Abstract
We explored the potential protective effects of tauroursodeoxycholic acid (TUDCA) on cone photoreceptor survival in a model of rapid retinal degeneration, the ß-Pde6rd1 (rd1) mouse model. We injected two strains of rd1 mice (B6.C3-Pde6brd1Hps4le/J and C57BL/6J-Pde6brd1−2/J mice) daily from postnatal day (P) 6 to P21 with TUDCA or vehicle. At P21, retinal function was evaluated with light-adapted electroretinography (ERG) and retinal structure was observed with plastic or frozen sections. TUDCA treatment partially preserved function and structure in B6.C3-Pde6brd1Hps4le/J mice but only partially preserved structure in C57BL/6J-Pde6brd1−2/J mice. Our results suggest a possible intervention for patients undergoing rapid retinal degeneration.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Tauroursodeoxycholic acid (TUDCA) is neuroprotective in several rodent models of neurodegeneration (reviewed in Boatright et al. 2009) and retinal degeneration (Boatright et al. 2006; Phillips et al. 2008) . In this study we explored the possible neuroprotective effects of daily injections of TUDCA on two strains of rd1 mice. The rd1 mouse , considered a model of retinitis pigmentosa, has a nonsense mutation in the ß-subunit of the rod cGMP phosphodiesterase, resulting in loss of rod photoreceptors beginning at postnatal day P10 and finishing by about P21 (Sancho-Pelluz et al. 2008) . Despite the rapid degeneration of rod photoreceptors , cone photoreceptors degenerate at a slower rate, providing potential therapeutic opportunities. In this study, we sought to examine whether TUDCA could preserve cone function. We were able to replicate that no functional protection was observed in C57BL-rd1 mice with TUDCA (Drack et al. 2012) , even though we found partial structural preservation. More importantly, we show functional and structural protection with daily injections of TUDCA from P6 to P21 in B6.C3-rd1 mice.
2 Material and Methods
2.1 Animals
All animal procedures were approved by the Institutional Animal Care and Use Committee at the Atlanta VA Medical Center and conform to the standards of the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. Two strains of ß-Pde6rd1 mice were obtained from Jackson Laboratories (Bar Harbor, ME): B6.C3-Pde6brd1Hps4le/J mice (B6.C3-rd1; Stock #: 000002) and C57BL/6J-Pde6brd1−2/J mice (C57BL-rd1; Stock #: 004766). All mice were housed under controlled lighting conditions on a 12 h light/12 h dark cycle.
2.2 TUDCA Treatments
rd1 litters were randomly divided at P6 to receive TUDCA (500 mg/kg, Calbiochem, San Diego, CA) or vehicle (0.15 M NaHCO3 1 ml/kg) treatment. TUDCA solution was made fresh daily and pH was adjusted to 7.4 using 0.1 M HCl. Daily intraperitoneal injections began at P6 as it has been previously shown that injections every 3 days have no protective effects on the rd1 retina, most likely due to the increased degeneration rate compared to other models (Boatright et al. 2009) . Treatments ended at P21 for each animal. Mice were weighed daily prior to injection to determine proper dosing of TUDCA and vehicle.
2.3 Electroretinography
Electroretinography was performed at P21, as previously detailed (Mocko et al. 2011) . Briefly, mice were anesthetized with ketamine (80 mg/kg) and xylazine (16 mg/kg), the cornea was anesthetized (1 % tetracaine), and the pupils dilated (1 % tropicamide) in both eyes. The body temperature was maintained at 37 °C via a heating pad while the recording electrode, a nylon-silver thread, contacted the cornea using 1 % methylcellulose. The responses were referenced and grounded to needle electrodes placed in the cheek and tail, respectively. A series of full-field flash stimuli (− 0.82 to 1.9 log cd-s/m2) were presented by a Ganzfeld dome under light-adapted conditions (30 cd/m2) to isolate cone responses. Acquired responses were stored on a commercial ERG system (UTAS 3000, LKC Technologies, Gaithersburg, MD).
