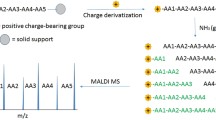
Abstract
Peptide microarrays are useful devices for the high throughput study of biomolecular or peptide–cell interactions. Whereas the synthesis of unmodified peptide libraries is an easy task and can be performed at reasonable cost, the synthesis of libraries of modified peptides remains expensive and time consuming. This bottleneck led us to examine the possibility to produce modified peptide microspots by in situ chemical modification of unmodified peptide microspots. The great advantage would be the preparation of a series of complex microarrays (daughter microarrays) starting from an easy-to-make and cost-effective unmodified peptide microarray (parent microarray). One step toward this goal has been presented in the accompanying chapter dealing with the in situ methylation methodology for studying the specificity of antibodies directed toward methylated epitopes. Here we describe the development of a novel desorption/ionization on silicon nanowires mass spectrometry (DIOSiNWs-MS) technique for characterizing the in situ chemical modification of peptides.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Yang, X. J., and Seto, E. (2008) Lysine acetylation: codified crosstalk with other posttranslational modifications. Mol. Cell 31, 449–61.
Wisniewski, J. R., Zougman, A., and Mann, M. (2008) Nepsilon-formylation of lysine is a widespread post-translational modification of nuclear proteins occurring at residues involved in regulation of chromatin function. Nucleic Acids Res. 36, 570–7.
Floor, E., Maples, A. M., Rankin, C. A., Yaganti, V. M., Shank, S. S., Nichols, G. S., O’Laughlin, M., Galeva, N. A., and Williams, T. D. (2006) A one-carbon modification of protein lysine associated with elevated oxidative stress in human substantia nigra. J. Neurochem. 97, 504–14.
Karas, M., and Hillenkamp, F. (1988) Laser desorption ionization of proteins with molecular masses exceeding 10,000 daltons. Anal. Chem. 60, 2299–301.
Wei, J., Buriak, J. M., and Siuzdak, G. (1999) Desorption-ionization mass spectrometry on porous silicon. Nature 399, 243–6.
Piret, G., Coffinier, Y., Roux, C., Melnyk, O., and Boukherroub, R. (2008) Biomolecule and nanoparticle transfer on patterned and heterogeneously wetted superhydrophobic silicon nanowire surfaces. Langmuir 24, 1670–2.
Douani, R., Hadjersi, T., Boukherroub, R., Adour, L., and Manseri, A. (2008). Formation of aligned silicon-nanowire on silicon in aqueous HF/(AgNO3 + Na2S2O8) solution. Appl. Surf. Sci. 254, 7219–22
Coffinier, Y., Janel, S., Addad, A., Blossey, R., Gengembre, L., Payen, E., and Boukherroub, R. (2007) Preparation of superhydrophobic silicon nanowires surfaces. Langmuir 23, 1608–11.
Locht, C., Hougardy, J. M., Rouanet, C., Place, S., and Mascart, F. (2006) Heparin-binding hemagglutinin, from an extrapulmonary dissemination factor to a powerful diagnostic and protective antigen against tuberculosis. Tuberculosis (Edinb) 86, 303–9.
Menozzi, F. D., Bischoff, R., Fort, E., Brennan, M. J., and Locht, C. (1998) Molecular characterization of the mycobacterial heparin-binding hemagglutinin, a mycobacterial adhesin. Proc. Natl Acad. Sci. U S A 95, 12625–30.
Menozzi, F. D., Rouse, J. H., Alavi, M., Laude-Sharp, M., Muller, J., Bischoff, R., Brennan, M. J., and Locht, C. (1996) Identification of a heparin-binding hemagglutinin present in mycobacteria. J. Exp. Med. 184, 993–1001.
Pethe, K., Bifani, P., Drobecq, H., Sergheraert, C., Debrie, A. S., Locht, C., and Menozzi, F. D. (2002) Mycobacterial heparin-binding hemagglutinin and laminin-binding protein share antigenic methyllysines that confer resistance to proteolysis. Proc. Natl Acad. Sci. U S A 99, 10759–64.
Temmerman, S., Pethe, K., Parra, M., Alonso, S., Rouanet, C., Pickett, T., Drowart, A., Debrie, A. S., Delogu, G., Menozzi, F. D., Sergheraert, C., Brennan, M. J., Mascart, F., and Locht, C. (2004) Methylation-dependent T cell immunity to Mycobacterium tuberculosis heparin-binding hemagglutinin. Nat. Med. 10, 935–41.
Acknowledgments
We gratefully acknowledge the financial support from CNRS, Université de Lille Nord de France, Institut Pasteur de Lille, IFR 142, Région Nord Pas de Calais, the European Community (FEDER), and from Cancéropôle Nord-Ouest. This research was performed using the Chemistry Systems Biology platform (http://csb.ibl.fr). GP thanks the DGA (Direction Générale de l’Armement) for support with a PhD scholarship.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Piret, G., Drobecq, H., Boukherroub, R., Melnyk, O. (2010). In Situ Chemical Modification of Peptide Microarrays: Characterization by Desorption/Ionization on Silicon Nanowires. In: Uttamchandani, M., Yao, S. (eds) Small Molecule Microarrays. Methods in Molecular Biology, vol 669. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-60761-845-4_10
Download citation
DOI: https://doi.org/10.1007/978-1-60761-845-4_10
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-60761-844-7
Online ISBN: 978-1-60761-845-4
eBook Packages: Springer Protocols