Abstract
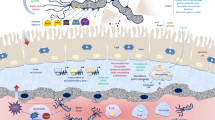
The cag pathogenicity island is a well-characterized virulence determinant. It is composed of 32 genes that encode a type IV bacterial secretion system and is linked with a more severe clinical outcome. The following chapters will explore the manipulation of bacterial factors in order to understand their role in gastric mucosal disease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Alm RA, Bina J, Andrews BM, Doig P, Hancock RE, Trust TJ (2000) Comparative genomics of Helicobacter pylori: analysis of the outer membrane protein families. Infect Immun 68:4155–4168
Go MF, Kapur V, Graham DY, Musser JM (1996) Population genetic analysis of Helicobacter pylori by multilocus enzyme electrophoresis: extensive allelic diversity and recombinational population structure. J Bacteriol 178:3934–3938
Salama N, Guillemin K, McDaniel TK, Sherlock G, Tompkins L, Falkow S (2000) A whole-genome microarray reveals genetic diversity among Helicobacter pylori strains. Proc Natl Acad Sci USA 97:14668–14673
Tomb JF, White O, Kerlavage AR, Clayton RA, Sutton GG, Fleischmann RD et al (1997) The complete genome sequence of the gastric pathogen Helicobacter pylori. Nature 388:539–547
Israel DA, Salama N, Krishna U, Rieger UM, Atherton JC, Falkow S et al (2001) Helicobacter pylori genetic diversity within the gastric niche of a single human host. Proc Natl Acad Sci USA 98:14625–14630
Akopyants NS, Clifton SW, Kersulyte D, Crabtree JE, Youree BE, Reece CA et al (1998) Analyses of the cag pathogenicity island of Helicobacter pylori. Mol Microbiol 28:37–53
Alm RA, Ling LS, Moir DT, King BL, Brown ED, Doig PC et al (1999) Genomic-sequence comparison of two unrelated isolates of the human gastric pathogen Helicobacter pylori. Nature 397:176–180
Censini S, Lange C, Xiang Z, Crabtree JE, Ghiara P, Borodovsky M et al (1996) Cag, a pathogenicity island of Helicobacter pylori, encodes type I specific and disease-associated virulence factors. Proc Natl Acad Sci USA 93:14648–14653
Blaser MJ, Perez-Perez GI, Kleanthous H, Cover TL, Peek RM, Chyou PH et al (1995) Infection with Helicobacter pylori strains possessing cagA is associated with an increased risk of developing adenocarcinoma of the stomach. Cancer Res 55:2111–2115
Cover TL, Dooley CP, Blaser MJ (1990) Characterization of and human serologic response to proteins in Helicobacter pylori broth culture supernatants with vacuolizing cytotoxin activity. Infect Immun 58:603–610
Crabtree JE, Taylor JD, Wyatt JI, Heatley RV, Shallcross TM, Tompkins DS et al (1991) Mucosal IgA recognition of Helicobacter pylori 120 kDa protein, peptic ulceration, and gastric pathology. Lancet 338:332–335
Crabtree JE, Wyatt JI, Sobala GM, Miller G, Tompkins DS, Primrose JN et al (1993) Systemic and mucosal humoral responses to Helicobacter pylori in gastric cancer. Gut 34:1339–1343
Kuipers EJ, Perez-Perez GI, Meuwissen SG, Blaser MJ (1995) Helicobacter pylori and atrophic gastritis: importance of the cagA status. J Natl Cancer Inst 87:1777–1780
Parsonnet J, Replogle M, Yang S, Hiatt R (1997) Seroprevalence of CagA-positive strains among Helicobacter pylori-infected, healthy young adults. J Infect Dis 175:1240–1242
