Abstract
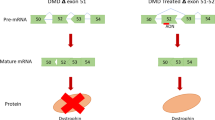
Exon skipping is a therapeutic approach that is feasible for various genetic diseases and has been studied and developed for over two decades. This approach uses antisense oligonucleotides (AON) to modify the splicing of pre-mRNA to correct the mutation responsible for a disease, or to suppress a particular gene expression, as in allergic diseases. Antisense-mediated exon skipping is most extensively studied in Duchenne muscular dystrophy (DMD) and has developed from in vitro proof-of-concept studies to clinical trials targeting various single exons such as exon 45 (casimersen), exon 53 (NS-065/NCNP-01, golodirsen), and exon 51 (eteplirsen). Eteplirsen (brand name Exondys 51), is the first approved antisense therapy for DMD in the USA, and provides a treatment option for ~14% of all DMD patients, who are amenable to exon 51 skipping. Eteplirsen is granted accelerated approval and marketing authorization by the US Food and Drug Administration (FDA), on the condition that additional postapproval trials show clinical benefit. Permanent exon skipping achieved at the DNA level using clustered regularly interspaced short palindromic repeats (CRISPR) technology holds promise in current preclinical trials for DMD. In hopes of achieving clinical success parallel to DMD, exon skipping and splice modulation are also being studied in other muscular dystrophies, such as Fukuyama congenital muscular dystrophy (FCMD), dysferlinopathy including limb-girdle muscular dystrophy type 2B (LGMD2B), Miyoshi myopathy (MM), and distal anterior compartment myopathy (DMAT), myotonic dystrophy, and merosin-deficient congenital muscular dystrophy type 1A (MDC1A). This chapter also summarizes the development of antisense-mediated exon skipping therapy in diseases such as Usher syndrome, dystrophic epidermolysis bullosa, fibrodysplasia ossificans progressiva (FOP), and allergic diseases.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zhang L, Li X, Zhao R (2013) Structural analyses of the pre-mRNA splicing machinery. Protein Sci 22(6):677–692. https://doi.org/10.1002/pro.2266
Veltrop M, Aartsma-Rus A (2014) Antisense-mediated exon skipping: taking advantage of a trick from mother nature to treat rare genetic diseases. Exp Cell Res 325(1):50–55. https://doi.org/10.1016/j.yexcr.2014.01.026
Sardone V, Zhou H, Muntoni F et al (2017) Antisense oligonucleotide-based therapy for neuromuscular disease. Molecules 22(4):E563. https://doi.org/10.3390/molecules22040563
Stein CA, Castanotto D (2017) FDA-approved oligonucleotide therapies in 2017. Mol Ther 25(5):1069–1075. https://doi.org/10.1016/j.ymthe.2017.03.023
Nowak KJ, Davies KE (2004) Duchenne muscular dystrophy and dystrophin: pathogenesis and opportunities for treatment. EMBO Rep 5(9):872–876. https://doi.org/10.1038/sj.embor.7400221
Hoffman EP, Brown RH, Kunkel LM (1992) Dystrophin: the protein product of the Duchene muscular dystrophy locus. 1987. Biotechnology 24:457–466
Nichols B, Takeda S, Yokota T (2015) Nonmechanical roles of dystrophin and associated proteins in exercise, neuromuscular junctions, and brains. Brain Sci 5(3):275–298. https://doi.org/10.3390/brainsci5030275
Aoki Y, Nakamura A, Yokota T et al (2010) In-frame dystrophin following exon 51-skipping improves muscle pathology and function in the exon 52-deficient mdx mouse. Mol Ther 18(11):1995–2005. https://doi.org/10.1038/mt.2010.186
Wein N, Vulin A, Findlay AR et al (2017) Efficient skipping of single exon duplications in DMD patient-derived cell lines using an antisense oligonucleotide approach. J Neuromuscul Dis 4(3):199–207. https://doi.org/10.3233/JND-170233
Maruyama R, Echigoya Y, Caluseriu O et al (2017) Systemic delivery of morpholinos to skip multiple exons in a dog model of duchenne muscular dystrophy. Methods Mol Biol 1565:201–213. https://doi.org/10.1007/978-1-4939-6817-6_17
Yokota T, Duddy W, Echigoya Y et al (2012) Exon skipping for nonsense mutations in Duchenne muscular dystrophy: too many mutations, too few patients? Expert Opin Biol Ther 12(9):1141–1152. https://doi.org/10.1517/14712598.2012.693469
