Summary
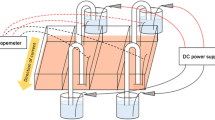
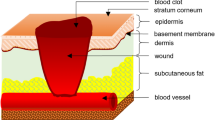
Electric fields were measured at human skin wounds over one and half centuries ago. Modern techniques have verified and greatly extended our understanding of the existence of endogenous wound electric fields. In virtually all wounds studied, disruption of an epithelial layer instantaneously generates endogenous electric fields. As electric fields have the intrinsic property of being vectorial, it has long been proposed that these fields may serve as a directional signal guiding cell migration in wound healing. We have established several experimental systems to study the guidance effects and mechanisms of electric fields on cell migration. Most types of cells migrate directionally in a small electric field, a phenomenon called galvanotaxis/electrotaxis. Remarkably, electric fields of strength equal to those detected at in vivo wounds direct cell migration and override some other well-accepted coexistent guidance cues such as contact inhibition. The naturally occurring endogenous electric fields therefore may be an important signaling mechanism that regulates directional cell movement in vivo. Applied electric fields may have a potential clinical role in guiding cell migration in wound healing. The magnitude and direction of the electric field can be more precisely and quickly changed than most other guidance cues such as chemical cues. Application of electric fields thus offers a robust experimental system for study of directional cell migration with extensive flexibility. We present a brief review of the background and describe the experimental system for studying electrotaxis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Du Bois-Reymond, E. (1843) Vorläufiger Abriss einer Untersuchung uber den sogenannten Froschstrom und die electomotorischen Fische. Ann. Phy. U. Chem. 58, 1–30.
Du Bois-Reymond, E. (1860) Untersuchungen uber thierische Elektricitat, Zweiter Band, Zweite Abtheilung (Erste Lieferung). Georg Reimer, Berlin.
Borgens, R. B., Vanable, J. W., Jr. and Jaffe, L. F. (1977) Bioelectricity and regeneration: large currents leave the stumps of regenerating newt limbs. Proc. Natl. Acad. Sci. USA 74, 4528–4532.
Barker, A. T., Jaffe, L. F. and Vanable, J. W., Jr. (1982) The glabrous epidermis of cavies contains a powerful battery. Am. J. Physiol. 242, R358–366.
Jaffe, L. F. and Vanable, J. W., Jr. (1984) Electric fields and wound healing. Clin. Dermatol. 2, 34–44.
Reid, B., Nuccitelli, R. and Zhao, M. (2007) Non-invasive measurement of bioelectric currents with a vibrating probe. Nat. Protoc. 2, 661–669.
Reid, B., Song, B., McCaig, C. D. and Zhao, M. (2005) Wound healing in rat cornea: the role of electric currents. FASEB J. 19, 379–386.
Nuccitelli, R. (2003) Endogenous electric fields in embryos during development, regeneration and wound healing. Radiat. Prot. Dosimetry 106, 375–383.
Wang, E., Reid, B., Lois, N., Forrester, J. V., McCaig, C. D. and Zhao, M. (2005) Electrical inhibition of lens epithelial cell proliferation: an additional factor in secondary cataract? FASEB J. 19, 842–844.
Mukerjee, E. V., Isseroff, R. R., Nuccitelli, R., Collins, S. D. and Smith, R. L. (2006) Microneedle array for measuring wound generated electric fields. Conf. Proc. IEEE Eng. Med. Biol. Soc. 1, 4326–4328.
Nuccitelli, R., Nuccitelli, P., Ramlatchan, S., Sanger, R. and Smith, P. J. (2008) Imaging the electric field associated with mouse and human skin wounds. Wound Repair Regen. 16, 432–441.
Chiang, M. C., Cragoe, E. J., Jr. and Vanable, J. W., Jr. (1991) Intrinsic electric fields promote. epithelization of wounds in the newt, Notophthalmus viridescens. Dev. Biol. 146, 377–385.
Nuccitelli, R. (1992) Endogenous ionic currents and DC electric fields in multicellular animal tissues. Bioelectromagnetics Suppl 1, 147–157.
McCaig, C. D., Rajnicek, A. M., Song, B. and Zhao, M. (2005) Controlling cell behavior electrically: current views and future potential. Physiol. Rev. 85, 943–978.
