenotype correlation. Am J Hum Genet 80(1):140–151
van Slegtenhorst M, de Hoogt R, Hermans C, Nellist M, Janssen B, Verhoef S, Lindhout D, van den Ouweland A, Halley D, Young J, Burley M, Jeremiah S, Woodward K, Nahmias J, Fox M, Ekong R, Osborne J, Wolfe J, Povey S, Snell RG, Cheadle J P, Jones AC, Tachataki M, Ravine D, Sampson JR, Reeve M P, Richardson P, Wilmer F, Munro C, Hawkins TL, Sepp T, Ali JB, Ward S, Green AJ, Yates JR, Kwiatkowska J, Henske E P, Short M P, Haines JH, Jozwiak S, Kwiatkowski DJ (1997) Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 277(5327):805–808
Vandenbroucke I, Van Oostveldt P, Coene E, De Paepe A, Messiaen L (2004) Neurofibromin is actively transported to the nucleus. FEBS Lett 560(1–3):98–102
Vanhaesebroeck B, Alessi DR (2000) The PI3K-PDK1 connection: more than just a road to PKB. Biochem J 346 Pt 3:561–576
Varley J (2003) TP53, hChk2, and the Li-Fraumeni syndrome. Methods Mol Biol 222:117–129
Vikhanskaya F, Lee MK, Mazzoletti M, Broggini M, Sabapathy K (2007) Cancer-derived p53 mutants suppress p53-target gene expression–potential mechanism for gain of function of mutant p53. Nucleic Acids Res 35(6):2093–2104
Viskochil D, Buchberg AM, Xu G, Cawthon RM, Stevens J, Wolff RK, Culver M, Carey JC, Copeland NG, Jenkins NA, et al. (1990) Deletions and a translocation interrupt a cloned gene at the neuro-fibromatosis type 1 locus. Cell 62(1):187–192
Viskochil D, Cawthon R, O'Connell P, Xu G F, Stevens J, Culver M, Carey J, White R (1991) The gene encoding the oligodendrocyte-myelin glycoprotein is embedded within the neurofibromatosis type 1 gene. Mol Cell Biol 11(2):906–912
von Deimling A, Louis DN, Menon AG, von Ammon K, Petersen I, Ellison D, Wiestler OD, Seizinger BR (1993) Deletions on the long arm of chromosome 17 in pilocytic astrocytoma. Acta Neuropathol (Berl) 86(1):81–85
Vousden KH, Lu X (2002) Live or let die: the cell's response to p53. Nat Rev Cancer 2(8): 594–604
Weber JD, Taylor LJ, Roussel M F, Sherr CJ, Bar-Sagi D (1999) Nucleolar Arf sequesters Mdm2 and activates p53. Nat Cell Biol 1(1):20–26
Welti S, Fraterman S, D'Angelo I, Wilm M, Scheffzek K (2007) The sec14 homology module of neurofibromin binds cellular glycerophos-pholipids: mass spectrometry and structure of a lipid complex. J Mol Biol 366(2):551–562
Wiederhold T, Lee M F, James M, Neujahr R, Smith N, Murthy A, Hartwig J, Gusella J F, Ramesh V (2004) Magicin, a novel cytoskeletal protein associates with the NF2 tumor suppressor merlin and Grb2. Oncogene 23(54):8815–8825
Wimmer K, Eckart M, Meyer-Puttlitz B, Fonatsch C, Pietsch T (2002) Mutational and expression analysis of the NF1 gene argues against a role as tumor suppressor in sporadic pilocytic astrocytomas. J Neuropathol Exp Neurol 61(10):896–902
Winston LA, Hunter T (1995) JAK2, Ras, and Raf are required for activation of extracellular signal-regulated kinase/mitogen-activated protein kinase by growth hormone. J Biol Chem 270(52): 30837–30840
Xiao ZX, Chen J, Levine AJ, Modjtahedi N, Xing J, Sellers WR, Livingston DM (1995) Interaction between the retinoblastoma protein and the onco-protein MDM2. Nature 375(6533):694–698
Xu HM, Gutmann DH (1998) Merlin differentially associates with the microtubule and actin cytoskeleton. J Neurosci Res 51(3):403–415
Zhang Y, Gao X, Saucedo LJ, Ru B, Edgar BA, Pan D (2003) Rheb is a direct target of the tuberous sclerosis tumour suppressor proteins. Nat Cell Biol 5(6):578–581.
Zhu Y, Guignard F, Zhao D, Liu L, Burns DK, Mason R P, Messing A, Parada LF (2005a) Early inacti-vation of p53 tumor suppressor gene cooperating with NF1 loss induces malignant astrocytoma. Cancer Cell 8(2):119–130
Zhu Y, Harada T, Liu L, Lush ME, Guignard F, Harada C, Burns DK, Bajenaru ML, Gutmann DH, Parada LF (2005b) Inactivation of NF1 in CNS causes increased glial progenitor proliferation and optic glioma formation. Development 132(24):5577–5588
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Reuss, D., von Deimling, A. (2009). Hereditary Tumor Syndromes and Gliomas. In: von Deimling, A. (eds) Gliomas. Recent Results in Cancer Research, vol 171. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-31206-2_5
Download citation
DOI: https://doi.org/10.1007/978-3-540-31206-2_5
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-31205-5
Online ISBN: 978-3-540-31206-2
eBook Packages: MedicineMedicine (R0)