Abstract
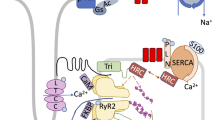
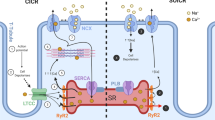
Regulation of Calcium (Ca) cycling by the sarcoplasmic reticulum (SR) underlies the control of cardiac contraction during excitation-contraction (E-C) coupling. Moreover, alterations in E-C coupling occurring in cardiac hypertrophy and heart failure are characterized by abnormal Ca-cycling through the SR network. A large body of evidence points to the central role of: a) SERCA and its regulator phospholamban (PLN) in the modulation of cardiac relaxation; b) calsequestrin in the regulation of SR Ca-load; and c) the ryanodine receptor (RyR) Ca-channel in the control of SR Ca-release. The levels or activity of these key Ca-handling proteins are altered in cardiomyopathies, and these changes have been linked to the deteriorated cardiac function and remodeling. Furthermore, genetic variants in these SR Ca-cycling proteins have been identified, which may predispose to heart failure or fatal arrhythmias. This chapter concentrates on the pivotal role of SR Ca-cycling proteins in health and disease with specific emphasis on their recently reported genetic modifiers
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Anderson, M.E., Braun, A.P., Schulman, H., et al., 1994, Multifunctional Ca 2+ /calmodulin-dependent protein kinase mediates Ca(2+)-induced enhancement of the L-type Ca 2+ current in rabbit ventricular myocytes. Circ Res, 75(5), pp854–61.
Bassani, J.W., Yuan, W., andBers, D.M., 1995, Fractional SR Ca release is regulated by trigger Ca and SR Ca content in cardiac myocytes. Am J Physiol, 268(5 Pt 1), ppC1313–9.
Bers, D.M., 2001,Excitation-Contraction Coupling and Cardiac Contractile Force. 2nd ed: Kluwer Academic Press, Boston, Massachusetts.
Bossuyt, J., Ai, X., Moorman, J.R., et al., 2005, Expression and phosphorylation of the na-pump regulatory subunit phospholemman in heart failure. Circ Res, 97(6), pp558–65.
Brittsan, A.G., andKranias, E.G., 2000, Phospholamban and cardiac contractile function. J. Mol. Cell. Cardiol., 32, pp2131–2139.
Dash, R., Frank, K.F., Carr, A.N., et al., 2001, Gender influences on sarcoplasmic reticulum Ca 2+ -handling in failing human myocardium. J Mol Cell Cardiol, 33(7), pp1345–53.
Dash, R., Kadambi, V., Schmidt, A.G., et al., 2001, Interactions between phospholamban and beta-adrenergic drive may lead to cardiomyopathy and early mortality. Circulation, 103(6), pp889–96.
de Tombe, P.P., 1998, Altered contractile function in heart failure. Cardiovasc Res, 37(2), pp367–80.
del Monte, F., Harding, S.E., Schmidt, U., et al., 1999, Restoration of contractile function in isolated cardiomyocytes from failing human hearts by gene transfer of SERCA2a. Circulation, 100, pp2308–11.
Desai, K.H., Schauble, E., Luo, W., et al., 1999, Phospholamban deficiency does not compromise exercise capacity. Amer. J. Physiol, 276(4), ppH1172–7.
Farrell, E.F., Antaramian, A., Rueda, A., et al., 2003, Sorcin inhibits calcium release and modulates excitation-contraction coupling in the heart. J Biol Chem, 278, pp34660–66.
Franz, W.M., Muller, O.J., andKatus, H.A., 2001. Cardiomyopathies: from genetics to the prospect of treatment. Lancet, 358, pp1627–1637.
Garvey, J.L., Kranias, E.G., andSolaro, R.J., 1988, Phosphorylation of C-protein, troponin I and phospholamban in isolated rabbit hearts. Biochem J, 249(3), pp709–14.