Zhao, M., Song, B., Pu, J., Forrester, J. V. and McCaig, C. D. (2003) Direct visualization of a stratified epithelium reveals that wounds heal by unified sliding of cell sheets. FASEB J. 17, 397–406.
Robinson, K. R. (1985) The responses of cells to electrical fields: a review. J. Cell Biol. 101, 2023–2027.
Ferrier, J., Ross, S. M., Kanehisa, J. and Aubin, J. E. (1986) Osteoclasts and osteoblasts migrate in opposite directions in response to a constant electrical field. J. Cell. Physiol. 129, 283–288.
Soong, H. K., Parkinson, W. C., Bafna, S., Sulik, G. L. and Huang, S. C. (1990) Movements of cultured corneal epithelial cells and stromal fibroblasts in electric fields. Invest. Ophthalmol. Vis. Sci. 31, 2278–2282.
Sillman, A. L., Quang, D. M., Farboud, B., Fang, K. S., Nuccitelli, R. and Isseroff, R. R. (2003) Human dermal fibroblasts do not exhibit directional migration on collagen I in direct-current electric fields of physiological strength. Exp. Dermatol. 12, 396–402.
Zhao, M., Song, B., Pu, J., Wada, T., Reid, B., Tai, G., et al. (2006) Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-γ and PTEN. Nature 442, 457–460.
Zhao, M., Pu, J., Forrester, J. V. and McCaig, C. D. (2002) Membrane lipids, EGF receptors, and intracellular signals colocalize and are polarized in epithelial cells moving directionally in a physiological electric field. FASEB J. 16, 857–859.
Bourne, H. R. (2006) G-proteins and GPCRs: from the beginning (2006) Ernst Schering Found. Symp. Proc. 2006(2), 1–21.
Wu, L., Valkema, R., Van Haastert, P. J. and Devreotes, P. N. (1995) The G protein beta subunit is essential for multiple responses to chemoattractants in Dictyostelium. J. Cell Biol. 129, 1667–1675.
Zhao, M., Jin, T., McCaig, C. D., Forrester, J. V. and Devreotes, P. N. (2002) Genetic analysis of the role of G protein-coupled receptor signaling in electrotaxis. J. Cell Biol. 157, 921–927.
Servant, G., Weiner, O. D., Herzmark, P., Balla, T., Sedat, J. W. and Bourne, H. R. (2000) Polarization of chemoattractant receptor signaling during neutrophil chemotaxis. Science 287, 1037–1040.
Zhao, M., Agius-Fernandez, A., Forrester, J. V. and McCaig, C. D. (1996) Orientation and directed migration of cultured corneal epithelial cells in small electric fields are serum dependent. J. Cell Sci. 109, 1405–1414.
Gipson, I. K. and Grill, S. M. (1982) A tech.nique for obtaining sheets of intact rabbit corneal epithelium. Invest. Ophthalmol. Vis. Sci. 23, 269–273.
Zhao, M., Agius-Fernandez, A., Forrester, J. V. and McCaig, C. D. (1996) Directed migration of corneal epithelial sheets in physiological electric fields. Invest. Ophthalmol. Vis. Sci. 37, 2548–2558.
Gruler, H. and Nuccitelli, R. (1991) Neural crest cell galvanotaxis: new data and a novel approach to the analysis of both galvanotaxis and chemotaxis. Cell Motil. Cytoskeleton 19, 121–133.
Acknowledgments
The authors’ work was supported by research grants from the Wellcome Trust, London, UK, and a startup support from the University of California, Davis (to M.Z). We thank Drs. Servant and Bourne for providing HL60 cells and transfected HL60 cells. We thank Drs. Fei Wang, Jingsong Xu, and Paul Herzmark for helping us with some experiments on HL60 cells.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Humana Press
About this protocol
Cite this protocol
Tai, G., Reid, B., Cao, L., Zhao, M. (2009). Electrotaxis and Wound Healing: Experimental Methods to Study Electric Fields as a Directional Signal for Cell Migration. In: Jin, T., Hereld, D. (eds) Chemotaxis. Methods in Molecular Biology™, vol 571. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-60761-198-1_5
Download citation
DOI: https://doi.org/10.1007/978-1-60761-198-1_5
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-60761-197-4
Online ISBN: 978-1-60761-198-1
eBook Packages: Springer Protocols