Lu QL, Rabinowitz A, Chen YC et al (2005) Systemic delivery of antisense oligoribonucleotide restores dystrophin expression in body-wide skeletal muscles. Proc Natl Acad Sci U S A 102(1):198–203. https://doi.org/10.1073/pnas.0406700102
Monaco AP, Bertelson CJ, Liechti-Gallati S et al (1988) An explanation for the phenotypic differences between patients bearing partial deletions of the DMD locus. Genomics 2(1):90–95
Aartsma-Rus A, Van Deutekom JC, Fokkema IF et al (2006) Entries in the Leiden Duchenne muscular dystrophy mutation database: an overview of mutation types and paradoxical cases that confirm the reading-frame rule. Muscle Nerve 34(2):135–144. https://doi.org/10.1002/mus.20586
Bladen CL, Salgado D, Monges S et al (2015) The TREAT-NMD DMD global database: analysis of more than 7,000 Duchenne muscular dystrophy mutations. Hum Mutat 36(4):395–402. https://doi.org/10.1002/humu.22758
Aartsma-Rus A, Fokkema I, Verschuuren J et al (2009) Theoretic applicability of antisense-mediated exon skipping for Duchenne muscular dystrophy mutations. Hum Mutat 30(3):293–299. https://doi.org/10.1002/humu.20918
Release FN (2016) FDA grants accelerated approval to first drug for Duchenne muscular dystrophy. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm521263.htm
Aartsma-Rus A, Krieg AM (2017) FDA approves eteplirsen for duchenne muscular dystrophy: the next chapter in the eteplirsen saga. Nucleic Acid Ther 27(1):1–3. https://doi.org/10.1089/nat.2016.0657
Mitrpant C, Fletcher S, Iversen PL et al (2009) By-passing the nonsense mutation in the 4 CV mouse model of muscular dystrophy by induced exon skipping. J Gene Med 11(1):46–56. https://doi.org/10.1002/jgm.1265
Yokota T, Lu QL, Partridge T et al (2009) Efficacy of systemic morpholino exon-skipping in Duchenne dystrophy dogs. Ann Neurol 65(6):667–676. https://doi.org/10.1002/ana.21627
Niks EH, Aartsma-Rus A (2017) Exon skipping: a first in class strategy for Duchenne muscular dystrophy. Expert Opin Biol Ther 17(2):225–236. https://doi.org/10.1080/14712598.2017.1271872
Shimo T, Maruyama R, Yokota T (2018) Designing effective antisense oligonucleotides for exon skipping. Methods Mol Biol 1687:143–155. https://doi.org/10.1007/978-1-4939-7374-3_10
Maruyama R, Echigoya Y, Nakamura A et al. (2017) Systemic injections of peptide-conjugated morpholinos improve cardiac symptoms of a dog model of duchenne muscular dystrophy. Paper presented at the MOLECULAR THERAPY,
Aartsma-Rus A, Janson AA, Kaman WE et al (2004) Antisense-induced multiexon skipping for Duchenne muscular dystrophy makes more sense. Am J Hum Genet 74(1):83–92. https://doi.org/10.1086/381039
Beroud C, Tuffery-Giraud S, Matsuo M et al (2007) Multiexon skipping leading to an artificial DMD protein lacking amino acids from exons 45 through 55 could rescue up to 63% of patients with Duchenne muscular dystrophy. Hum Mutat 28(2):196–202. https://doi.org/10.1002/humu.20428
van Vliet L, de Winter CL, van Deutekom JC et al (2008) Assessment of the feasibility of exon 45-55 multiexon skipping for Duchenne muscular dystrophy. BMC Med Genet 9:105. https://doi.org/10.1186/1471-2350-9-105
Echigoya Y, Aoki Y, Miskew B et al (2015) Long-term efficacy of systemic multiexon skipping targeting dystrophin exons 45-55 with a cocktail of vivo-morpholinos in mdx52 mice. Mol Ther Nucleic Acids 4:e225. https://doi.org/10.1038/mtna.2014.76
Echigoya Y, Nakamura A, Nagata T et al (2017) Effects of systemic multiexon skipping with peptide-conjugated morpholinos in the heart of a dog model of Duchenne muscular dystrophy. Proc Natl Acad Sci U S A 114(16):4213–4218. https://doi.org/10.1073/pnas.1613203114
Nakamura A, Shiba N, Miyazaki D et al (2017) Comparison of the phenotypes of patients harboring in-frame deletions starting at exon 45 in the Duchenne muscular dystrophy gene indicates potential for the development of exon skipping therapy. J Hum Genet 62(4):459–463. https://doi.org/10.1038/jhg.2016.152
Yokota T, Takeda S, Lu QL et al (2009) A renaissance for antisense oligonucleotide drugs in neurology: exon skipping breaks new ground. Arch Neurol 66(1):32–38. https://doi.org/10.1001/archneurol.2008.540
