Abstract
Low-temperature stress is considered as the major abiotic constraint limiting plant’s growth and the potential land cultivation. Crop adaptation to limiting temperature is thus an important breeding objective because it determines yield stability in environment-friendly cultivation practices. Conventional breeding methods had limited success in improving the cold tolerance of important crop plants because of the complexity of stress tolerance traits, low genetic variance, and lack of efficient selection criteria. The knowledge of physiology, of genetics, and of the DNA technology has improved substantially nowadays, and these advancements will allow the breeder to predict the breeding value of best genotypes by using physiology, genetics, and molecular information. The perspective for selecting more effectively cold-tolerant crops will involve efficient genotyping, reliable phenotyping and envirotyping, and adequate statistical models.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
9.1 Introduction
Low-temperature stress is considered as the major abiotic constraint limiting plants’ growth and the exploitation of the potential land cultivation (Jha et al. 2017). Crops evolved, through domestication, generally from warm areas of the planet (Feldman and Levy 2009), thanks to acclimation and adaptation mechanisms that made it possible for them to survive at higher latitudes. As adaptation to cold was undertaken by most of the crop plants, the traits that allow a satisfactory yield are shared among different species and present functional conservation among them. These common tolerance traits can be the effect of convergent evolution via independent paths to a similar outcome or of monophyletic origin if they all descend from a common ancestor (Mickelbart et al. 2015). Indeed, several specific adaptations are involved in the maximal exploitation of yield potential of a crop (Huner et al. 2014). Among them, we name the modulation of the amount of energy that can reach the plant, the light interception efficiency, the energy conversion efficiency, and the partitioning efficiency of products into commercial yield. All those steps can be limited by less than optimal temperature. In addition, to adapt to new, cooler environments, plants need to modify a number of traits, like photoperiod sensitivity, as in case of wheat (Guo et al. 2018), maize, and potato, or vernalization, e.g., for wheat, barley, and carrots (Mickelbart et al. 2015).
Crop adaptation to limiting low temperature still is an important breeding objective, as observed by Bradshaw (2017) who also noticed that “breeders still need to apply appropriate breeding methods to the right germplasm for the right objectives”. The objective of breeding is determined by the characteristics of the new and more efficient farming systems and of the innovative uses of crops, in turn devised to face the increased need of food and energy expected in the near future. When environmental conditions deviate from the optimal temperature, growth is limited, and there is risk of tissue damage or even of impaired survival ability (Korner 2016). In general, conditions with temperatures lower than optimum can be found at high elevation or at high latitude, but there are cold periods in temperate zones, too. Genotypes able to survive in extreme environments, like in case of alpine trees (Korner et al. 2016), are able to actively grow during the period with acceptable temperature. Plants can adapt to temperatures that become low only seasonally by means of modification of phenology and morphology. In higher latitudes, plants can basically develop only by using the warmest periods, and thus they need to grow faster to take advantage of the otherwise short season. Morphological modifications concern usually plant size, in that smaller plants are generally less susceptible than taller ones.
Abiotic stresses impose negative deviations from the maximum yield potential, and thus they are important factors determining crop yield stability (Mickelbart et al. 2015). Traditionally, increased yield stability has been attained through selective breeding, and selection has been successful in changing allelic frequencies of favorable alleles, although taking a long time to accomplish the change. However, since abiotic stresses, including cold stress, are somehow unpredictable in the field, sometimes it is more advisable to select genotypes with high potential per se and wide adaptability and stability, instead of trying to develop varieties designed for a narrow agroecological region and a specific stress (Arief et al. 2015; Bennett et al. 2012; Stojakovic et al. 2015).
In recent decades, crops had to face changing environmental conditions, with a general increase in the mean temperature and especially a higher temperature fluctuation (Xu et al. 2018). Even though, at first glance, the global warming might seem to reduce frost damage in crops (Cattivelli 2011), the fluctuation of winter temperatures, with warm periods, has a strong impact on the frost tolerance both in crops and natural plant species. In fact, warm days during the winter can strongly interfere with dormancy and vernalization, so plants start an active growth and lose most of their freezing tolerance. In addition, with warmer winters, temperate tree species tend to show earlier flowering, with increased risk of damage during the spring frost (Eccel et al. 2009). Modification in the cultivation practices to cope with new environmental conditions have already been observed, with significant changes in tillage techniques and in the timing of cultivation toward earlier sowing, to avoid dry periods during the summer and to use as much as possible of the winter and spring precipitations (Olesen et al. 2011, 2012; Trnka et al. 2011). Indeed, temperature changes will trigger the need for optimization of the crop growth phases, to minimize the stress and to maximize the productivity of the agronomic resources employed. New tillage practices are focused on soil–water conservation and protection against soil erosion, as these issues are believed to become increasingly important (Falloon and Betts 2010). These practices, however, leave the soil colder than the traditional ones for the sowing of spring crops, and thus genotypes tolerant to low temperature at germination will better suit the new practices. As for the timing of cultivation, a study regarding the major trends in Europe (Olesen et al. 2012) pointed out that “climate change will affect the timing of cereal crop development, but exact changes will also depend on changes in varieties as affected by plant breeding and variety choices”. The modification of the timing of crop growth and of the agronomic practices can be made possible through plant breeding by selecting for different traits, such as the response to photoperiod or the response to temperature critical for each growth phase. Genotypes selected for tolerance to suboptimal temperature are also particularly useful when cultivation is addressed to the low input agriculture or for high elevated lands (Sthapit and Witcombe 1998). This is the case of both the low input climate-smart agriculture and the highly efficient sustainable intensification (Campbell et al. 2014; Garnett et al. 2013; Nemali et al. 2015; Thakur and Uphoff 2017). Agronomic models have been developed to describe the growth phases of major crops under different climate conditions (Waha et al. 2012) and for the characterization of breeding needs in more adapted varieties through the identification of an appropriate ideotype (Olesen et al. 2012; Semenov et al. 2014; Semenov 2009).
This chapter will review (1) how cold tolerance can be the key of the crop adaptation to low input and highly efficient agricultural techniques and (2) how plant breeding can integrate classical and molecular methods to select genotypes maximizing crop performance in cold environments.
9.2 Cold Tolerance in Adaptation to Environment
Selection of alleles for adaptation to favorable and unfavorable environments already led to a general improvement of crops yield. This achievement was accomplished through effective stress adaptation, namely, to low and subfreezing temperatures, high temperature, flooding, drought, salinity, ion toxicity, ion deficiency, and ozone (Mickelbart et al. 2015). Adaptation of plants to abiotic stresses, characterizing diverse and variable environments, involves a variety of plasticity mechanisms and provides plants with increased yield stability. In case the suboptimal condition occurs when the plant is growing slowly, the effect may be not as great as if it occurs during plant fast growing (Dolferus 2014; Mansouri-Far et al. 2010). Moreover, stresses often occur in combinations (i.e., high temperature and drought, low temperature and flooding), so yield adaptation usually implies enhancement of multiple-stress tolerances. Oftentimes, constraints imposed by one stress are the same as another. With reference to cold, it can involve membrane damage as in case of high temperature (Bita and Gerats 2013; Dhillon et al. 2010), so the maintenance of membrane function is crucial to achieve the tolerance to both. Accordingly, a large overlapping among adaptive responses to cold and drought have been reported (Hussain et al. 2018). As an example, maintenance of water potential is a factor enhancing tolerance to cold, and such a mechanism is shared with other stresses such as drought and salinity. The response to all those environmental factors has been studied and proved to involve cross talk between various stress signaling metabolic pathways (Cramer et al. 2011; Nakashima et al. 2014; Seki et al. 2003). Tolerance or susceptibility to these stresses are complex traits, as stress may affect multiple stages of plant development, and often several stresses concurrently affect the plants (Chinnusamy et al. 2004; Li et al. 2014). Moreover, it has been reported in a number of studies that the same genes can be responsible for reaction to multiple stresses (Bai et al. 2018; Banerjee and Roychoudhury 2018; Dubois et al. 2018; Hossain et al. 2018; Mustafavi et al. 2018).