Peek RM Jr, Miller GG, Tham KT, Perez-Perez GI, Zhao X, Atherton JC et al (1995) Heightened inflammatory response and cytokine expression in vivo to cagA + Helicobacter pylori strains. Lab Invest 73:760–770
Queiroz DM, Mendes EN, Rocha GA, Oliveira AM, Oliveira CA, Magalhaes PP et al (1998) CagA-positive Helicobacter pylori and risk for developing gastric carcinoma in Brazil. Int J Cancer 78:135–139
Rudi J, Kolb C, Maiwald M, Zuna I, von Herbay A, Galle PR et al (1997) Serum antibodies against Helicobacter pylori proteins VacA and CagA are associated with increased risk for gastric adenocarcinoma. Dig Dis Sci 42:1652–1659
Shimoyama T, Fukuda S, Tanaka M, Mikami T, Munakata A, Crabtree JE (1998) CagA seropositivity associated with development of gastric cancer in a Japanese population. J Clin Pathol 51:225–228
Torres J, Perez-Perez GI, Leal-Herrera Y, Munoz O (1998) Infection with CagA+ Helicobacter pylori strains as a possible predictor of risk in the development of gastric adenocarcinoma in Mexico. Int J Cancer 78:298–300
Vorobjova T, Nilsson I, Kull K, Maaroos HI, Covacci A, Wadstrom T et al (1998) CagA protein seropositivity in a random sample of adult population and gastric cancer patients in Estonia. Eur J Gastroenterol Hepatol 10:41–46
Camorlinga-Ponce M, Romo C, Gonzalez-Valencia G, Munoz O, Torres J (2004) Topographical localisation of cagA positive and cagA negative Helicobacter pylori strains in the gastric mucosa; an in situ hybridisation study. J Clin Pathol 57:822–828
Kwok T, Zabler D, Urman S, Rohde M, Hartig R, Wessler S et al (2007) Helicobacter exploits integrin for type IV secretion and kinase activation. Nature 449:862–866
Jimenez-Soto LF, Kutter S, Sewald X, Ertl C, Weiss E, Kapp U et al (2009) Helicobacter pylori type IV secretion apparatus exploits beta1 integrin in a novel RGD-independent manner. PLoS Pathogens 5:e1000684
Odenbreit S, Puls J, Sedlmaier B, Gerland E, Fischer W, Haas R (2000) Translocation of Helicobacter pylori CagA into gastric epithelial cells by type IV secretion. Science 287:1497–1500
Ohnishi N, Yuasa H, Tanaka S, Sawa H, Miura M, Matsui A et al (2008) Transgenic expression of Helicobacter pylori CagA induces gastrointestinal and hematopoietic neoplasms in mouse. Proc Natl Acad Sci USA 105:1003–1008
Stein M, Bagnoli F, Halenbeck R, Rappuoli R, Fantl WJ, Covacci A (2002) c-Src/Lyn kinases activate Helicobacter pylori CagA through tyrosine phosphorylation of the EPIYA motifs. Mol Microbiol 43:971–980
Hatakeyama M (2004) Oncogenic mechanisms of the Helicobacter pylori CagA protein. Nat Rev Cancer 4:688–694
Higashi H, Yokoyama K, Fujii Y, Ren S, Yuasa H, Saadat I et al (2005) EPIYA motif is a membrane-targeting signal of Helicobacter pylori virulence factor CagA in mammalian cells. J Biol Chem 280:23130–23137
Naito M, Yamazaki T, Tsutsumi R, Higashi H, Onoe K, Yamazaki S et al (2006) Influence of EPIYA-repeat polymorphism on the phosphorylation-dependent biological activity of Helicobacter pylori CagA. Gastroenterology 130:1181–1190
Basso D, Zambon CF, Letley DP, Stranges A, Marchet A, Rhead JL et al (2008) Clinical relevance of Helicobacter pylori cagA and vacA gene polymorphisms. Gastroenterology 135:91–99
Schneider N, Krishna U, Romero-Gallo J, Israel DA, Piazuelo MB, Camargo MC et al (2009) Role of Helicobacter pylori CagA molecular variations in induction of host phenotypes with carcinogenic potential. J Infect Dis 199:1218–1221