Ginsburg, K.S., andBers, D.M., 2004, Modulation of excitation-contraction coupling by isoproterenol in cardiomyocytes with controlled SR Ca load and I_Ca trigger. J Physiol, 556(Pt 2), pp463–80.
Go, L.O., Moschella, M.C., Watras, J., et al. 1995, Differential regulation of two types of intracellular calcium release channels during end-stage heart failure. J Clin Invest, 95(2), pp888–94.
Guo, T., Zhang, T., Mestril, R., Bers, D.M., 2006, Ca/Calmodulin-Dependent Protein Kinase II Phosphorylation of Ryanodine Receptor Does Affect Calcium Sparks in Mouse Ventricular Myocytes. Circ Res, 99(4), pp398–406.
Gyorke, I., Hester, N., Jones, L.R., Gyorke, S., 2004, The role of calsequestrin, triadin, and junctin in conferring cardiac ryanodine receptor responsiveness to luminal calcium. Biophys J., 86(4), pp2121–8.
Hasenfuss, G. 1998, Alterations of calcium-regulatory proteins in heart failure. Cardiovasc Res, 37(2), pp279–89.
Hobai, I.A., O’Rourke, B. 2001, Decreased sarcoplasmic reticulum calcium content is responsible for defective excitation-contraction coupling in canine heart failure. Circulation, 103(11), pp1577–84.
Hoit, B.D., Khoury, S.F., Kranias, E.G., et al., 1995, In vivo echocardiographic detection of enhanced left ventricular function in gene-targeted mice with phospholamban deficiency. Circ Res,77(3), pp632–7.
Houser, S.R., Piacentino, V. 3rd., Mattiello, J., et al., 2000, Functional properties of failing human ventricular myocytes. Trends Cardiovasc Med, 10(3), pp101–7.
Huang, B., Wang, S., Qin, D., et al., 1999, Diminished basal phosphorylation level of phospholamban in the postinfarction remodeled rat ventricle: role of beta-adrenergic pathway, G(i) protein, phosphodiesterase, and phosphatases. Circ Res, 85(9), pp848–55.
Kadambi, V.J., Ponniah, S., Harrer, J.M., et al., 1996, Cardiac-specific overexpression of phospholamban alters calcium kinetics and resultant cardiomyocyte mechanics in transgenic mice. J Clin Invest, 97(2), pp533–9.
Kentish, J.C., McCloskey, D.T., Layland, J., et al. 2001, Phosphorylation of troponin I by protein kinase A accelerates relaxation and crossbridge cycle kinetics in mouse ventricular muscle. Circ Res, 88(10), pp1059–65.
Kranias, E.G., andSolaro, R.J., 1982, Phosphorylation of troponin I and phospholamban during catecholamine stimulation of rabbit heart. Nature, 298(5870), pp182–4.
Kranias, E.G., Steenaart, N.A., andDi Salvo, J., 1988, Purification and characterization of phospholamban phosphatase from cardiac muscle. J Biol Chem, 263(30), pp15681–7.
Leenhardt, A., Lucet, V., Denjoy, I., et al., 1995, Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients. Circulation, 91(5), pp1512–9.
Lehnart, S.E., Wehrens, X.H., Laitinen, P.J., et al., 2004, Sudden death in familial polymorphic ventricular tachycardia associated with calcium release channel (ryanodine receptor) leak. Circulation, 109(25), pp3208–14.
Li, L., Chu, G., Kranias, E.G., et al., 1998, Cardiac myocyte calcium transport in phospholamban knockout mouse: relaxation and endogenous CaMKII effects. Am J Physiol, 274(4 Pt 2), ppH1335–47.
Li, L., Desantiago, J., Chu, G., et al., 2000, Phosphorylation of phospholamban and troponin I in beta-adrenergic-induced acceleration of cardiac relaxation. Am J Physiol Heart Circ Physiol, 278(3), ppH769–79.