9.2.1 Cold Limits Crop Growth in Less than Optimal Environments
The ability of crops to grow in different environment/climates is one of more powerful tools enabling farmer to satisfy the growing demand of food and the changing needs of industrial crops. Plant breeding is a key process in the achievement of plants adapted to different climates (Atlin et al. 2017). Breeding for resilient crops, therefore, is one of the major strategies to cope with the increasing challenges in world agriculture (Obata et al. 2015). It is important to note that abiotic stresses inherent with the environment adversely interfere with growth and with agronomic performance through modifications regarding morphological, physiological, and molecular adjustments (Sanghera et al. 2011). Different environmental constraint exacerbates the need of cropping systems continually updated. As already noted, although it may seem counterintuitive, even the effects of increase of temperature can be overcome by means of cold tolerance. In fact, one way to cope with high temperatures and shortage of water is either to change the sowing date or to move to higher latitudes. Moreover, the rise of temperatures can affect cold acclimation and thus even impair plant overwintering survival (Arora 2018; Rapacz et al. 2014). Widespread efforts to identify major genes controlling tolerance to stress lead to the identification of some alleles with large effects for tolerance (Thoen et al. 2017). On the contrary, traits controlling stress responses are more often controlled by many genes with small effects (Haak et al. 2017).
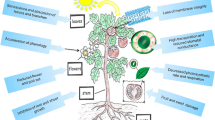
Understanding tolerance to stress should take into account “deciphering the environmental impact on plants” (Bloomfield et al. 2014; Xu 2016). With the new appealing term envirotyping, the fine study of the genotype-by-environment interaction (GEI) has been proposed as a tool useful to the modeling of crop and to phenotype prediction (Xu 2016). The characterization of the crop target environment can be seen as a third “typing” technology, complementing with genotyping and phenotyping to study abiotic stress and to accomplish selection. Environmental information can be collected through multiple environmental trials, geographic and soil information systems, measurement of soil and canopy properties, and evaluation of companion organisms. All the elements of envirotyping are represented in Fig. 9.1. Envirotyping contributes to crop modeling and phenotype prediction through its functional components, including GEI (Cooper et al. 2014). Envirotyping is driven by information and support systems; it has a wide range of applications, including environmental characterization, GEI analysis, phenotype prediction, near-iso-environment construction, agronomic genomics, precision agriculture and breeding. Envirotyping contributes to the development of a four-dimensional profile of the crops, involving genotype, phenotype, envirotype, and time, considered as developmental stage (Xu 2016). The reaction of a trait to different environments, or reaction norm, is also referred as phenotypic plasticity, and its genotypic variation can parallel GEI, as reviewed by Marais et al. (2013). Advances in the area of integrate analysis of environment profiling already led to the development of environmental indexes, to be integrated in comprehensive crop models (Li et al. 2018a).
Envirotyping. Advances in the area of integrate analysis of environment profiling contributes to crop modeling and phenotype prediction through its functional components. Envirotyping is the result of integration of environmental data by means of information and support systems. Factors to be considered include geographical and meteorological data, soil properties, data from living organisms characterizing the environment, crop management, and status of the crop, each containing several subgroups describing factors affecting plant growth and development (Xu 2016).
9.2.2 Genetic Control of Tolerance to Low Temperature
The review of Takeda and Matsuoka (2008) focused on the promising genetic tools available for crop improvements to cope with environmental stress and to maintain yield to feed a growing human population. In this context, the study of cold tolerance genetic control is the basis for implementing modern breeding programs and thus for improving the adaptation of plants to limiting environments. As pointed out by Revilla et al. (2005), low temperature can affect plant growth in different ways, according to the range of temperature experienced. In fact, tolerance to cold and chilling (0–15 °C) or to freezing (<0 °C) temperature is very different from a genetic and physiological perspective. Usually, cold tolerance is typical of annual warm-season crops, while freezing tolerance is found in annual cold-season or perennial crops. Most plants do not suffer chilling injury when temperatures are above 10 °C, although some important crops, such as rice, can be damaged even at 15 °C. Other than differences among species, there is also seasonal variation of cold tolerance as in the case of a woody plant, the coastal Douglas-fir, where factors controlling frost tolerance in the autumn are different from those controlling frost tolerance in the spring (Jermstad et al. 2001).
It is not completely clear which genes or biochemical processes are essential to the achievement of freezing tolerance and which ones affect responses to the low, non-freezing, temperatures but are not involved in freezing tolerance. For example, the loci involved in signal cascades mediating most aspects of cold acclimation, such as increases in abscisic acid, synthesis of compatible osmolytes, and changes in membrane lipid composition, are mostly unknown, besides for the induction of some COR genes (Xin and Browse 2000). Plants respond and adapt to survive under cold stress conditions by adjusting at the molecular and cellular levels, as well as at the physiological and biochemical levels (Sanghera et al. 2011). However, complexity of the response in terms of gene expression has been demonstrated to be widely variable in different genetic backgrounds. In other words, the response to the stress is different depending on the genotype, because the factor limiting tolerance can be different in each background (Canas et al. 2017). The role of phenotypic plasticity in adaptation to nonoptimal environments and its genetic control has been debated for long time (Josephs 2018; Marais et al. 2013; Via et al. 1995). Estimates of phenotypic plasticity were used to identify loci associated with GEI or to stress tolerance. The results obtained so far indicate that traits per se and plasticity may be controlled by different sets of genes (Kusmec et al. 2017). On the other hand, candidate genes for stress tolerance, like those involved in phytohormone-mediated processes, were proved to be involved in multiple stress responses (Kusmec et al. 2017). Some of them showed contrasting effects with different stresses (adaptive genes), while other showed consistent effects across different stress conditions (constitutive genes) (Thoen et al. 2017).
Several studies suggest that in some cases cold tolerance may be under quite simple genetic control. Stone et al. (1993) found that freezing tolerance and acclimation capacity are under control of relatively few genes in populations derived from interspecific crosses between Solanum commersonii and Solanum cardiophyllum. Extensive examples of cold tolerance genetic control is reported by Revilla et al. (2005), who pointed out that tolerance at different growth stages are definitely under the control of different gene sets. Since low temperature resistance in plants seems to be most of the times a very complex trait, involving many different metabolic pathways and cell compartments, physiological breeding has been suggested as a possible way to bring together different components of tolerance, starting with crosses of genotypes with complementary traits (Reynolds and Langridge 2016). To assess the predictive value of those physiological traits for yield and its components, correlation studies and network analyses have been performed (Obata et al. 2015).
9.3 Cold Tolerance in Plant Breeding
As already mentioned in the previous chapters, plant breeding is the key process for the achievement of plants adapted to different climates. Conventional breeding methods have met limited success in improving the cold tolerance of important crop plants based on interspecific or intergeneric hybridization (Jha et al. 2017). The conventional breeding approaches are limited by the complexity of stress tolerance traits, low genetic variance of yield, low heritability of yield components under stress conditions, and lack of efficient selection criteria (Sanghera et al. 2011). However, ample genetic reservoir for cold tolerance can be available in well-adapted breeding populations. Moreover, germplasm collected from high-altitude and low-temperature areas, cold-tolerant mutant, somaclonal variants, and wild species can be exploited for breeding improved cold-tolerant genotypes (Sanghera et al. 2011).