2.4 Histology
Retinal morphology was assessed as previously reported (Mocko et al. 2011) . Briefly, mice were euthanized and eyes enucleated, injected with 4 % paraformaldehyde, and immersion fixed in the same fixative for 45 min. Eyecups of B6.C3-rd1 mice were rinsed with 0.1 M phosphate buffer, processed through a graded alcohol series, and embedded in plastic resin (Embed 812/DER 736, Electron Microscopy Science, Inc, Hatfield, PA). Sections (0.5 µm) bisecting the optic disc at the superior-inferior axis were cut using an ultramicrotome (Reichert Ultracut, Leica Inc., Buffalo Grove, IL) with a histo-diamond knife. Eyecups of C57BL-rd1 mice were frozen in OCT and cryosectioned (10 µm thickness) onto glass slides. Both plastic and cryosections sections were stained with 1 % aqueous toluidine blue (Sigma; St. Louis MO) and imaged using a phase contrast microscope (Leica DM LB, Leica Inc., Buffalo Grove, IL) at 20 × power. Photoreceptor nuclei cells were counted using an image analysis program (Image-Pro Plus 5.0; MediaCybernetics; Rockville, MD). For each retinal section, we quantified photoreceptor nuclei across the retina moving outwards superiorly and inferiorly from the optic nerve. The number of photoreceptor nuclei were averaged across three retinal sections for each eye.
2.5 Statistical Analyses
We performed two-way repeated measures ANOVAs with Holm-Sidak post-hoc comparisons and Student’s t-tests using commercial statistical analysis software (SigmaStat 3.5; Systat Software; Chicago, IL). Significance was set at p < 0.05 for all analyses and values are expressed as mean ± standard error of the mean (sem).
3 Results
3.1 TUDCA Injections Preserved Retinal Function to P21 in B6.C3-rd1 Mice
Light-adapted ERG waveforms from representative mice in each group showed larger amplitudes with TUDCA treatment for B6.C3-rd1 mice compared to B6.C3-rd1 vehicle treated (Fig. 57.1a). Within the B6.C3-rd1 mice, those injected with TUDCA had significantly preserved light adapted b-wave amplitudes compared to vehicle treated mice at the brightest flash stimuli (Fig. 57.1b; 0.4, 0.9, 1.4, and 1.9 log cd s/m2; two-way repeated measures ANOVA; F(1, 41) = 16.986, p = 0.005). C57BL-rd1 mice exhibited no measurable a-or b-waveforms at P21, regardless of treatment.
TUDCA protects cone photoreceptor function in B6.C3-rd1 mice at P21. a Representative light-adapted ERG waveforms from B6.C3-rd1 mice across flash stimuli (− 0.81 to 1.9 log cd s/m2). b B6.C3-rd1 TUDCA-treated mice have significantly larger b-wave amplitudes responses at the brightest flash stimuli compared to vehicle-treated mice (two-way repeated measures ANOVA; F(1, 41) = 16.986, p = 0.005). C57BL-rd1 mice did not exhibit measurable b-wave responses at any flash stimulus
3.2 TUDCA Injections Preserved Photoreceptor Cell Counts in Both rd1 Strains
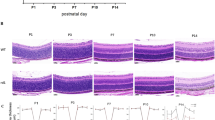
After TUDCA injections, the retinas of B6.C3-rd1 and C57BL-rd1 mice maintained a thicker outer nuclear layer (ONL) of about 2 rows of photoreceptor nuclei (Fig. 57.2b and 57.3b) compared to vehicle treated mice, which degenerated to a sparse single row of photoreceptor nuclei (Fig. 57.2a and 57.3a). The summed photoreceptor nuclei across the retina in TUDCA-treated B6.C3-rd1 mice was significantly greater compared to vehicle-treated littermates (Student’s t-test; p = 0.005; Fig. 57.2c). C57BL-rd1 mice also showed significantly more photoreceptor nuclei with TUDCA treatment (Student’s t-test; p = 0.038; Fig. 57.3c).
