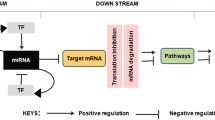
Abstract
MicroRNAs exert their effects in the context of gene regulatory networks. The recent development of high-throughput experimental approaches and the growing availability of gene expression data have permitted comprehensive functional studies of miRNAs. However, the data interpretation is often challenging due to the fact that miRNAs not only act cooperatively with other miRNAs but also participate in complex networks by interacting with other functional elements, including non-coding RNAs or transcription factors that often have extensive effects on cell biology. This chapter provides detailed practical procedures on how to use miRNet 2.0 (https://www.mirnet.ca) to perform miRNA regulatory network analytics to gain functional insights.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bracken CP, Scott HS, Goodall GJ (2016) A network-biology perspective of microRNA function and dysfunction in cancer. Nature Reviews Genetics 17. https://doi.org/10.1038/nrg.2016.134
Anastasiadou E, Jacob LS, Slack FJ (2017) Non-coding RNA networks in cancer. Nature Reviews Cancer 18. https://doi.org/10.1038/nrc.2017.99
Fan Y, Siklenka K, Arora SK, Ribeiro P, Kimmins S, Xia JJ (2016) miRNet-dissecting miRNA-target interactions and functional associations through network-based visual analysis. Nucleic Acids Res 44(W1):W135–W141
Fan Y, Habib M, Xia J (2018) Xeno-mirNet: a comprehensive database and analytics platform to explore xeno-miRNAs and their potential targets. PeerJ 2018. https://doi.org/10.7717/peerj.5650
Fan Y, Xia J (2018) miRNet—functional analysis and visual exploration of miRNA–target interactions in a network context. In: Computational cell biology. Springer, pp 215–233
Chang L, Zhou G, Soufan O, Xia J (2020) miRNet 2.0: network-based visual analytics for miRNA functional analysis and systems biology. Nucleic Acids Res 48(W1):W244–W251. https://doi.org/10.1093/nar/gkaa467
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (2011) A ceRNA hypothesis: the Rosetta stone of a hidden RNA language? Cell 146(3):353–358
Zhu X, Gerstein M, Snyder M (2007) Getting connected: analysis and principles of biological networks. Genes Dev 21(9):1010–1024
Nuzziello N, Vilardo L, Pelucchi P, Consiglio A, Liuni S, Trojano M, Liguori MJ (2018) Investigating the role of MicroRNA and transcription factor co-regulatory networks in multiple sclerosis pathogenesis. Ijoms 19(11):3652
Jacomy M, Venturini T, Heymann S, Bastian M (2014) ForceAtlas2, a continuous graph layout algorithm for handy network visualization designed for the Gephi software. PLoS One 9(6):e98679. https://doi.org/10.1371/journal.pone.0098679
Karagkouni D, Paraskevopoulou MD, Chatzopoulos S, Vlachos IS, Tastsoglou S, Kanellos I, Papadimitriou D, Kavakiotis I, Maniou S, Skoufos G (2018) DIANA-TarBase v8: a decade-long collection of experimentally supported miRNA–gene interactions. Nucleic Acids Res 46(D1):D239–D245
Huang H-Y, Lin Y-C-D, Li J, Huang K-Y, Shrestha S, Hong H-C, Tang Y, Chen Y-G, Jin C-N, Yu Y (2020) miRTarBase 2020: updates to the experimentally validated microRNA–target interaction database. Nucleic Acids Res 48(D1):D148–D154
Xiao F, Zuo Z, Cai G, Kang S, Gao X, Li T (2009) miRecords: an integrated resource for microRNA–target interactions. Nucleic Acids Res 37(suppl_1):D105–D110
Betel D, Koppal A, Agius P, Sander C, Leslie C (2010) Comprehensive modeling of microRNA targets predicts functional non-conserved and non-canonical sites. Genome Biol 11(8):R90
Li J-H, Liu S, Zhou H, Qu L-H, Yang J-H (2014) starBase v2. 0: decoding miRNA-ceRNA, miRNA-ncRNA and protein–RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res 42(D1):D92–D97
Jiang Q, Wang Y, Hao Y, Juan L, Teng M, Zhang X, Li M, Wang G, Liu Y (2009) miR2Disease: a manually curated database for microRNA deregulation in human disease. Nucleic Acids Res 37(suppl_1):D98–D104