Argent RH, Hale JL, El-Omar EM, Atherton JC (2008) Differences in Helicobacter pylori CagA tyrosine phosphorylation motif patterns between western and East Asian strains, and influences on interleukin-8 secretion. J Med Microbiol 57:1062–1067
Asahi M, Azuma T, Ito S, Ito Y, Suto H, Nagai Y et al (2000) Helicobacter pylori CagA protein can be tyrosine phosphorylated in gastric epithelial cells. J Exp Med 191:593–602
Backert S, Ziska E, Brinkmann V, Zimny-Arndt U, Fauconnier A, Jungblut PR et al (2000) Translocation of the Helicobacter pylori CagA protein in gastric epithelial cells by a type IV secretion apparatus. Cell Microbiol 2:155–164
Segal ED, Cha J, Lo J, Falkow S, Tompkins LS (1999) Altered states: involvement of phosphorylated CagA in the induction of host cellular growth changes by Helicobacter pylori. Proc Natl Acad Sci USA 96:14559–14564
Selbach M, Moese S, Hauck CR, Meyer TF, Backert S (2002) Src is the kinase of the Helicobacter pylori CagA protein in vitro and in vivo. J Biol Chem 277:6775–6778
Stein M, Rappuoli R, Covacci A (2000) Tyrosine phosphorylation of the Helicobacter pylori CagA antigen after cag-driven host cell translocation. Proc Natl Acad Sci USA 97:1263–1268
Higashi H, Nakaya A, Tsutsumi R, Yokoyama K, Fujii Y, Ishikawa S et al (2004) Helicobacter pylori CagA induces Ras-independent morphogenetic response through SHP-2 recruitment and activation. J Biol Chem 279:17205–17216
Higashi H, Tsutsumi R, Fujita A, Yamazaki S, Asaka M, Azuma T et al (2002) Biological activity of the Helicobacter pylori virulence factor CagA is determined by variation in the tyrosine phosphorylation sites. Proc Natl Acad Sci USA 99:14428–14433
Higashi H, Tsutsumi R, Muto S, Sugiyama T, Azuma T, Asaka M et al (2002) SHP-2 tyrosine phosphatase as an intracellular target of Helicobacter pylori CagA protein. Science 295:683–686
Tsutsumi R, Higashi H, Higuchi M, Okada M, Hatakeyama M (2002) Attenuation of Helicobacter pylori CagA-SHP-2 signaling by interaction between CagA and C-terminal Src kinase. J Biol Chem 21:21
Tammer I, Brandt S, Hartig R, Konig W, Backert S (2007) Activation of Abl by Helicobacter pylori: a novel kinase for CagA and crucial mediator of host cell scattering. Gastroenterology 132:1309–1319
Selbach M, Moese S, Hurwitz R, Hauck CR, Meyer TF, Backert S (2003) The Helicobacter pylori CagA protein induces cortactin dephosphorylation and actin rearrangement by c-Src inactivation. EMBO J 22:515–528
Moese S, Selbach M, Brinkmann V, Karlas A, Haimovich B, Backert S et al (2007) The Helicobacter pylori CagA protein disrupts matrix adhesion of gastric epithelial cells by dephosphorylation of vinculin. Cell Microbiol 9:1148–1161
Selbach M, Moese S, Backert S, Jungblut PR, Meyer TF (2004) The Helicobacter pylori CagA protein induces tyrosine dephosphorylation of ezrin. Proteomics 4:2961–2968
Moese S, Selbach M, Kwok T, Brinkmann V, Konig W, Meyer TF et al (2004) Helicobacter pylori induces AGS cell motility and elongation via independent signaling pathways. Infect Immun 72:3646–3649
Tsutsumi R, Takahashi A, Azuma T, Higashi H, Hatakeyama M (2006) Focal adhesion kinase is a substrate and downstream effector of SHP-2 complexed with Helicobacter pylori CagA. Mol Cell Biol 26:261–276
