Abstract
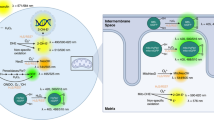
Multicellular organisms initiate adaptive responses when oxygen (O2) availability decreases. The underlying mechanisms of O2 sensing remain unclear. Mitochondria have been implicated in many hypoxia-inducible factor (HIF) -dependent and -independent hypoxic responses. However, the role of mitochondria in mammalian cellular O2 sensing has remained controversial, particularly regarding the use pharmacologic agents to effect hypoxic HIFα stabilization, which has produced conflicting data in the literature. Using murine embryonic cells lacking cytochrome c, we show that mitochondrial reactive O2 species (ROS) are essential for O2 sensing and subsequent HIFα stabilization at 1.5% O2. In the absence of this signal, HIFα subunits continue to be hydroxylated and degraded via the proteasome. Importantly, exogenous treatment with H2O2 and severe O2 deprivation is sufficient to stabilize HIFα even in the absence of functional mitochondrial. These results demonstrate that mitochondria function as O2 sensors and signal hypoxic HIFα stabilization by releasing ROS to the cytoplasm. The cytochrome c mutant embryonic cells provide a unique reagent to further dissect the role of mitochondria in O2 mediated-intracellular events.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Archer SL, Reeve HL, Michelakis E, Puttagunta L, Waite R, Nelson DP, Dinauer MC, and Weir EK. O2 sensing is preserved in mice lacking the gp91 phox subunit of NADPH oxidase. Proc Natl Acad Sci U S A 96: 7944–7949, 1999.
Chandel NS, Maltepe E, Goldwasser E, Mathieu CE, Simon MC, and Schumacker PT. Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA 95: 11715–11720, 1998.
Chandel NS, McClintock DS, Feliciano CE, Wood TM, Melendez JA, Rodriguez AM, and Schumacker PT. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-lalpha during hypoxia: a mechanism of O2 sensing. J Biol Chem 275: 25130–25138., 2000.
Epstein AC, Gleadle JM, McNeill LA, Hewitson KS, O’Rourke J, Mole DR, Mukherji M, Metzen E, Wilson MI, Dhanda A, Tian YM, Masson N, Hamilton DL, Jaakkola P, Barstead R, Hodgkin J, Maxwell PH, Pugh CW, Schofield CJ, and Ratcliffe PJ. C. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 107: 43–54, 2001.
Huang LE, Gu J, Schau M, and Bunn HE Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin-proteasome pathway. Proc Natl Acad Sci U S A 95: 7987–7992, 1998.
Ivan M, Kondo K, Yang H, Kim W, Valiando J, Ohh M, Salic A, Asara JM, Lane WS, and Kaelin WG, Jr. HIFalpha targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science 292: 464–468., 2001.
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, Kriegsheim A, Hebestreit HF, Mukherji M, Schofield CJ, Maxwell PH, Pugh CW, and Ratcliffe PJ. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292: 468–472., 2001.
Jones RD, Hancock JT, and Morice AH. NADPH oxidase: a universal oxygen sensor? Free Radie Biol Med 29: 416–424, 2000.
Mansfield KD, Guzy RD, Pan Y, Young RD, Schumacker PT, and Simon MC. Cytochrome C is required for cellular oxygen sensing and hypoxic HIF activation. Cell Metabolism, manuscript submitted, 2005.
Michiels C, Minet E, Mottet D, and Raes M. Regulation of gene expression by oxygen: NF-kappaB and HIF-1, two extremes. Free Radie Biol Med 33: 1231–1242, 2002.
Schroedl C, McClintock DS, Budinger GR, and Chandel NS. Hypoxic but not anoxic stabilization of HIF-1alpha requires mitochondrial reactive oxygen species. Am J Physiol Lung Cell Mol Physiol 283: L922–931., 2002.
Semenza GL. Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol 15: 551–578, 1999.
Srinivas V, Leshchinsky I, Sang N, King MP, Minchenko A, and Caro J. Oxygen sensing and HIF-1 activation does not require an active mitochondrial respiratory chain electron-transfer pathway. J Biol Chem 276: 21995–21998, 2001.
Vaux E, Metzen E, Yeates K, and Ratcliffe P. Regulation of hypoxia-inducible factor is preserved in the absence of a functioning mitochondrial respiratory chain. Blood 98: 296–302, 2001.
Zhu H and Bunn H. Oxygen Sensing and signaling: impact on the regulation of physiologically important genes. Respiration Physiology 115: 239–247, 1999.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer Science+Business Media, LLC
About this paper
Cite this paper
Celeste Simon, M. (2006). Mitochondrial Reactive Oxygen Species are Required for Hypoxic HIFα Stabilization. In: Roach, R.C., Wagner, P.D., Hackett, P.H. (eds) Hypoxia and Exercise. Advances in Experimental Medicine and Biology, vol 588. Springer, Boston, MA. https://doi.org/10.1007/978-0-387-34817-9_15
Download citation
DOI: https://doi.org/10.1007/978-0-387-34817-9_15
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-34816-2
Online ISBN: 978-0-387-34817-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)