9.3.1 Genetic Variability
In order to successfully apply any breeding program, what is needed first is a suitable genetic diversity, but it is also crucial to develop a deep understanding of the trait(s) to select for. So, according to Dwivedi et al. (2017), assessing the proper functional diversity determining yield in less than optimal environment implies to decide what are the targets of selection and what are the best methods to use. As the first step is to find genetic variability, genetic resources can be considered to access favorable alleles available in the germplasm collections. As pointed out by Revilla et al. (2005), genes for cold tolerance can be transferred from a source to a recipient genotype more easily in case the genetic distance between the two is not very high. The most common way to include a donor genotype in a breeding program is to cross a tolerant accession and a susceptible élite genotype and to select within the segregating population. For example, Zhang et al. (2014) were able to obtain a significant response to selection for cold tolerance in rice from crosses of susceptible by resistant parents. Recurrent selection programs (Frascaroli and Landi 2013; Zhang et al. 2014) already obtained appreciable responses in the segregating populations developed from a cross between two inbreds of different origin, one elite and one locally adapted, both with an average tolerance to cold. In that case, the response to selection was due to the recovery of transgressive individuals originating from recombination. In other cases, favorable alleles were transferred within the same species, from one maize type (i.e., field maize) to another specialty maize (i.e., sweet maize) (Revilla et al. 1998) or from winter to spring wheat (Braun et al. 1996). Exploitation of natural variation for tolerance to abiotic stresses can also rely on the use of landraces (Dwivedi et al. 2013, 2016).
To improve the chances of a successful breeding, stress adaptation loci involved in yield stability and in field performances under environmental extremes were investigated. In particular, for quantitative traits, quantitative traits loci (QTL) involved in the control of adaptation to cold can be identified through linkage mapping based on a mapping population or through genome-wide association studies (GWAS) on genetic panels with genotypes of different origin. According to Thoen et al. (2017), investigation on plant QTL controlling tolerance to stress brings some inherent difficulties due to the complexity of the response. In fact, different stresses are often present simultaneously, as in case of cold and drought, anoxia, and so on. Moreover, phenotype expression in response to two biotic stresses could not be predicted on the basis of existing information regarding interactions between underlying signaling pathways. In addition, cross talk among the responses to various stress complicate further the picture. In spite of those limitations, the study and the dissection of the genetic control of cold tolerance have been pursued for different species. Several studies on QTL analysis for cold tolerance have been already reported by Revilla et al. (2005). In more recent days, thanks to the advancements of the knowledge and to the lowering of genotyping costs, studies gained higher impact, counting on the size of mapping populations. A wide review of the literature is reported by Jha et al. (2017). For barley, QTL analysis in the “Nure” (winter) × “Tremois” (spring) cross (Francia et al. 2004) and the fine mapping on more than 1800 recombinants was carried out (Francia et al. 2007) for the two major low-temperature tolerance QTL, i.e., Fr-H1 and Fr-H2. In case of winter wheat, Zhao et al. (2013) analyzed a large mapping population of 1739 genotypes. In maize some work was focused on the variation in response to cold for chlorophyll content or photosynthesis (Fracheboud et al. 2004; Hund et al. 2005; Rodriguez et al. 2014; Strigens et al. 2013). Given the complexity of the trait, QTL mapping must take advantage of large and complex populations. In maize, for example, 720 double haploids were investigated for adaptation to chilling conditions to map genome regions involved in tolerance (Presterl et al. 2007). In rice almost 2000 lines were considered for fine mapping (Andaya and Mackill 2003). Complex mapping populations, like connected populations, have been used for mapping QTL for tolerance to cold, as in case of maize at germination phase (Li et al. 2018b) where 650 families allowed to map up to 43 QTL, that reduced to three after a meta-analysis of the three connected populations.
New techniques aimed at the identification of specific genes for cold tolerance can be pursued by using transcriptomic and proteomic approaches and/or QTL validation and cloning (Marla et al. 2017; Salvi and Tuberosa 2015; Sheng et al. 2017). A rich analysis of the plethora of results obtained with those methods can be found in dedicated chapters. However, an important effort is needed to integrate all genomics information of crop and model species into databases to make it possible the comparative analysis of genomes, also in response to stress conditions (Naithani et al. 2017; Tello-Ruiz et al. 2018).
9.3.2 Breeding Programs
The objective of breeding is to obtain new varieties that are improved for their tolerance or resistance to different stresses (Trachsel et al. 2017).The definition of selection criteria is an essential step in any breeding program and particularly so when trying to improve cold tolerance (Paleari et al. 2017). In turn, to choose those criteria, plant breeders made effort to model and understand GEI (Crespo-Herrera et al. 2017; Lado et al. 2016) in order to well define mega-environments, in the attempt to select for the tolerance to the more frequent stresses limiting adaptation to a particular region.
Successful strategies must be devised to improve the élite varieties, depending on the genetic control of the trait. In case tolerance is due to just one locus, the good allele, usually found in a non-élite genotype, can be introgressed into the élite one. Introgression is carried out by repeated backcrosses aimed at transferring a genetic determinant (allele) from the donor to the recipient genotype. Introgression can be accelerated by the use of molecular markers for the donor chromosomal region, as well as of markers surrounding that region and the other chromosomes of the recipient genotype. Altogether, backcross breeding is the most common strategy to improve single-target traits, particularly the high heritable ones (Bernardo 2016b). This method proved to be successful to improve yield and cold tolerance together in rice (Meng et al. 2013; Zhang et al. 2014) and to select for cold tolerance even in case of complex genetic control (Zhang et al. 2014).
Breeding for stress tolerance or avoidance, and especially for cold tolerance, has proved to be challenging, at least partly because tolerance mechanisms are often environment-specific, and screening methods that integrate the multiple spatial and temporal variations relevant to this stress are difficult to establish. For the need to adapt to all these multiple spatial and temporal variations characterizing cold environment, tolerance is often controlled by many loci, whose small effects combined confer tolerance to stress. This complex control implies that breeding for tolerant genotypes cannot be pursued as for the monogenic traits . Moreover, the small effect of each quantitative locus is usually subjected to a relevant genotype-by-environment interaction, thus reducing the effectiveness of multiple loci detection when analyzed in multiple environments (Makumburage et al. 2013). Selection can be effective both for high and low tolerance, as in case of Landi et al. (1992) who obtained significant responses in divergent full-sib recurrent selection for the difference between germination at low and at optimal temperature, i.e., for the reaction norm, in maize. Response to selection was appreciable in cold conditions both in controlled environment and in the field (Frascaroli and Landi 2013) but not when the selected genotypes were compared at warm temperature. Later studies evaluated the same populations focusing on associated changes in mitochondrial properties and found a correlated response concerning the interaction between membrane lipids and cytochrome-c-oxidase content (De Santis et al. 1999; Tampieri et al. 2011).