TUDCA protects cone photoreceptor structure in B6.C3-rd1 mice. Retinal micrographs of plastic sections taken 1.0 mm from the optic nerve from B6.C3-rd1 mice shows that mice injected with TUDCA had a thicker ONL (b) compared to those injected with vehicle (a). c TUDCA-treated B6.C3-rd1 mice had significantly more photoreceptor nuclei compared to vehicle-treated mice (Student’s t-test; p = 0.005)
TUDCA protects cone photoreceptor structure in C57BL-rd1 mice. Retinal micrographs of cryosections taken 1.0 mm from the optic nerve in C57BL-rd1 mice injected with TUDCA had a thicker ONL (b) compared to those injected with vehicle (a). c TUDCA-treated C57BL-rd1 mice had significantly more photoreceptor nuclei compared to vehicle-treated mice (Student’s t-test; p = 0.038)
4 Discussion
Here we demonstrate that significant cone protection in rd1 mice is possible with TUDCA injections. Daily injections of TUDCA were sufficient to protect both retinal function (specifically light-adapted ERGs) and structure in B6.C3-rd1 mice. However, in C57BL-rd1 mice, photoreceptor nuclei were preserved, but not retinal function. A previous report also observed no functional preservation in C57BL-rd1 mice (Dr. Val Sheffield, personal communication) with TUDCA injections at P21 (Drack et al. 2012) , however, structural preservation was not explored. The differences in efficacy of TUDCA between the two strains of rd1 mice may be due to the different rates of degeneration. It is possible that C57BL-rd1 have a more aggressive degeneration compared to B6.C3-rd1 mice , as C57BL-rd1 mice have no measurable ERG response at any age (Chang et al. 2007) , while B6.C3-rd1 mice treated with vehicle still have residual ERG responses at P21 (Fig. 57.1). Nonetheless, our findings illustrate the protective effects of TUDCA on cone photoreceptors in a model of rapid retinal degeneration, and suggest a possible intervention for aggressive forms of retinitis pigmentosa.
References
Boatright JH, Moring AG, McElroy C et al (2006) Tool from ancient pharmacopoeia prevents vision loss. Mol Vis 12:1706–1714
Boatright JH, Nickerson JM, Moring AG et al (2009) Bile acids in treatment of ocular disease. J Ocul Biol Dis Inform 2:149–159
Chang B, Hawes NL, Pardue MT et al (2007) Two mouse retinal degenerations caused by missense mutations in the beta-subunit of rod cGMP phosphodiesterase gene. Vis Res 47:624–633
Drack AV, Dumitrescu AV, Bhattarai S et al (2012) TUDCA slows retinal degeneration in two different mouse models of retinitis pigmentosa and prevents obesity in Bardet-Biedl syndrome type 1 mice. Invest Ophthalmol Vis Sci 53:100–106
Mocko JA, Kim M, Faulkner AE et al (2011) Effects of subretinal electrical stimulation in mer-KO mice. Invest Ophthalmol Vis Sci 52:4223–4230
Phillips MJ, Walker TA, Choi HY et al (2008) Tauroursodeoxycholic acid preservation of photoreceptor structure and function in the rd10 mouse through postnatal day 30. Invest Ophthalmol Vis Sci 49:2148–2155
Sancho-Pelluz J, Arango-Gonzalez B, Kustermann S et al (2008) Photoreceptor cell death mechanisms in inherited retinal degeneration. Mol Neurobiol 38:253–269
Acknowledgments
Grant support provided by NIH P30 EY006360, NIH R01 EY014026 and a grant from the Abraham J. & Phyllis Katz Foundation (to J.H.B.), Rehabilitation Research and Development Service Veterans Affairs Research Career Scientist Award (to M.T.P.), Atlanta VA Center of Excellence in Vision and Neurocognitive Rehabilitation, and Departmental Award from Research to Prevent Blindness.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Lawson, E. et al. (2016). Tauroursodeoxycholic Acid Protects Retinal Function and Structure in rd1 Mice. In: Bowes Rickman, C., LaVail, M., Anderson, R., Grimm, C., Hollyfield, J., Ash, J. (eds) Retinal Degenerative Diseases. Advances in Experimental Medicine and Biology, vol 854. Springer, Cham. https://doi.org/10.1007/978-3-319-17121-0_57
Download citation
DOI: https://doi.org/10.1007/978-3-319-17121-0_57
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-17120-3
Online ISBN: 978-3-319-17121-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)