Aoki Y, Yokota T, Wood MJ (2013) Development of multiexon skipping antisense oligonucleotide therapy for Duchenne muscular dystrophy. Biomed Res Int 2013:402369. https://doi.org/10.1155/2013/402369
Aoki Y, Yokota T, Nagata T et al (2012) Bodywide skipping of exons 45-55 in dystrophic mdx52 mice by systemic antisense delivery. Proc Natl Acad Sci U S A 109(34):13763–13768. https://doi.org/10.1073/pnas.1204638109
Arechavala-Gomeza V, Graham IR, Popplewell LJ et al (2007) Comparative analysis of antisense oligonucleotide sequences for targeted skipping of exon 51 during dystrophin pre-mRNA splicing in human muscle. Hum Gene Ther 18(9):798–810. https://doi.org/10.1089/hum.2006.061
Mendell JR, Rodino-Klapac LR, Sahenk Z et al (2013) Eteplirsen for the treatment of Duchenne muscular dystrophy. Ann Neurol 74(5):637–647. https://doi.org/10.1002/ana.23982
Miyatake S, Mizobe Y, Takizawa H et al (2018) Exon skipping therapy using phosphorodiamidate morpholino oligomers in the mdx52 mouse model of duchenne muscular dystrophy. Methods Mol Biol 1687:123–141. https://doi.org/10.1007/978-1-4939-7374-3_9
Lim KR, Maruyama R, Yokota T (2017) Eteplirsen in the treatment of Duchenne muscular dystrophy. Drug Des Devel Ther 11:533–545. https://doi.org/10.2147/DDDT.S97635
Mendell JR, Goemans N, Lowes LP et al (2016) Longitudinal effect of eteplirsen versus historical control on ambulation in Duchenne muscular dystrophy. Ann Neurol 79(2):257–271. https://doi.org/10.1002/ana.24555
Nguyen Q, Yokota T (2017) Immortalized muscle cell model to test the exon skipping efficacy for duchenne muscular dystrophy. J Pers Med 7(4):13
Echigoya Y, Lim KRQ, Trieu N et al (2017) Quantitative antisense screening and optimization for exon 51 skipping in duchenne muscular dystrophy. Mol Ther 25(11):2561–2572. https://doi.org/10.1016/j.ymthe.2017.07.014
Therapeutics S (2017) Sarepta therapeutics announces positive results in its study evaluating gene expression, dystrophin production, and dystrophin localization in patients with duchenne muscular dystrophy (DMD) amenable to skipping exon 53 treated with golodirsen (SRP-4053). http://investorrelations.sarepta.com/news-releases/news-release-details/sarepta-therapeutics-announces-positive-results-its-study. Accessed Sep 2017
Mojica FJ, Diez-Villasenor C, Garcia-Martinez J et al (2005) Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements. J Mol Evol 60(2):174–182. https://doi.org/10.1007/s00239-004-0046-3
Jansen R, Embden JD, Gaastra W et al (2002) Identification of genes that are associated with DNA repeats in prokaryotes. Mol Microbiol 43(6):1565–1575
Barrangou R, Fremaux C, Deveau H et al (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315(5819):1709–1712. https://doi.org/10.1126/science.1138140
Maeder ML, Gersbach CA (2016) Genome-editing technologies for gene and cell therapy. Mol Ther 24(3):430–446. https://doi.org/10.1038/mt.2016.10
Gaj T, Gersbach CA, Barbas CF 3rd (2013) ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 31(7):397–405. https://doi.org/10.1016/j.tibtech.2013.04.004
Li HL, Fujimoto N, Sasakawa N et al (2015) Precise correction of the dystrophin gene in duchenne muscular dystrophy patient induced pluripotent stem cells by TALEN and CRISPR-Cas9. Stem Cell Reports 4(1):143–154. https://doi.org/10.1016/j.stemcr.2014.10.013
Tabebordbar M, Zhu K, Cheng JKW et al (2016) In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science 351(6271):407–411. https://doi.org/10.1126/science.aad5177
Nelson CE, Hakim CH, Ousterout DG et al (2016) In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy. Science 351(6271):403–407. https://doi.org/10.1126/science.aad5143
Long C, Amoasii L, Mireault AA et al (2016) Postnatal genome editing partially restores dystrophin expression in a mouse model of muscular dystrophy. Science 351(6271):400–403. https://doi.org/10.1126/science.aad5725
Ousterout DG, Kabadi AM, Thakore PI et al (2015) Multiplex CRISPR/Cas9-based genome editing for correction of dystrophin mutations that cause Duchenne muscular dystrophy. Nat Commun 6:6244. https://doi.org/10.1038/ncomms7244