Breeding for cold tolerance can be limited by major bias due to environmental conditions during the evaluation of the trait. In fact, field-breeding programs suffer environmental variations that limit selection progress (Ly et al. 2018), while programs carried out under controlled conditions in the laboratory are limited by the correlation between field and laboratory performance. An accurate, consistent technique would therefore be very helpful. There are examples where reliable methods or indexes can be used as indicators of tolerance in crops under selection (Thapa et al. 2008). Cold chambers are often used for selecting for cold tolerance and for integrating evaluations made in the field (Frascaroli and Landi 2013; Revilla et al. 2014, 2016). The evaluation of reaction to cold during selection can also be impaired by large experimental errors, often a side effect of the stressful condition itself, in addition to the experimental error due to the methodologies and the interaction with unpredictable environmental variation. To overcome this drawback, high-throughput phenotyping systems can be considered, once the experimental procedures are adequately optimized (Junker et al. 2015; Tschiersch et al. 2017).
There are not many recent published reports on the results obtained with the most common breeding methods, probably because these applied methods are designed for improving varieties and not for publishing scientific articles, owing to the lack of systematic design or the absence of novelty. Moreover, uncontrolled environmental variation in breeding programs can limit genetic gains for cold tolerance achievable through selection. Indeed, as previously explained, selection entails some problems of evaluation because of the unpredictable climatic variation of field trials and the inconsistent correlation between controlled environment and field performance. However, breeding designed to adapt maize to Northern Europe has resulted in the release of compact maize hybrids (Frei 2000). Similarly, in Canada most of the genetic yield improvement of maize is attributable to increased cold stress tolerance (Assefa et al. 2017; Tollenaar and Wu 1999). In tomato, Foolad and Lin (2001) obtained with mass selection significant improvements in germination under cold conditions. On the contrary, according to Hensleigh et al. (1992), conventional breeding methods have not been successful in developing barley cultivars with adequate winter hardiness for many northern regions.
9.4 Prospective for Breeding
Selection has been an important human activity since the domestication of crops, but after the advent of the genetic studies, the application of statistics and of the scientific method, breeding has developed its own theory, and selection programs can be planned and developed successfully. The more recent ability to dissect tolerance loci by means of molecular genetics gave rise to a burst of speed in the understanding of the mechanism of tolerance, even though this knowledge is not immediately translatable into a useful breeding procedure when many loci are involved. The availability of élite cultivars and the ability to perform rapid breeding cycles, providing the farmer with new cultivars for specific conditions, are important elements to adapt the cropping system to a new environment or agronomic technique (Atlin et al. 2017). To be ready and flexible for new environmental constraints, breeding programs should evaluate the potential of new cultivars in a wide range of climatic conditions. To reach this goal, according to the plant breeding theory, the main steps are (i) the improvement of base populations, (ii) the selection of commercial élite varieties, and (iii) the dissemination of the new varieties. Recurrent selection is the breeding method of choice to enrich a population with favorable alleles for a polygenic trait (Bernardo 2010; Gorjanc et al. 2018; Mueller et al. 2018). This method has been successfully employed to improve breeding population for stress tolerance, as reported, for example, by Meng et al. (2013) who was able to obtain a large numbers of cold-tolerant lines in rice. Successful breeding programs were carried out in maize (Frascaroli and Landi 2016; Sezegen and Carena 2009; Viesselmann et al. 2014), chickpea (Jha et al. 2017), ryegrass (Iraba et al. 2013), and alfalfa (Castonguay et al. 2009). The selected genotype can also be considered useful materials for genetic, physiological, and molecular dissection of cold tolerance traits using DNA markers and other -omic tools (Frascaroli and Landi 2018; Meng et al. 2013) to obtain indication on the genetic control of the trait and the genomic regions involved in the response to selection.
It has been observed that in most of the cases, the genetic improvement currently possible with conventional approaches does not exceed the 1% per year (Fischer and Edmeades 2010). For this reason different strategies for improving the selection efficiency were explored. As observed by Mickelbart et al. (2015), a combination of different approaches is advisable to accelerate the identification and characterization of specific loci that can be moved by molecular marker-assisted selection (MAS) into elite varieties, minimizing yield-adverse linkages. According to Reynolds and Langridge (2016), physiological breeding can be the strategic tool to speed up the improvement in crop adaptation and, ultimately, the ability to provide food and other products needed for the world improving population. As already mentioned, the choice of the more appropriate method must be made according to the genetic control of the traits. Indeed, selection approaches are different for traits at different levels of integration and genetic complexity, as shown in Fig. 9.2, from Reynolds and Langridge (2016). The figure represents plant selection approaches that may be used for traits controlled at different levels of complexity, starting with simple metabolites and culminating in polygenic productivity traits such as yield and biomass. Current research is expected to yield (i) basic insights on cold tolerance, with advanced information on the affected molecular and physiological processes, and (ii) applied tools such as the identification and the characterization of useful genes for improving tolerance to cold stress. This information could be useful to obtain allele sequences of functionally characterized genes from which functional motifs affecting plant phenotype can be identified and may be used as functional markers (Andersen and Lubberstedt 2003; Brenner et al. 2013). However, it has also been observed that QTL mapping and its application in breeding are most useful for traits, for example, wheat Fusarium head blight resistance or soybean cyst nematode resistance, which might have one or a few underlying major loci (Bernardo 2008).
Traits with different levels of integration and genetic complexity are selected with different approaches. Traits represented by simple metabolites can be followed; thanks to biochemical markers, simple traits controlled by major genes are conveniently selected by means of marker-assisted selection (MAS), while selection for complex traits, such as most of the cold tolerance and productivity traits, must rely on comprehensive methods like genomic selection (GS). The whole picture takes into account also regulatory factors that may interact with the expression of traits at any level as they interact with the environment (epigenetics) and thus enhance the genotype-by-environment interaction (GEI). (From Reynolds and Langridge (2016))
In spite of the huge effort made to understand all the components determining a trait, the consensus among plant breeders today is still that it is usually more efficient to select for the primary trait itself, rather than to select for multiple secondary traits that are components of or associated with it (Bernardo 2016a). As for the use of single genes, so far, the transgenic approach has not been able to handle polygenic traits, while it may be usefully utilized in cultivars transformed with a single transgene (Bernardo 2016a). In the recent years, we witnessed the enhancement of knowledge of plant genomics, and especially of sequence technology, and the shifting of plant science from “explanatory” to “predictive.” Indeed, the possibility to predict the optimal genotype based on genomic information would greatly enhance the efficiency of plant breeding programs. Dense genotyping, that provided a very large amount of SNP (Single Nucleotide Polymorphism) markers, made it possible to develop new promising methods for improving complex traits that are controlled by many QTL with small effects (Bernardo 2016a). Genomic selection (GS) is a breeding method that accelerate the selection of genotypes carrying favorable alleles at loci undetectable by means of mapping models (Meuwissen et al. 2001) and can be integrated into recurrent selection (Gorjanc et al. 2018; Mueller et al. 2018). Genome-wide selection is expected to be particularly efficient when phenotypic selection is nonexistent or ineffective. Simulation and empirical studies revealed that GS can even be more efficient than MAS (Bernardo and Yu 2007), especially for traits that are difficult to measure such as cold tolerance, particularly in the field. The most advanced prediction techniques (Crossa et al. 2017; Heffner et al. 2010; Montesinos-Lopez et al. 2018) are promising in the perspective of selection implementation for cold tolerance in combination with other agronomic traits.
In conclusion, crop adaptation is needed to increase production and stability under cold conditions that are getting worse with climatic change. To improve crop plants for complex traits, such as cold tolerance, the key will be the combination of classical plant breeding with the advances in genomics, crop physiology, and modeling in an integrated profile involving genotype, phenotype, and envirotype. In particular, the most promising approach for selection will involve high-precision, high-throughput phenotyping in controlled growth chambers and platforms, and multi-environment field trials combining agronomic, morphological, physiological, and biochemical data. The exploitation of big data will involve updated statistical models for mapping genomic regions controlling cold tolerance or for predicting the breeding value of genotypes.