Amieva MR, Vogelmann R, Covacci A, Tompkins LS, Nelson WJ, Falkow S (2003) Disruption of the epithelial apical-junctional complex by Helicobacter pylori CagA. Science 300:1430–1434
Bagnoli F, Buti L, Tompkins L, Covacci A, Amieva MR (2005) Helicobacter pylori CagA induces a transition from polarized to invasive phenotypes in MDCK cells. Proc Natl Acad Sci USA 102:16339–16344
Franco AT, Israel DA, Washington MK, Krishna U, Fox JG, Rogers AB et al (2005) Activation of beta-catenin by carcinogenic Helicobacter pylori. Proc Natl Acad Sci USA 102:10646–10651
Murata-Kamiya N, Kurashima Y, Teishikata Y, Yamahashi Y, Saito Y, Higashi H et al (2007) Helicobacter pylori CagA interacts with E-cadherin and deregulates the beta-catenin signal that promotes intestinal transdifferentiation in gastric epithelial cells. Oncogene 26:4617–4626
Saadat I, Higashi H, Obuse C, Umeda M, Murata-Kamiya N, Saito Y et al (2007) Helicobacter pylori CagA targets PAR1/MARK kinase to disrupt epithelial cell polarity. Nature 447:330–333
Suzuki M, Mimuro H, Suzuki T, Park M, Yamamoto T, Sasakawa C (2005) Interaction of CagA with Crk plays an important role in Helicobacter pylori-induced loss of gastric epithelial cell adhesion. J Exp Med 202:1235–1247
Churin Y, Al-Ghoul L, Kepp O, Meyer TF, Birchmeier W, Naumann M (2003) Helicobacter pylori CagA protein targets the c-Met receptor and enhances the mitogenic response. J Cell Biol 161:249–255
Mimuro H, Suzuki T, Tanaka J, Asahi M, Haas R, Sasakawa C (2002) Grb2 is a key mediator of Helicobacter pylori CagA protein activities. Mol Cell 10:745–755
Lu H, Murata-Kamiya N, Saito Y, Hatakeyama M (2009) Role of partitioning-defective 1/microtubule affinity-regulating kinases in the morphogenetic activity of Helicobacter pylori CagA. J Biol Chem 284:23024–23036
Umeda M, Murata-Kamiya N, Saito Y, Ohba Y, Takahashi M, Hatakeyama M (2009) Helicobacter pylori CagA causes mitotic impairment and induces chromosomal instability. J Biol Chem 284:22166–22172
Kurashima Y, Murata-Kamiya N, Kikuchi K, Higashi H, Azuma T, Kondo S et al (2008) Deregulation of beta-catenin signal by Helicobacter pylori CagA requires the CagA-multimerization sequence. Int J Cancer 122:823–831
Suzuki M, Mimuro H, Kiga K, Fukumatsu M, Ishijima N, Morikawa H et al (2009) Helicobacter pylori CagA phosphorylation-independent function in epithelial proliferation and inflammation. Cell Host Microbe 5:23–34
Nesic D, Miller MC, Quinkert ZT, Stein M, Chait BT, Stebbins CE (2010) Helicobacter pylori CagA inhibits PAR1-MARK family kinases by mimicking host substrates. Nat Struct Mol Biol 17:130–132
Brandt S, Kwok T, Hartig R, Konig W, Backert S (2005) NF-kappaB activation and potentiation of proinflammatory responses by the Helicobacter pylori CagA protein. Proc Natl Acad Sci USA 102:9300–9305
Kim SY, Lee YC, Kim HK, Blaser MJ (2006) Helicobacter pylori CagA transfection of gastric epithelial cells induces interleukin-8. Cell Microbiol 8:97–106
Lamb A, Yang XD, Tsang YH, Li JD, Higashi H, Hatakeyama M et al (2009) Helicobacter pylori CagA activates NF-kappaB by targeting TAK1 for TRAF6-mediated Lys 63 ubiquitination. EMBO Rep 10:1242–1249