Li, L., Satoh, H., Ginsburg, K.S., et al., 1997, The effect of Ca(2+)-calmodulin-dependent protein kinase II on cardiac excitation-contraction coupling in ferret ventricular myocytes. J Physiol. 501(Pt 1), pp17–31.
Li, Y., Kranias, E.G., Mignery, G.A., et al., 2002, Protein kinase A phosphorylation of the ryanodine receptor does not affect calcium sparks in mouse ventricular myocytes. Circ Res, 90(3), pp309–16.
Lindemann, J.P., Jones, L.R., Hathaway, D.R., et al., 1983, beta-Adrenergic stimulation of phospholamban phosphorylation and Ca 2+ -ATPase activity in guinea pig ventricles. J Biol Chem, 258(1), pp464–71.
Lindner, M., Erdmann, E., andBeuckelmann, D.J., 1998, Calcium content of the sarcoplasmic reticulum in isolated ventricular myocytes from patients with terminal heart failure. J Mol Cell Cardiol, 30(4), pp743–9.
Lorenz, J.N., andKranias, E.G., 1997, Regulatory effects of phospholamban on cardiac function in intact mice. Am J Physiol, 273(6 Pt 2), ppH2826–31.
Luo, W., Grupp, I.L., Harrer, J., et al., 1994, Targeted ablation of the phospholamban gene is associated with markedly enhanced myocardial contractility and loss of beta-agonist stimulation. Circ Res, 75(3), pp401–9.
Luo, W., Wolska, B.M., Grupp, I.L., et al., 1996, Phospholamban gene dosage effects in the mammalian heart. Circ Res, 78(5), pp839–47.
Luo, W., Chu, G., Sato, Y., et al., 1998, Transgenic approaches to define the functional role of dual site phospholamban phosphorylation. J. Biol. Chem., 273, pp4734–4739.
MacLennan, D.H., andKranias, E.G., 2003, Phospholamban: a crucial regulator of cardiac contractility. Nat Rev Mol Cell Biol, 4(7), pp566–77.
Maier, L.S., Zhang, T., Chen, L., et al., 2003, Transgenic CaMKIIdeltaC overexpression uniquely alters cardiac myocyte Ca 2+ handling: reduced SR Ca 2+ load and activated SR Ca 2+ release. Circ Res, 92(8), pp904–11.
Marx, S.O., Reiken, S., Hisamatsu, Y. et al., 2000, PKA phosphorylation dissociates FKBP12.6 from the calcium release channel (ryanodine receptor): defective regulation in failing hearts. Cell, 101(4), pp365–76.
McCall, E., Li, L., Satoh, H., et al., 1996, Effects of FK-506 on contraction and Ca 2+ transients in rat cardiac myocytes. Circ Res, 79(6), pp1110–21.
McTiernan, C.F., Frye, C.S., Lemster, B.H., et al., 1999, The human phospholamban gene: structure and expression. J. Mol. Cell. Cardiol, 31, pp679–692.
Minamisawa, S., Sato, Y., Tatsuguchi, et al., 2003, Mutation of the phospholamban promoter associated with hypertrophic cardiomyopathy. Biochem Biophys Res Commun, 304(1), pp1–4.
Mitchell, R.D., Simmerman, H.K., andJones, L.R., 1988, Ca 2+ binding effects on protein conformation and protein interactions of canine cardiac calsequestrin. J Biol Chem, 263(3), pp1376–81.
Miyamoto, M.I., del Monte, F., Schmidt, U., et al., 2000, Adenoviral gene transfer of SERCA2a improves left-ventricular function in aortic-banded rats in transition to heart failure. Proc Natl Acad Sci U S A, 97(2), pp793–8.
Mukherjee, R., andSpinale, F.G., 1998, L-type calcium channel abundance and function with cardiac hypertrophy and failure: a review. J Mol Cell Cardiol, 30(10), pp1899–916.