References
Andaya VC, Mackill DJ (2003) Mapping of QTLs associated with cold tolerance during the vegetative stage in rice. J Exp Bot 54(392):2579–2585
Andersen JR, Lübberstedt T (2003) Functional markers in plants. Trends in Plant Science 8 (11):554–560
Arief VN, DeLacy IH, Crossa J, Payne T, Singh R, Braun HJ, Tian T, Basford KE, Dieters MJ (2015) Evaluating testing strategies for plant breeding field trials: redesigning a CIMMYT international wheat nursery. Crop Sci 55(1):164–177
Arora R (2018) Mechanism of freeze-thaw injury and recovery: A cool retrospective and warming up to new ideas. Plant Sci 270:301–313
Assefa Y, Prasad PVV, Carter P, Hinds M, Bhalla G, Schon R, Jeschke M, Paszkiewicz S, Ciampitti IA (2017) A new insight into corn yield: trends from 1987 through 2015. Crop Sci 57(5):2799–2811
Atlin GN, Cairns JE, Das B (2017) Rapid breeding and varietal replacement are critical to adaptation of cropping systems in the developing world to climate change. Glob Food Sec 12:31–37
Bai YL, Sunarti S, Kissoudis C, Visser RGF, van der Linden CG (2018) The role of tomato WRKY genes in plant responses to combined abiotic and biotic stresses. Front Plant Sci 9:801
Banerjee A, Roychoudhury A (2018) Strigolactones: multi-level regulation of biosynthesis and diverse responses in plant abiotic stresses. Acta Physiol Plant 40(5):86
Bennett D, Reynolds M, Mullan D, Izanloo A, Kuchel H, Langridge P, Schnurbusch T (2012) Detection of two major grain yield QTL in bread wheat (Triticum aestivum L.) under heat, drought and high yield potential environments. Theor Appl Genet 125(7):1473–1485
Bernardo R, Yu J (2007) Prospects for Genomewide Selection for Quantitative Traits inMaize. Crop Science 47(3):1082
Bernardo R (2008) Molecular markers and selection for complex traits in plants: learning from the last 20 years. Crop Sci 48(5):1649–1664
Bernardo R (2010) Breeding for quantitative traits in plants. Stemma Press, Woodbury
Bernardo R (2016a) Bandwagons I, too, have known. Theor Appl Genet 129(12):2323–2332
Bernardo R (2016b) Genomewide predictions for backcrossing a quantitative trait from an exotic to an adapted line. Crop Sci 56(3):1067–1075
Bita CE, Gerats T (2013) Plant tolerance to high temperature in a changing environment: scientific fundamentals and production of heat stress-tolerant crops. Front Plant Sci 4:273
Bloomfield JA, Rose TJ, King GJ (2014) Sustainable harvest: managing plasticity for resilient crops. Plant Biotechnol J 12(5):517–533
Bradshaw JE (2017) Plant breeding: past, present and future. Euphytica 213(3):60
Braun HJ, Rajaram S, vanGinkel M (1996) CIMMYT's approach to breeding for wide adaptation. Euphytica 92(1–2):175–183
Brenner EA, Beavis WD, Andersen JR, Lübberstedt T (2013) Prospects and limitations fordevelopment and application of functional markers in plants. In Diagnostics in plant breeding,edited by Lübberstedt, T and R K Varshney, 329–346. Dordrecht: Springer
Campbell BM, Thornton P, Zougmore R, van Asten P, Lipper L (2014) Sustainable intensification: what is its role in climate smart agriculture? Curr Opin Environ Sustain 8:39–43
Canas RA, Yesbergenova-Cuny Z, Simons M, Chardon F, Armengaud P, Quillere I, Cukier C, Gibon Y, Limami AM, Nicolas S, Brule L, Lea PJ, Maranas CD, Hirel B (2017) Exploiting the genetic diversity of maize using a combined metabolomics, enzyme activity profiling, and metabolic modeling approach to link leaf physiology to kernel yield. Plant Cell 29(5):919–943
Castonguay Y, Michaud R, Nadeau P, Bertrand A (2009) An indoor screening method for improvement of freezing tolerance in alfalfa. Crop Sci 49(3):809–818
Cattivelli L (2011) More cold tolerant plants for a warmer world. Plant Sci 180(1):1–2
Chinnusamy V, Schumaker K, Zhu JK (2004) Molecular genetic perspectives on cross-talk and specificity in abiotic stress signalling in plants. J Exp Bot 55(395):225–236
Cooper M, Messina CD, Podlich D, Totir LR, Baumgarten A, Hausmann NJ, Wright D, Graham G (2014) Predicting the future of plant breeding: complementing empirical evaluation with genetic prediction. Crop Pasture Sci 65(4):311–336
Cramer GR, Urano K, Delrot S, Pezzotti M, Shinozaki K (2011) Effects of abiotic stress on plants: a systems biology perspective. BMC Plant Biol 11:163
Crespo-Herrera LA, Crossa J, Huerta-Espino J, Autrique E, Mondal S, Velu G, Vargas M, Braun HJ, Singh RP (2017) Genetic yield gains in CIMMYT’s international elite spring wheat yield trials by modeling the genotype x environment interaction. Crop Sci 57(2):789–801
Crossa J, Pérez-Rodríguez P, Cuevas J, Montesinos-López O, Jarquín D,Gustavo de los Campos, Burgueño J, Juan M. González-Camacho, Pérez-Elizalde S,Yoseph Beyene, Susanne Dreisigacker, Ravi Singh, Xuecai Zhang, Manje Gowda, Manish Roorkiwal, Jessica Rutkoski, Rajeev K. Varshney (2017) Genomic Selection in Plant Breeding: Methods, Models, and Perspectives. Trends in Plant Sci 22 (11):961–975
De Santis A, Landi P, Genchi G (1999) Changes of mitochondrial properties in maize seedlings associated with selection for germination at low temperature. Fatty acid composition, cytochrome c oxidase, and adenine nucleotide translocase activities. Plant Physiol 119(2):743–754
Dhillon T, Pearce SP, Stockinger EJ, Distelfeld A, Li CX, Knox AK, Vashegyi I, Vagujfalvi A, Galiba G, Dubcovsky J (2010) Regulation of freezing tolerance and flowering in temperate cereals: the VRN-1 connection. Plant Physiol 153(4):1846–1858
Dolferus R (2014) To grow or not to grow: a stressful decision for plants. Plant Sci 229:247–261
Dubois M, Van den Broeck L, Inze D (2018) The pivotal role of ethylene in plant growth. Trends Plant Sci 23(4):311–323
Dwivedi S, Sahrawat K, Upadhyaya H, Ortiz R (2013) Food, nutrition and agrobiodiversity under global climate change. In: Sparks DL (ed) Advances in agronomy, vol 120. Advances in Agronomy, San Diego, pp 1–128
Dwivedi SL, Ceccarelli S, Blair MW, Upadhyaya HD, Are AK, Ortiz R (2016) Lancrace germplasm for improving yield and abiotic stress adaptation. Trends Plant Sci 21(1):31–42
Dwivedi SL, Scheben A, Edwards D, Spillane C, Ortiz R (2017) Assessing and exploiting functional diversity in germplasm pools to enhance abiotic stress adaptation and yield in cereals and food legumes. Front Plant Sci 8:1461
Eccel E, Rea R, Caffarra A, Crisci A (2009) Risk of spring frost to apple production under future climate scenarios: the role of phenological acclimation. Int J Biometeorol 53(3):273–286