Huang Z, Shi J, Gao Y, Cui C, Zhang S, Li J, Zhou Y, Cui Q (2019) HMDD v3. 0: a database for experimentally supported human microRNA–disease associations. Nucleic Acids Res 47(D1):D1013–D1017
Ruepp A, Kowarsch A, Schmidl D, Buggenthin F, Brauner B, Dunger I, Fobo G, Frishman G, Montrone C, Theis FJ (2010) PhenomiR: a knowledgebase for microRNA expression in diseases and biological processes. Genome Biol 11(1):R6
Liu X, Wang S, Meng F, Wang J, Zhang Y, Dai E, Yu X, Li X, Jiang W (2013) SM2miR: a database of the experimentally validated small molecules’ effects on microRNA expression. Bioinformatics 29(3):409–411
Rukov JL, Wilentzik R, Jaffe I, Vinther J, Shomron N (2014) Pharmaco-miR: linking microRNAs and drug effects. Brief Bioinform 15(4):648–659
Dai E, Yu X, Zhang Y, Meng F, Wang S, Liu X, Liu D, Wang J, Li X, Jiang W (2014) EpimiR: a database of curated mutual regulation between miRNAs and epigenetic modifications. Database (Oxford) 2014(2014):bau023. https://doi.org/10.1093/database/bau023
Tong Z, Cui Q, Wang J, Zhou Y (2019) TransmiR v2. 0: an updated transcription factor-microRNA regulation database. Nucleic Acids Res 47(D1):D253–D258
Oak N, Ghosh R, Kl H, Wheeler DA, Ding L, Plon SE (2019) Framework for microRNA variant annotation and prioritization using human population and disease datasets. Hum Mutat 40(1):73–89
Bhattacharya A, Ziebarth JD, Cui Y (2014) PolymiRTS database 3.0: linking polymorphisms in microRNAs and their target sites with human diseases and biological pathways. Nucleic Acids Res 42(D1):D86–D91
Xu T, Su N, Liu L, Zhang J, Wang H, Zhang W, Gui J, Yu K, Li J, Le TD (2018) miRBaseConverter: an R/Bioconductor package for converting and retrieving miRNA name, accession, sequence and family information in different versions of miRBase. BMC bioinformatics 19(19):179–188
Li J, Han X, Wan Y, Zhang S, Zhao Y, Fan R, Cui Q, Zhou Y (2018) TAM 2.0: tool for MicroRNA set analysis. Nucleic Acids Res 46(W1):W180–W185
Bleazard T, Lamb JA, Griffiths-Jones S (2015) Bias in microRNA functional enrichment analysis. Bioinformatics 31(10):1592–1598
Csardi G, Nepusz T (2006) The igraph software package for complex network research. Inter J Complex Syst 1695(5):1–9
Smyth GK (2005) Limma: linear models for microarray data. In: Bioinformatics and computational biology solutions using R and Bioconductor. Springer, pp 397–420
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140
Dvinge H, Bertone P (2009) HTqPCR: high-throughput analysis and visualization of quantitative real-time PCR data in R. Bioinformatics 25(24):3325–3326
Guo Z, Maki M, Ding R, Yang Y, Zhang B, Xiong L (2014) Genome-wide survey of tissue-specific microRNA and transcription factor regulatory networks in 12 tissues. Sci Rep 4:5150. https://doi.org/10.1038/srep05150
Palmieri V, Backes C, Ludwig N, Fehlmann T, Kern F, Meese E, Keller A (2018) IMOTA: an interactive multi-omics tissue atlas for the analysis of human miRNA-target interactions. Nucleic Acids Res 46(D1):D770–d775. https://doi.org/10.1093/nar/gkx701
Mathivanan S, Simpson RJ (2009) ExoCarta: a compendium of exosomal proteins and RNA. Proteomics 9:4997–5000. https://doi.org/10.1002/pmic.200900351
Acknowledgments
Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant, NSERC-CREATE-MATRIX Scholarship, and Canada Research Chairs (CRC) Program.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Chang, L., Xia, J. (2023). MicroRNA Regulatory Network Analysis Using miRNet 2.0. In: Song, Q., Tao, Z. (eds) Transcription Factor Regulatory Networks. Methods in Molecular Biology, vol 2594. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2815-7_14
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2815-7_14
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2814-0
Online ISBN: 978-1-0716-2815-7
eBook Packages: Springer Protocols