Mundina-Weilenmann, C., Vittone, L., Ortale, M., et al. 1996, Immunodetection of phosphorylation sites gives new insights into the mechanisms underlying phospholamban phosphorylation in the intact heart. J Biol Chem, 271(52), pp33561–7.
Nabauer, M., andKaab, S., 1998, Potassium channel down-regulation in heart failure. Cardiovasc Res, 37(2), pp324–34.
Phillips, R.M., Narayan, P., Gomez, A.M., et al., 1998, Sarcoplasmic reticulum in heart failure: central player or bystander? Cardiovasc Res, 37(2), pp346–51.
Pi, Y., Kemnitz, K.R., Zhang, D., et al., 2002, Phosphorylation of troponin I controls cardiac twitch dynamics: evidence from phosphorylation site mutants expressed on a troponin I-null background in mice. Circ Res, 90(6), pp649–56.
Piacentino, V. 3rd, Weber, C.R., Chen, X., et al., 2003, Cellular basis of abnormal calcium transients of failing human ventricular myocytes. Circ Res., 92(6), pp651–8.
Pogwizd, S.M., Qi, M., Yuan, W., et al., 1999, Upregulation of Na(+)/Ca(2+) exchanger expression and function in an arrhythmogenic rabbit model of heart failure. Circ Res, 85(11), pp1009–19.
Pogwizd, S.M., Schlotthauer, K., Li, L., et al., 2001, Arrhythmogenesis and contractile dysfunction in heart failure: Roles of sodium-calcium exchange, inward rectifier potassium current, and residual beta-adrenergic responsiveness. Circ Res., 88(11), pp1159–67.
Richard, S., Leclercq, F., Lemaire, S., et al., 1998, Ca 2+ currents in compensated hypertrophy and heart failure. Cardiovasc Res, 37(2), pp300–11.
Sainte, Beuve C., Allen, P.D., Dambrin, G., et al., 1997, Cardiac calcium release channel (ryanodine receptor) in control and cardiomyopathic human hearts: mRNA and protein contents are differentially regulated. J Mol Cell Cardiol., 29(4), pp1237–46.
Sato, Y., Kiriazis, H., Yatani, A., et al., 2001, Rescue of contractile parameters and myocyte hypertrophy in calsequestrin overexpressing myocardium by phospholamban ablation. J. Biol. Chem., 276(12), pp9392–9,
Schillinger, W., Meyer, M., Kuwajima, G., et al., 1996, Unaltered ryanodine receptor protein levels in ischemic cardiomyopathy. Mol Cell Biochem, 160–161, pp297–302.
Schmidt, A.G., Haghighi, K., Frank, B., et al., 2003, Polymorphic SERCA2a variants do not account for inter-individual differences in phospholamban –SERCA2a interaction in human heart failure. J. Mol. Cell. Cardiol, 35, pp867–870.
Schmitt, J.P., Kamisago, M., Asahi, M., et al., 2003, Dilated cardiomyopathy and heart failure caused by a mutation in phospholamban. Science, 299(5611), pp1410–1413.
Schwinger, R.H., Munch, G., Bolck, B., et al., 1999, Reduced Ca(2+)-sensitivity of SERCA 2a in failing human myocardium due to reduced serin-16 phospholamban phosphorylation. J Mol Cell Cardiol, 31(3), pp479–91.
Shannon, T.R., Ginsburg, K.S., andBers, D.M., 2000, Potentiation of fractional sarcoplasmic reticulum calcium release by total and free intra-sarcoplasmic reticulum calcium concentration. Biophys J, 78(1), pp334–43.
Simmerman, H.K., andJones, L.R., 1998, Phospholamban: protein structure, mechanism of action, and role in cardiac function. Physiol Rev, 78(4), pp921–47.
Solaro, R.J., andBriggs, F.N., 1974, Estimating the functional capabilitities of sarcoplasmic reticulum in cardiac muscle. Circ. Res., 34, pp531–540.