Falloon P, Betts R (2010) Climate impacts on European agriculture and water management in the context of adaptation and mitigation-the importance of an integrated approach. Sci Total Environ 408(23):5667–5687
Feldman M, Levy AA (2009) Genome evolution in allopolyploid wheat-a revolutionary reprogramming followed by gradual changes. J Genet Genomics 36(9):511–518
Fischer RA, Edmeades GO (2010) Breeding and cereal yield progress. Crop Sci 50(2):S85–S98
Foolad MR, Lin GY (2001) Genetic analysis of cold tolerance during vegetative growth in tomato, Lycopersicon esculentum Mill. Euphytica 122(1):105–111
Fracheboud Y, Jompuk C, Ribaut JM, Stamp P, Leipner J (2004) Genetic analysis of cold-tolerance of photosynthesis in maize. Plant Mol Biol 56(2):241–253
Francia E, Barabaschi D, Tondelli A, Laido G, Rizza F, Stanca AM, Busconi M, Fogher C, Stockinger EJ, Pecchioni N (2007) Fine mapping of a HvCBF gene cluster at the frost resistance locus Fr-H2 in barley. Theor Appl Genet 115(8):1083–1091
Francia E, Rizza F, Cattivelli L, Stanca AM, Galiba G, Toth B, Hayes PM, Skinner JS, Pecchioni N (2004) Two loci on chromosome 5H determine low-temperature tolerance in a ‘Nure’ (winter) x ‘Tremois’ (spring) barley map. Theor Appl Genet 108(4):670–680
Frascaroli E, Landi P (2013) Divergent selection in a maize population for germination at low temperature in controlled environment: study of the direct response, of the trait inheritance and of correlated responses in the field. Theor Appl Genet 126(3):733–746
Frascaroli E, Landi P (2016) Cold tolerance in field conditions, its inheritance, agronomic performance and genetic structure of maize lines divergently selected for germination at low temperature. Euphytica 209(3):771–788
Frascaroli E, Landi P (2018) Signatures of divergent selection for cold tolerance in maize. Euphytica 214(5):80
Frei OM (2000) Changes in yield physiology of corn as a result of breeding in northern Europe. Maydica 45(3):173–183
Garnett T, Appleby MC, Balmford A, Bateman IJ, Benton TG, Bloomer P, Burlingame B, Dawkins M, Dolan L, Fraser D, Herrero M, Hoffmann I, Smith P, Thornton PK, Toulmin C, Vermeulen SJ, Godfray HCJ (2013) Sustainable intensification in agriculture: premises and policies. Science 341(6141):33–34
Gorjanc G, Gaynor RC, Hickey JM (2018) Optimal cross selection for long-term genetic gain in two-part programs with rapid recurrent genomic selection. Theor Appl Genet 131:1953
Guo Z, Chen D, Schnurbusch T (2018) Plant and floret growth at distinct developmental stages during the stem elongation phase in wheat. Front Plant Sci 9:330
Haak DC, Fukao T, Grene R, Hua Z, Ivanov R, Perrella G, Li S (2017) Multilevel regulation of abiotic stress responses in plants. Front Plant Sci 8:1564
Heffner EL, Lorenz , Jean-Luc Jannink, Mark E. Sorrells, (2010) Plant Breeding with Genomic Selection: Gain per Unit Time and Cost. Crop Science 50 (5):1681
Hensleigh PF, Blake TK, Welty LE (1992) Natural-selection on winter barley composite cross-XXVI affects winter survival and associated traits. Crop Sci 32(1):57–62
Hossain MA, Li ZG, Hoque TS, Burritt DJ, Fujita M, Munne-Bosch S (2018) Heat or cold priming-induced cross-tolerance to abiotic stresses in plants: key regulators and possible mechanisms. Protoplasma 255(1):399–412
Hund A, Frascaroli E, Leipner J, Jompuk C, Stamp P, Fracheboud Y (2005) Cold tolerance of the photosynthetic apparatus: pleiotropic relationship between photosynthetic performance and specific leaf area of maize seedlings. Mol Breed 16(4):321–331
Huner NPA, Dahal K, Kurepin LV, Savitch L, Singh J, Ivanov AG, Kane K, Sarhan F (2014) Potential for increased photosynthetic performance and crop productivity in response to climate change: role of CBFs and gibberellic acid. Front Chem 2:18
Hussain HA, Hussain S, Khaliq A, Ashraf U, Anjum SA, Men SN, Wang LC (2018) Chilling and drought stresses in crop plants: implications, cross talk, and potential management opportunities. Front Plant Sci 9:393
Iraba A, Castonguay Y, Bertrand A, Floyd DJ, Cloutier J, Belzile F (2013) Characterization of populations of turf-type perennial ryegrass recurrently selected for superior freezing tolerance. Crop Sci 53(5):2225–2238
Jermstad KD, Bassoni DL, Wheeler NC, Anekonda TS, Aitken SN, Adams WT, Neale DB (2001) Mapping of quantitative trait loci controlling adaptive traits in coastal Douglas-fir. II. Spring and fall cold-hardiness. Theor Appl Genet 102(8):1152–1158
Jha UC, Bohra A, Jha R (2017) Breeding approaches and genomics technologies to increase crop yield under low-temperature stress. Plant Cell Rep 36(1):1
Josephs EB (2018) Determining the evolutionary forces shaping G*E. New Phytol 219(1):31–36
Junker A, Muraya MM, Weigelt-Fischer K, Arana-Ceballos F, Klukas C, Melchinger AE, Meyer RC, Riewe D, Altmann T (2015) Optimizing experimental procedures for quantitative evaluation of crop plant performance in high throughput phenotyping systems. Front Plant Sci 5:770
Korner C (2016) Plant adaptation to cold climates. F1000Research 5:F1000
Korner C, Basler D, Hoch G, Kollas C, Lenz A, Randin CF, Vitasse Y, Zimmermann NE (2016) Where, why and how? Explaining the low-temperature range limits of temperate tree species. J Ecol 104(4):1076–1088
Kusmec A, Srinivasan S, Nettleton D, Schnable PS (2017) Distinct genetic architectures for phenotype means and plasticities in Zea mays. Nat Plants 3(9):715–723
Lado B, Gonzalez Barrios P, Quincke M, Silva P, Gutierrez L (2016) Modeling genotype x environment interaction for genomic selection with unbalanced data from a wheat breeding program. Crop Sci 56(5):2165–2179
Landi P, Frascaroli E, Lovato A (1992) Divergent full-sib recurrent selection for germination at low-temperature in a maize population. Euphytica 64(1–2):21–29
Li X, Guo T, Mu Q, Li X, Yu J (2018a) Genomic and environmental determinants and their interplay underlying phenotypic plasticity. Proc Natl Acad Sci U S A 115(26):6679–6684
Li X-l, Yang X, Hu Y-x, Yu X-d, Li Q-l (2014) A novel NAC transcription factor from Suaeda liaotungensis K. enhanced transgenic Arabidopsis drought, salt, and cold stress tolerance. Plant Cell Rep 33(5):767–778