Zetsche B, Gootenberg JS, Abudayyeh OO et al (2015) Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163(3):759–771. https://doi.org/10.1016/j.cell.2015.09.038
Shmakov S, Abudayyeh OO, Makarova KS et al (2015) Discovery and functional characterization of diverse class 2 CRISPR-Cas systems. Mol Cell 60(3):385–397. https://doi.org/10.1016/j.molcel.2015.10.008
Zhang Y, Long C, Li H et al (2017) CRISPR-Cpf1 correction of muscular dystrophy mutations in human cardiomyocytes and mice. Sci Adv 3(4):e1602814. https://doi.org/10.1126/sciadv.1602814
Kamoshita S, Konishi Y, Segawa M et al (1976) Congenital muscular dystrophy as a disease of the central nervous system. Arch Neurol 33(7):513–516
Toda T, Segawa M, Nomura Y et al (1993) Localization of a gene for Fukuyama type congenital muscular dystrophy to chromosome 9q31-33. Nat Genet 5(3):283–286. https://doi.org/10.1038/ng1193-283
Watanabe M, Kobayashi K, Jin F et al (2005) Founder SVA retrotransposal insertion in Fukuyama-type congenital muscular dystrophy and its origin in Japanese and northeast Asian populations. Am J Med Genet A 138(4):344–348. https://doi.org/10.1002/ajmg.a.30978
Kobayashi K, Nakahori Y, Miyake M et al (1998) An ancient retrotransposal insertion causes Fukuyama-type congenital muscular dystrophy. Nature 394(6691):388–392. https://doi.org/10.1038/28653
Hayashi YK, Ogawa M, Tagawa K et al (2001) Selective deficiency of alpha-dystroglycan in Fukuyama-type congenital muscular dystrophy. Neurology 57(1):115–121
Lee JJA, Yokota T (2016) Translational research in nucleic acid therapies for muscular dystrophies. In: Takeda SI, Miyagoe-Suzuki Y, Mori-Yoshimura M (eds) Translational research in muscular dystrophy. Springer, Japan, Tokyo, pp 87–102. https://doi.org/10.1007/978-4-431-55678-7_6
Colombo R, Bignamini AA, Carobene A et al (2000) Age and origin of the FCMD 3′-untranslated-region retrotransposal insertion mutation causing Fukuyama-type congenital muscular dystrophy in the Japanese population. Hum Genet 107(6):559–567
Lee JJ, Yokota T (2013) Antisense therapy in neurology. J Pers Med 3(3):144–176. https://doi.org/10.3390/jpm3030144
Taniguchi-Ikeda M, Kobayashi K, Kanagawa M et al (2011) Pathogenic exon-trapping by SVA retrotransposon and rescue in Fukuyama muscular dystrophy. Nature 478(7367):127–131. https://doi.org/10.1038/nature10456
Harper PS (1975) Congenital myotonic dystrophy in Britain. II. Genetic basis. Arch Dis Child 50(7):514–521
Klein AF, Dastidar S, Furling D et al (2015) Therapeutic approaches for dominant muscle diseases: highlight on myotonic dystrophy. Curr Gene Ther 15(4):329–337
Brook JD, McCurrach ME, Harley HG et al (1992) Molecular basis of myotonic dystrophy: expansion of a trinucleotide (CTG) repeat at the 3′ end of a transcript encoding a protein kinase family member. Cell 68(4):799–808
Mahadevan M, Tsilfidis C, Sabourin L et al (1992) Myotonic dystrophy mutation: an unstable CTG repeat in the 3′ untranslated region of the gene. Science 255(5049):1253–1255
Fu YH, Pizzuti A, Fenwick RG Jr et al (1992) An unstable triplet repeat in a gene related to myotonic muscular dystrophy. Science 255(5049):1256–1258
Liquori CL, Ricker K, Moseley ML et al (2001) Myotonic dystrophy type 2 caused by a CCTG expansion in intron 1 of ZNF9. Science 293(5531):864–867. https://doi.org/10.1126/science.1062125
Klein AF, Gasnier E, Furling D (2011) Gain of RNA function in pathological cases: focus on myotonic dystrophy. Biochimie 93(11):2006–2012. https://doi.org/10.1016/j.biochi.2011.06.028
Charlet BN, Savkur RS, Singh G et al (2002) Loss of the muscle-specific chloride channel in type 1 myotonic dystrophy due to misregulated alternative splicing. Mol Cell 10(1):45–53
Savkur RS, Philips AV, Cooper TA (2001) Aberrant regulation of insulin receptor alternative splicing is associated with insulin resistance in myotonic dystrophy. Nat Genet 29(1):40–47. https://doi.org/10.1038/ng704