Stange, M., Xu, L., Balshaw, D., et al., 2003, Characterization of recombinant skeletal muscle (Ser-., 2843) and cardiac muscle (Ser-., 2809) ryanodine receptor phosphorylation mutants. J Biol Chem, 278(51), pp51693–702.
Talosi, L., Edes. I., andKranias, E.G., 1993, Intracellular mechanisms mediating reversal of beta-adrenergic stimulation in intact beating hearts. Am J Physiol, 264(3 Pt 2), ppH791–7.
Valdivia, H.H., Kaplan, J.H., Ellis-Davies, G.C., et al., 1995, Rapid adaptation of cardiac ryanodine receptors: modulation by Mg2+ and phosphorylation. Science, 267, pp997–2000.
Vatner, D.E., Sato, N., Kiuchi, K., et al., 1994, Decrease in myocardial ryanodine receptors and altered excitation-contraction coupling early in the development of heart failure. Circulation, 90(3), pp1423–30.
Wang, J., Maertz, N.A., Lokua, A.J., et al., 2001, Regulation of cardiac Ryanodine Receptors activity by calsequestrin. Biophys J, 80, pp590a.
Wegener, A.D., Simmerman, H.K., Lindemann, J.P., et al., 1989, Phospholamban phosphorylation in intact ventricles. Phosphorylation of serine 16 and threonine 17 in response to beta-adrenergic stimulation. J Biol Chem, 264(19), pp11468–74.
Wehrens, X.H., Lehnart, S.E., Huang, F., et al., 2003, FKBP12.6 deficiency and defective calcium release channel (ryanodine receptor) function linked to exercise-induced sudden cardiac death. Cell, 113(7), pp829–40.
Wickenden, A.D., Kaprielian, R., Kassiri, Z., et al., 1998, The role of action potential prolongation and altered intracellular calcium handling in the pathogenesis of heart failure. Cardiovasc Res, 37(2), pp312–23.
Wolska, B.M., Stojanovic, M.O., Luo, W., et al., 1996, Effect of ablation of phospholamban on dynamics of cardiac myocyte contraction and intracellular Ca 2+ . Am J Physiol, 271(1 Pt 1), ppC391–7.
Xiao, B., Sutherland, C., Walsh, M.P., et al. 2004, Protein kinase A phosphorylation at serine-2808 of the cardiac Ca 2+ -release channel (ryanodine receptor) does not dissociate 12.6-kDa FK506-binding protein (FKBP12.6). Circ Res, 94(4), pp487–95.
Xiao, R.P., Cheng, H., Lederer, W.J., et al. 1994, Dual regulation of Ca 2+/calmodulin-dependent kinase II activity by membrane voltage and by calcium influx. Proc Natl Acad Sci U S A. 91(20), pp9659–63.
Yano, M., Ono, K., Ohkusa, T., et al. 2000, Altered stoichiometry of FKBP12.6 versus ryanodine receptor as a cause of abnormal Ca(2+) leak through ryanodine receptor in heart failure. Circulation, 102(17), pp2131–6.
Yuan, W., Bers, D.M. 1994, Ca-dependent facilitatiosn of cardiac Ca current is due to Ca-calmodulin-dependent protein kinase. Am J Physiol. 267(3 Pt 2), ppH982–93.
Zhang, L., Kelley, J., Schmeisser, G., et al. 1997, Complex formation between junctin, triadin, calsequestrin, and the ryanodine receptor. Proteins of the cardiac junctional sarcoplasmic reticulum membrane. J Biol Chem, 272(37), pp23389–97.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Springer
About this chapter
Cite this chapter
KRANIAS, E., BERS, D. (2007). Calcium and Cardiomyopathies. In: Carafoli, E., Brini, M. (eds) Calcium Signalling and Disease. Subcellular Biochemistry, vol 45. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-6191-2_20
Download citation
DOI: https://doi.org/10.1007/978-1-4020-6191-2_20
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-6190-5
Online ISBN: 978-1-4020-6191-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)