Li XH, Wang GH, Fu JJ, Li L, Jia GY, Ren LS, Lubberstedt T, Wang GY, Wang JH, Gu RL (2018b) QTL mapping in three connected populations reveals a set of consensus genomic regions for low temperature germination ability in Zea mays L. Front Plant Sci 9:65
Ly D, Huet S, Gauffreteau A, Rincent R, Touzy G, Mini A, Jannink JL, Cormier F, Paux E, Lafarge S, Le Gouis J, Charmet G (2018) Whole-genome prediction of reaction norms to environmental stress in bread wheat (Triticum aestivum L.) by genomic random regression. Field Crop Res 216:32–41
Makumburage GB, Richbourg HL, LaTorre KD, Capps A, Chen C, Stapleton AE (2013) Genotype to phenotype maps: multiple input abiotic signals combine to produce growth effects via attenuating signaling interactions in maize. G3-Genes Genomes Genet 3(12):2195–2204
Mansouri-Far C, Sanavy S, Saberali SF (2010) Maize yield response to deficit irrigation during low-sensitive growth stages and nitrogen rate under semi-arid climatic conditions. Agric Water Manag 97(1):12–22
Marais DLD, Hernandez KM, Juenger TE (2013) Genotype-by-environment interaction and plasticity: exploring genomic responses of plants to the abiotic environment. Annu Rev Ecol Evol Syst 44(44):5−+
Marla SR, Shiva S, Welti R, Liu SZ, Burke JJ, Morris GP (2017) Comparative transcriptome and lipidome analyses reveal molecular chilling responses in chilling-tolerant sorghums. Plant Genome 10(3):16
Meng LJ, Lin XY, Wang JM, Chen K, Cui YR, Xu JL, Li ZK (2013) Simultaneous improvement in cold tolerance and yield of temperate japonica rice (Oryza sativa L.) by introgression breeding. Plant Breed 132(6):604–612
Meuwissen TH, Hayes EBJ, ME Goddard (2001) Prediction of total genetic valueusing genome-wide dense marker maps. Genetics 157 (4):1819–1829
Mickelbart MV, Hasegawa PM, Bailey-Serres J (2015) Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat Rev Genet 16(4):237–251
Montesinos-López OA, Montesinos-López A, Crossa J, Montesinos-López JC, Mota-Sanchez D, Estrada-González F, Gillberg J, Singh R, Mondal S, Juliana P (2018) Prediction of multiple-trait and multiple-environment genomic data using recommender systems. G3: Genes, Genomes, Genetics 8(1):131–147
Mueller D, Schopp P, Melchinger AE (2018) Selection on expected maximum haploid breeding values can increase genetic gain in recurrent genomic selection. G3-Genes Genomes Genet 8(4):1173–1181
Mustafavi SH, Badi HN, Sekara A, Mehrafarin A, Janda T, Ghorbanpour M, Rafiee H (2018) Polyamines and their possible mechanisms involved in plant physiological processes and elicitation of secondary metabolites. Acta Physiol Plant 40(6):102
Naithani S, Preece J, D'Eustachio P, Gupta P, Amarasinghe V, Dharmawardhana PD, Wu G, Fabregat A, Elser JL, Weiser J, Keays M, Fuentes AM-P, Petryszak R, Stein LD, Ware D, Jaiswal P (2017) Plant reactome: a resource for plant pathways and comparative analysis. Nucleic Acids Res 45(D1):D1029–D1039
Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K (2014) The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front Plant Sci 5:170
Nemali KS, Bonin C, Dohleman FG, Stephens M, Reeves WR, Nelson DE, Castiglioni P, Whitsel JE, Sammons B, Silady RA, Anstrom D, Sharp RE, Patharkar OR, Clay D, Coffin M, Nemeth MA, Leibman ME, Luethy M, Lawson M (2015) Physiological responses related to increased grain yield under drought in the first biotechnology-derived drought-tolerant maize. Plant Cell Environ 38(9):1866–1880
Obata T, Witt S, Lisec J, Palacios-Rojas N, Florez-Sarasa I, Yousfi S, Araus JL, Cairns JE, Fernie AR (2015) Metabolite profiles of maize leaves in drought, heat, and combined stress field trials reveal the relationship between metabolism and grain yield. Plant Physiol 169(4):2665–2683
Olesen JE, Borgesen CD, Elsgaard L, Palosuo T, Rotter RP, Skjelvag AO, Peltonen-Sainio P, Borjesson T, Trnka M, Ewert F, Siebert S, Brisson N, Eitzinger J, van Asselt ED, Oberforster M, van der Fels-Klerx HJ (2012) Changes in time of sowing, flowering and maturity of cereals in Europe under climate change. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 29(10):1527–1542
Olesen JE, Trnka M, Kersebaum KC, Skjelvag AO, Seguin B, Peltonen-Sainio P, Rossi F, Kozyra J, Micale F (2011) Impacts and adaptation of European crop production systems to climate change. Eur J Agron 34(2):96–112
Paleari L, Movedi E, Cappelli G, Wilson LT, Confalonieri R (2017) Surfing parameter hyperspaces under climate change scenarios to design future rice ideotypes. Glob Chang Biol 23(11):4651–4662
Presterl T, Ouzunova M, Schmidt W, Moller EM, Rober FK, Knaak C, Ernst K, Westhoff P, Geiger HH (2007) Quantitative trait loci for early plant vigour of maize grown in chilly environments. Theor Appl Genet 114(6):1059–1070
Rapacz M, Ergon A, Hoglind M, Jorgensen M, Jurczyk B, Ostrem L, Rognli OA, Tronsmo AM (2014) Overwintering of herbaceous plants in a changing climate. Still more questions than answers. Plant Sci 225:34–44
Revilla P, Butrón A, Rodríguez VM, Malvar RA, Ordás A (2005) Breeding for cold tolerance. In: Ashraf M, Harris PJC (eds) Abiotic stresses. Plant resistance through breedingand molecular approaches. The Haworth Press, Inc, New York, pp 301–398
Revilla P, Malvar RA, Cartea ME, Ordas A (1998) Identifying open-pollinated populations of field corn as sources of cold tolerance for improving sweet corn. Euphytica 101(2):239–247
Revilla P, Rodriguez VM, Ordas A, Rincent R, Charcosset A, Giauffret C, Melchinger AE, Schon CC, Bauer E, Altmann T, Brunel D, Moreno-Gonzalez J, Campo L, Ouzunova M, Alvarez A, de Galarreta JIR, Laborde J, Malvar RA (2016) Association mapping for cold tolerance in two large maize inbred panels. BMC Plant Biol 16:127
Revilla P, Rodriguez VM, Ordas A, Rincent R, Charcosset A, Giauffret C, Melchinger AE, Schon CC, Bauer E, Altmann T, Brunel D, Moreno-Gonzalez J, Campo L, Ouzunova M, Laborde J, Alvarez A, de Galarreta JIR, Malvar RA (2014) Cold tolerance in two large maize inbred panels adapted to European climates. Crop Sci 54(5):1981–1991
Reynolds M, Langridge P (2016) Physiological breeding. Curr Opin Plant Biol 31:162–171
Rodriguez V, Butron A, Rady MOA, Soengas P, Revilla P (2014) Identification of quantitative trait loci involved in the response to cold stress in maize (Zea mays L.). Mol Breed 33(2):363–371
Salvi S, Tuberosa R (2015) The crop QTLome comes of age. Curr Opin Biotechnol 32:179–185
Sanghera GS, Wani SH, Hussain W, Singh NB (2011) Engineering cold stress tolerance in crop plants. Curr Genomics 12(1):30–43