Fugier C, Klein AF, Hammer C et al (2011) Misregulated alternative splicing of BIN1 is associated with T tubule alterations and muscle weakness in myotonic dystrophy. Nat Med 17(6):720–725. https://doi.org/10.1038/nm.2374
Rau F, Laine J, Ramanoudjame L et al (2015) Abnormal splicing switch of DMD's penultimate exon compromises muscle fibre maintenance in myotonic dystrophy. Nat Commun 6:7205. https://doi.org/10.1038/ncomms8205
Wheeler TM, Lueck JD, Swanson MS et al (2007) Correction of ClC-1 splicing eliminates chloride channelopathy and myotonia in mouse models of myotonic dystrophy. J Clin Invest 117(12):3952–3957. https://doi.org/10.1172/JCI33355
Letter from Ionis Pharmaceuticals & Biogen to the MDF Community (2017.) http://us8.campaign-archive2.com/?u=8f5969cac3271759ce78c8354&id=1109538bcf&e=[UNIQID]. Accessed Sep 2017
Koebis M, Kiyatake T, Yamaura H et al (2013) Ultrasound-enhanced delivery of morpholino with bubble liposomes ameliorates the myotonia of myotonic dystrophy model mice. Sci Rep 3:2242. https://doi.org/10.1038/srep02242
Anderson LV, Davison K, Moss JA et al (1999) Dysferlin is a plasma membrane protein and is expressed early in human development. Hum Mol Genet 8(5):855–861
Glover L, Brown RH Jr (2007) Dysferlin in membrane trafficking and patch repair. Traffic 8(7):785–794. https://doi.org/10.1111/j.1600-0854.2007.00573.x
Aoki M (2004) Dysferlinopathy. GeneReviews®, Seattle (WA). University of Washington, Seattle, pp 1993–2017
Liu J, Aoki M, Illa I et al (1998) Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nat Genet 20(1):31–36. https://doi.org/10.1038/1682
Illa I, Serrano-Munuera C, Gallardo E et al (2001) Distal anterior compartment myopathy: a dysferlin mutation causing a new muscular dystrophy phenotype. Ann Neurol 49(1):130–134
Patel NJ, Van Dyke KW, Espinoza LR (2017) Limb-girdle muscular dystrophy 2B and Miyoshi presentations of dysferlinopathy. Am J Med Sci 353(5):484–491. https://doi.org/10.1016/j.amjms.2016.05.024
Lee JJA, Maruyama R, Sakurai H et al (2018) Cell membrane repair assay using a two-photon laser microscope. J Vis Exp 131:e56999–e56999. https://doi.org/10.3791/56999
Sinnreich M, Therrien C, Karpati G (2006) Lariat branch point mutation in the dysferlin gene with mild limb-girdle muscular dystrophy. Neurology 66(7):1114–1116. https://doi.org/10.1212/01.wnl.0000204358.89303.81
Aartsma-Rus A, Singh KH, Fokkema IF et al (2010) Therapeutic exon skipping for dysferlinopathies? Eur J Hum Genet 18(8):889–894. https://doi.org/10.1038/ejhg.2010.4
Barthelemy F, Blouin C, Wein N et al (2015) Exon 32 skipping of dysferlin rescues membrane repair in patients' cells. J Neuromuscul Dis 2(3):281–290. https://doi.org/10.3233/JND-150109
Durbeej M (2015) Laminin-alpha2 chain-deficient congenital muscular dystrophy: pathophysiology and development of treatment. Curr Top Membr 76:31–60. https://doi.org/10.1016/bs.ctm.2015.05.002
Collins J, Bonnemann CG (2010) Congenital muscular dystrophies: toward molecular therapeutic interventions. Curr Neurol Neurosci Rep 10(2):83–91. https://doi.org/10.1007/s11910-010-0092-8
Zhang X, Vuolteenaho R, Tryggvason K (1996) Structure of the human laminin alpha2-chain gene (LAMA2), which is affected in congenital muscular dystrophy. J Biol Chem 271(44):27664–27669
Siala O, Louhichi N, Triki C et al (2007) Severe MDC1A congenital muscular dystrophy due to a splicing mutation in the LAMA2 gene resulting in exon skipping and significant decrease of mRNA level. Genet Test 11(3):199–207. https://doi.org/10.1089/gte.2006.0517
Aoki Y, Nagata T, Yokota T et al (2013) Highly efficient in vivo delivery of PMO into regenerating myotubes and rescue in laminin-alpha2 chain-null congenital muscular dystrophy mice. Hum Mol Genet 22(24):4914–4928. https://doi.org/10.1093/hmg/ddt341
Kimberling WJ, Hildebrand MS, Shearer AE et al (2010) Frequency of usher syndrome in two pediatric populations: implications for genetic screening of deaf and hard of hearing children. Genet Med 12(8):512–516. https://doi.org/10.1097/GIM.0b013e3181e5afb8
Yan D, Liu XZ (2010) Genetics and pathological mechanisms of usher syndrome. J Hum Genet 55(6):327–335. https://doi.org/10.1038/jhg.2010.29