Keates S, Keates AC, Warny M, Peek RM, Murray PG, Kelly CP (1999) Differential activation of mitogen-activated protein kinases in AGS gastric epithelial cells by cag + and cag - Helicobacter pylori. J Immunol 163:5552–5559
Keates S, Sougioultzis S, Keates AC, Zhao D, Peek RM, Shaw LM et al (2001) Cag + Helicobacter pylori induce transactivation of the epidermal growth factor receptor in AGS gastric epithelial cells. J Biol Chem 276:48127–48134
Meyer-Ter-Vehn T, Covacci A, Kist M, Pahl HL (2000) Helicobacter pylori activates mitogen-activated protein kinase cascades and induces expression of the proto-oncogenes c-fos and c-jun. J Biol Chem 275:16064–16072
Naumann M, Wessler S, Bartsch C, Wieland B, Covacci A, Haas R et al (1999) Activation of activator protein 1 and stress response kinases in epithelial cells colonized by Helicobacter pylori encoding the cag pathogenicity island. J Biol Chem 274:31655–31662
Kaparakis M, Turnbull L, Carneiro L, Firth S, Coleman HA, Parkington HC et al (2010) Bacterial membrane vesicles deliver peptidoglycan to NOD1 in epithelial cells. Cell Microbiol 12:372–385
Boughan PK, Argent RH, Body-Malapel M, Park JH, Ewings KE, Bowie AG et al (2006) Nucleotide-binding oligomerization domain-1 and epidermal growth factor receptor: critical regulators of beta-defensins during Helicobacter pylori infection. J Biol Chem 281:11637–11648
Viala J, Chaput C, Boneca IG, Cardona A, Girardin SE, Moran AP et al (2004) Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nat Immunol 5:1166–1174
Allison CC, Kufer TA, Kremmer E, Kaparakis M, Ferrero RL (2009) Helicobacter pylori induces MAPK phosphorylation and AP-1 activation via a NOD1-dependent mechanism. J Immunol 183:8099–8109
Watanabe T, Asano N, Fichtner-Feigl S, Gorelick PL, Tsuji Y, Matsumoto Y et al (2010) NOD1 contributes to mouse host defense against Helicobacter pylori via induction of type I IFN and activation of the ISGF3 signaling pathway. J Clin Invest 120:1645–1662
Strober W, Murray PJ, Kitani A, Watanabe T (2006) Signalling pathways and molecular interactions of NOD1 and NOD2. Nat Rev Immunol 6:9–20
Nagy TA, Frey MR, Yan F, Israel DA, Polk DB, Peek RM (2009) Helicobacter pylori regulates cellular migration and apoptosis by activation of phosphatidylinositol 3-kinase signaling. J Infect Dis 199:641–651
Wang G, Olczak A, Forsberg LS, Maier RJ (2009) Oxidative stress-induced peptidoglycan deacetylase in Helicobacter pylori. J Biol Chem 284:6790–6800
Franco AT, Friedman DB, Nagy TA, Romero-Gallo J, Krishna U, Kendall A et al (2009) Delineation of a carcinogenic Helicobacter pylori proteome. Mol Cell Proteomics 8:1947–1958
Wang G, Maier SE, Lo LF, Maier G, Dosi S, Maier RJ (2010) Peptidoglycan deacetylation in Helicobacter pylori contributes to bacterial survival by mitigating host immune responses. Infect Immun 78:4660–4666
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Noto, J.M., Peek, R.M. (2012). The Helicobacter pylori cag Pathogenicity Island. In: Houghton, J. (eds) Helicobacter Species. Methods in Molecular Biology, vol 921. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-005-2_7
Download citation
DOI: https://doi.org/10.1007/978-1-62703-005-2_7
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-004-5
Online ISBN: 978-1-62703-005-2
eBook Packages: Springer Protocols