Seki M, Kamei A, Yamaguchi-Shinozaki K, Shinozaki K (2003) Molecular responses to drought, salinity and frost: common and different paths for plant protection. Curr Opin Biotechnol 14(2):194–199
Semenov MA (2009) Impacts of climate change on wheat in England and Wales. J R Soc Interface 6(33):343–350
Semenov MA, Stratonovitch P, Alghabari F, Gooding MJ (2014) Adapting wheat in Europe for climate change. J Cereal Sci 59(3):245–256
Sezegen B, Carena MJ (2009) Divergent recurrent selection for cold tolerance in two improved maize populations. Euphytica 167(2):237–244
Sheng J, Zheng X, Wang J, Zeng X, Zhou F, Jin S, Hu Z, Diao Y (2017) Transcriptomics and proteomics reveal genetic and biological basis of superior biomass crop Miscanthus. Sci Rep 7:13777
Sthapit BR, Witcombe JR (1998) Inheritance of tolerance to chilling stress in rice during germination and plumule greening. Crop Sci 38(3):660–665
Stojakovic M, Mitrovic B, Zoric M, Ivanovic M, Stanisavljevic D, Nastasic A, Dodig D (2015) Grouping pattern of maize test locations and its impact on hybrid zoning. Euphytica 204(2):419–431
Stone JM, Palta JP, Bamberg JB, Weiss LS, Harbage JF (1993) Inheritance of freezing resistance in tuber-bearing solanum species – evidence for independent genetic-control of nonacclimated freezing tolerance and cold-acclimation capacity. Proc Natl Acad Sci U S A 90(16):7869–7873
Strigens A, Freitag NM, Gilbert X, Grieder C, Riedelsheimer C, Schrag TA, Messmer R, Melchinger AE (2013) Association mapping for chilling tolerance in elite flint and dent maize inbred lines evaluated in growth chamber and field experiments. Plant Cell Environ 36(10):1871–1887
Takeda S, Matsuoka M (2008) Genetic approaches to crop improvement: responding to environmental and population changes. Nat Rev Genet 9(6):444–457
Tampieri E, Baraldi E, Carnevali F, Frascaroli E, De Santis A (2011) The activity of plant inner membrane anion channel (PIMAC) can be performed by a chloride channel (CLC) protein in mitochondria from seedlings of maize populations divergently selected for cold tolerance. J Bioenerg Biomembr 43(6):611–621
Tello-Ruiz MK, Naithani S, Stein JC, Gupta P, Campbell M, Olson A, Wei S, Preece J, Geniza MJ, Jiao Y, Lee YK, Wang B, Mulvaney J, Chougule K, Elser J, Al-Bader N, Kumari S, Thomason J, Kumar V, Bolser DM, Naamati G, Tapanari E, Fonseca N, Huerta L, Iqbal H, Keays M, Fuentes AM-P, Tang A, Fabregat A, D’Eustachio P, Weiser J, Stein LD, Petryszak R, Papatheodorou I, Kersey PJ, Lockhart P, Taylor C, Jaiswal P, Ware D (2018) Gramene 2018: unifying comparative genomics and pathway resources for plant research. Nucleic Acids Res 46(D1):D1181–D1189
Thakur AK, Uphoff NT (2017) How the system of rice intensification can contribute to climate-smart agriculture. Agron J 109(4):1163–1182
Thapa B, Arora R, Knapp AD, Brummer EC (2008) Applying freezing test to quantify cold acclimation in Medicago truncatula. J Am Soc Hortic Sci 133(5):684–691
Thoen MPM, Olivas NHD, Kloth KJ, Coolen S, Huang P-P, Aarts MGM, Bac-Molenaar JA, Bakker J, Bouwmeester HJ, Broekgaarden C, Bucher J, Busscher-Lange J, Cheng X, Fradin EF, Jongsma MA, Julkowska MM, Keurentjes JJB, Ligterink W, Pieterse CMJ, Ruyter-Spira C, Smant G, Testerink C, Usadel B, van Loon JJA, van Pelt JA, van Schaik CC, van Wees SCM, Visser RGF, Voorrips R, Vosman B, Vreugdenhil D, Warmerdam S, Wiegers GL, van Heerwaarden J, Kruijer W, van Eeuwijk FA, Dicke M (2017) Genetic architecture of plant stress resistance: multi-trait genome-wide association mapping. New Phytol 213(3):1346–1362
Tollenaar M, Wu J (1999) Yield improvement in temperate maize is attributable to greater stress tolerance. Crop Sci 39(6):1597–1604
Trachsel S, Burgueno J, Suarez EA, San Vicente FM, Rodriguez CS, Dhliwayo T (2017) Interrelations among early vigor, flowering time, physiological maturity, and grain yield in tropical maize (Zea mays L.) under multiple abiotic stresses. Crop Sci 57(1):229–242
Trnka M, Olesen JE, Kersebaum KC, Skjelvag AO, Eitzinger J, Seguin B, Peltonen-Sainio P, Rotter R, Iglesias A, Orlandini S, Dubrovsky M, Hlavinka P, Balek J, Eckersten H, Cloppet E, Calanca P, Gobin A, Vucetic V, Nejedlik P, Kumar S, Lalic B, Mestre A, Rossi F, Kozyra J, Alexandrov V, Semeradova D, Zalud Z (2011) Agroclimatic conditions in Europe under climate change. Glob Chang Biol 17(7):2298–2318
Tschiersch H, Junker A, Meyer RC, Altmann T (2017) Establishment of integrated protocols for automated high throughput kinetic chlorophyll fluorescence analyses. Plant Methods 13:54
Via S, Gomulkiewicz R, Dejong G, Scheiner SM, Schlichting CD, Vantienderen PH (1995) Adaptive phenotypic plasticity – consensus and controversy. Trends Ecol Evol 10(5):212–217
Viesselmann LM, De Vries B, Dodson HG, Tracy WF (2014) Recurrent selection for seedling growth of sweet corn in cool temperatures. Crop Sci 54(3):1033–1040
Waha K, van Bussel LGJ, Muller C, Bondeau A (2012) Climate-driven simulation of global crop sowing dates. Glob Ecol Biogeogr 21(2):247–259
Xin Z, Browse J (2000) Cold comfort farm: the acclimation of plants to freezing temperatures. Plant Cell Environ 23(9):893–902
Xu WH, Li QX, Jones P, Wang XLL, Trewin B, Yang S, Zhu C, Zhai PM, Wang JF, Vincent L, Dai AG, Gao Y, Ding YH (2018) A new integrated and homogenized global monthly land surface air temperature dataset for the period since 1900. Clim Dyn 50(7–8):2513–2536
Xu Y (2016) Envirotyping for deciphering environmental impacts on crop plants. Theor Appl Genet 129(4):653–673
Zhang F, Ma XF, Gao YM, Hao XB, Li ZK (2014) Genome-wide response to selection and genetic basis of cold tolerance in rice (Oryza sativa L.). BMC Genet 15:55
Zhao YS, Gowda M, Wurschum T, Longin CFH, Korzun V, Kollers S, Schachschneider R, Zeng J, Fernando R, Dubcovsky J, Reif JC (2013) Dissecting the genetic architecture of frost tolerance in Central European winter wheat. J Exp Bot 64(14):4453–4460
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Frascaroli, E. (2018). Breeding Cold-Tolerant Crops. In: Wani, S., Herath, V. (eds) Cold Tolerance in Plants. Springer, Cham. https://doi.org/10.1007/978-3-030-01415-5_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-01415-5_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-01414-8
Online ISBN: 978-3-030-01415-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)