Millan JM, Aller E, Jaijo T et al (2011) An update on the genetics of usher syndrome. J Ophthalmol 417217:2011. https://doi.org/10.1155/2011/417217
Vache C, Besnard T, le Berre P et al (2012) Usher syndrome type 2 caused by activation of an USH2A pseudoexon: implications for diagnosis and therapy. Hum Mutat 33(1):104–108. https://doi.org/10.1002/humu.21634
Slijkerman RW, Vache C, Dona M et al (2016) Antisense oligonucleotide-based splice correction for ush2a-associated retinal degeneration caused by a frequent deep-intronic mutation. Mol Ther Nucleic Acids 5(10):e381. https://doi.org/10.1038/mtna.2016.89
Therapeutics P (2017) Innovation programs. http://www.proqr.com/innovation-programs/. Accessed 25 Sep 2017
Sakai LY, Keene DR, Morris NP et al (1986) Type VII collagen is a major structural component of anchoring fibrils. J Cell Biol 103(4):1577–1586
Turczynski S, Titeux M, Tonasso L et al (2016) Targeted exon skipping restores type VII collagen expression and anchoring fibril formation in an in vivo RDEB model. J Invest Dermatol 136(12):2387–2395. https://doi.org/10.1016/j.jid.2016.07.029
Murata T, Masunaga T, Ishiko A et al (2004) Differences in recurrent COL7A1 mutations in dystrophic epidermolysis bullosa: ethnic-specific and worldwide recurrent mutations. Arch Dermatol Res 295(10):442–447. https://doi.org/10.1007/s00403-003-0444-1
Gardella R, Castiglia D, Posteraro P et al (2002) Genotype-phenotype correlation in italian patients with dystrophic epidermolysis bullosa. J Invest Dermatol 119(6):1456–1462. https://doi.org/10.1046/j.1523-1747.2002.19606.x
Salas-Alanis JC, Amaya-Guerra M, McGrath JA (2000) The molecular basis of dystrophic epidermolysis bullosa in Mexico. Int J Dermatol 39(6):436–442
Csikos M, Szocs HI, Laszik A et al (2005) High frequency of the 425A-->G splice-site mutation and novel mutations of the COL7A1 gene in Central Europe: significance for future mutation detection strategies in dystrophic epidermolysis bullosa. Br J Dermatol 152(5):879–886. https://doi.org/10.1111/j.1365-2133.2005.06542.x
Tamai K, Murai T, Mayama M et al (1999) Recurrent COL7A1 mutations in Japanese patients with dystrophic epidermolysis bullosa: positional effects of premature termination codon mutations on clinical severity. Japanese collaborative study group on Epidermolysis Bullosa. J Invest Dermatol 112(6):991–993. https://doi.org/10.1046/j.1523-1747.1999.00601.x
Mohammedi R, Mellerio JE, Ashton GH et al (1999) A recurrent COL7A1 mutation, R2814X, in British patients with recessive dystrophic epidermolysis bullosa. Clin Exp Dermatol 24(1):37–39
Dang N, Murrell DF (2008) Mutation analysis and characterization of COL7A1 mutations in dystrophic epidermolysis bullosa. Exp Dermatol 17(7):553–568. https://doi.org/10.1111/j.1600-0625.2008.00723.x
Mellerio JE, Dunnill MG, Allison W et al (1997) Recurrent mutations in the type VII collagen gene (COL7A1) in patients with recessive dystrophic epidermolysis bullosa. J Invest Dermatol 109(2):246–249
Goto M, Sawamura D, Nishie W et al (2006) Targeted skipping of a single exon harboring a premature termination codon mutation: implications and potential for gene correction therapy for selective dystrophic epidermolysis bullosa patients. J Invest Dermatol 126(12):2614–2620. https://doi.org/10.1038/sj.jid.5700435
ProQR receives orphan drug designation from FDA for drug candidate QR-313 for dystrophic epidermolysis bullosa and will present data at two scientific conferences. (2017). https://globenewswire.com/news-release/2017/09/19/1124532/0/en/ProQR-Receives-Orphan-Drug-Designation-from-FDA-for-Drug-Candidate-QR-313-for-Dystrophic-Epidermolysis-Bullosa-and-will-Present-Data-at-two-Scientific-Conferences.html. Accessed Sep 2017
N.V PT (2017). http://www.proqr.com/qr-313-for-dystrophic-epidermolysis-bullosa/. Accessed Sep 2017
Galli SJ, Tsai M, Piliponsky AM (2008) The development of allergic inflammation. Nature 454(7203):445–454. https://doi.org/10.1038/nature07204
Brown JM, Wilson TM, Metcalfe DD (2008) The mast cell and allergic diseases: role in pathogenesis and implications for therapy. Clin Exp Allergy 38(1):4–18. https://doi.org/10.1111/j.1365-2222.2007.02886.x
Gilfillan AM, Tkaczyk C (2006) Integrated signalling pathways for mast-cell activation. Nat Rev Immunol 6(3):218–230. https://doi.org/10.1038/nri1782
Ishibashi K, Suzuki M, Sasaki S et al (2001) Identification of a new multigene four-transmembrane family (MS4A) related to CD20, HTm4 and beta subunit of the high-affinity IgE receptor. Gene 264(1):87–93
Cruse G, Kaur D, Leyland M et al (2010) A novel FcepsilonRIbeta-chain truncation regulates human mast cell proliferation and survival. FASEB J 24(10):4047–4057. https://doi.org/10.1096/fj.10-158378
Ra C, Jouvin MH, Kinet JP (1989) Complete structure of the mouse mast cell receptor for IgE (fc epsilon RI) and surface expression of chimeric receptors (rat-mouse-human) on transfected cells. J Biol Chem 264(26):15323–15327
Dombrowicz D, Lin S, Flamand V et al (1998) Allergy-associated FcRbeta is a molecular amplifier of IgE- and IgG-mediated in vivo responses. Immunity 8(4):517–529
Cruse G, Beaven MA, Ashmole I et al (2013) A truncated splice-variant of the FcepsilonRIbeta receptor subunit is critical for microtubule formation and degranulation in mast cells. Immunity 38(5):906–917. https://doi.org/10.1016/j.immuni.2013.04.007
Donnadieu E, Jouvin MH, Rana S et al (2003) Competing functions encoded in the allergy-associated F(c)epsilonRIbeta gene. Immunity 18(5):665–674
Cruse G, Yin Y, Fukuyama T et al (2016) Exon skipping of FcepsilonRIbeta eliminates expression of the high-affinity IgE receptor in mast cells with therapeutic potential for allergy. Proc Natl Acad Sci U S A 113(49):14115–14120. https://doi.org/10.1073/pnas.1608520113
Pignolo RJ, Kaplan FS (2017) Clinical staging of fibrodysplasia ossificans progressiva (FOP). Bone 109:111–114. https://doi.org/10.1016/j.bone.2017.09.014
Kaplan FS, Chakkalakal SA, Shore EM (2012) Fibrodysplasia ossificans progressiva: mechanisms and models of skeletal metamorphosis. Dis Model Mech 5(6):756–762. https://doi.org/10.1242/dmm.010280
Miao J, Zhang C, Wu S et al (2012) Genetic abnormalities in fibrodysplasia ossificans progressiva. Genes Genet Syst 87(4):213–219
Shi S, Cai J, de Gorter DJ et al (2013) Antisense-oligonucleotide mediated exon skipping in activin-receptor-like kinase 2: inhibiting the receptor that is overactive in fibrodysplasia ossificans progressiva. PLoS One 8(7):e69096. https://doi.org/10.1371/journal.pone.0069096
Shore EM, Xu M, Feldman GJ et al (2006) A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat Genet 38(5):525–527. https://doi.org/10.1038/ng1783
van Dinther M, Visser N, de Gorter DJ et al (2010) ALK2 R206H mutation linked to fibrodysplasia ossificans progressiva confers constitutive activity to the BMP type I receptor and sensitizes mesenchymal cells to BMP-induced osteoblast differentiation and bone formation. J Bone Miner Res 25(6):1208–1215. https://doi.org/10.1359/jbmr.091110
Kaplan FS, Xu M, Seemann P et al (2009) Classic and atypical fibrodysplasia ossificans progressiva (FOP) phenotypes are caused by mutations in the bone morphogenetic protein (BMP) type I receptor ACVR1. Hum Mutat 30(3):379–390. https://doi.org/10.1002/humu.20868
Lounev VY, Ramachandran R, Wosczyna MN et al (2009) Identification of progenitor cells that contribute to heterotopic skeletogenesis. J Bone Joint Surg Am 91(3):652–663. https://doi.org/10.2106/JBJS.H.01177
Medici D, Shore EM, Lounev VY et al (2010) Conversion of vascular endothelial cells into multipotent stem-like cells. Nat Med 16(12):1400–1406. https://doi.org/10.1038/nm.2252
Shi S, de Gorter DJ, Hoogaars WM et al (2013) Overactive bone morphogenetic protein signaling in heterotopic ossification and Duchenne muscular dystrophy. Cell Mol Life Sci 70(3):407–423. https://doi.org/10.1007/s00018-012-1054-x
Leblanc E, Trensz F, Haroun S et al (2011) BMP-9-induced muscle heterotopic ossification requires changes to the skeletal muscle microenvironment. J Bone Miner Res 26(6):1166–1177. https://doi.org/10.1002/jbmr.311
Dang ZC, van Bezooijen RL, Karperien M et al (2002) Exposure of KS483 cells to estrogen enhances osteogenesis and inhibits adipogenesis. J Bone Miner Res 17(3):394–405. https://doi.org/10.1359/jbmr.2002.17.3.394
de Gorter DJ, van Dinther M, Korchynskyi O et al (2011) Biphasic effects of transforming growth factor beta on bone morphogenetic protein-induced osteoblast differentiation. J Bone Miner Res 26(6):1178–1187. https://doi.org/10.1002/jbmr.313
Guncay A, Yokota T (2015) Antisense oligonucleotide drugs for Duchenne muscular dystrophy: how far have we come and what does the future hold? Future Med Chem 7(13):1631–1635. https://doi.org/10.4155/fmc.15.116
Khorkova O, Wahlestedt C (2017) Oligonucleotide therapies for disorders of the nervous system. Nat Biotechnol 35(3):249–263. https://doi.org/10.1038/nbt.3784
Goyenvalle A, Leumann C, Garcia L (2016) Therapeutic potential of Tricyclo-DNA antisense oligonucleotides. J Neuromuscul Dis 3(2):157–167. https://doi.org/10.3233/JND-160146
Goyenvalle A, Griffith G, Babbs A et al (2015) Functional correction in mouse models of muscular dystrophy using exon-skipping tricyclo-DNA oligomers. Nat Med 21(3):270–275. https://doi.org/10.1038/nm.3765
Relizani K, Griffith G, Echevarria L et al (2017) Efficacy and safety profile of tricyclo-DNA antisense oligonucleotides in duchenne muscular dystrophy mouse model. Mol Ther Nucleic Acids 8:144–157. https://doi.org/10.1016/j.omtn.2017.06.013
Cao L, Han G, Lin C et al (2016) Fructose promotes uptake and activity of oligonucleotides with different chemistries in a context-dependent manner in mdx mice. Mol Ther Nucleic Acids 5(6):e329. https://doi.org/10.1038/mtna.2016.46
Han G, Gu B, Cao L et al (2016) Hexose enhances oligonucleotide delivery and exon skipping in dystrophin-deficient mdx mice. Nat Commun 7:10981. https://doi.org/10.1038/ncomms10981
Coley WD, Bogdanik L, Vila MC et al (2016) Effect of genetic background on the dystrophic phenotype in mdx mice. Hum Mol Genet 25(1):130–145. https://doi.org/10.1093/hmg/ddv460
Fukada S, Morikawa D, Yamamoto Y et al (2010) Genetic background affects properties of satellite cells and mdx phenotypes. Am J Pathol 176(5):2414–2424. https://doi.org/10.2353/ajpath.2010.090887
Rodrigues M, Echigoya Y, Fukada S et al (2016) Current translational research and murine models for duchenne muscular dystrophy. J Neuromuscul Dis 3(1):29–48
Rodrigues M, Echigoya Y, Maruyama R et al (2016) Impaired regenerative capacity and lower revertant fibre expansion in dystrophin-deficient mdx muscles on DBA/2 background. Sci Rep 6:38371. https://doi.org/10.1038/srep38371
Nakamura A, Fueki N, Shiba N et al (2016) Deletion of exons 3-9 encompassing a mutational hot spot in the DMD gene presents an asymptomatic phenotype, indicating a target region for multiexon skipping therapy. J Hum Genet 61(7):663–667. https://doi.org/10.1038/jhg.2016.28
Echigoya Y, Yokota T (2014) Skipping multiple exons of dystrophin transcripts using cocktail antisense oligonucleotides. Nucleic Acid Ther 24(1):57–68. https://doi.org/10.1089/nat.2013.0451
Acknowledgments
This work was supported by the Friends of Garrett Cumming Research Chair Fund, the HM Toupin Neurological Science Research Chair Fund, the Muscular Dystrophy Canada, Canada Foundation for Innovation (CFI), Alberta Advanced Education and Technology (AET), Canadian Institutes of Health Research (CIHR), Jesse’s Journey - The Foundation for Gene and Cell Therapy, the University of Alberta Faculty of Medicine and Dentistry, and the Women and Children’s Health Research Institute (WCHRI).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Rodrigues, M., Yokota, T. (2018). An Overview of Recent Advances and Clinical Applications of Exon Skipping and Splice Modulation for Muscular Dystrophy and Various Genetic Diseases. In: Yokota, T., Maruyama, R. (eds) Exon Skipping and Inclusion Therapies. Methods in Molecular Biology, vol 1828. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8651-4_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8651-4_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8650-7
Online ISBN: 978-1-4939-8651-4
eBook Packages: Springer Protocols