20.1. Abstract
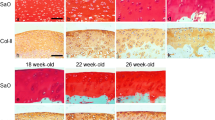
Objective of this study was to investigate the early wound healing reactions of immature and mature articular cartilage on experimental wound healing in the New Zealand White rabbit. The proliferation potential and glycosaminoglycan production of isolated chondrocytes of these animals was studied in an alginate culture system. A band of tissue with death chondrocytes was observed at wound edges of immature articular cartilage, whereas mature cartilage showed a significant smaller amount of dead chondrocytes. A general increase in TGFβ1, FGF2 and IGF1 was observed throughout cartilage tissue with the exception of lesion edges. The observed immunonegative area appeared to correlate with the observed cell death in lesion edges. Repair in immature cartilage was indicated by chondrocyte proliferation in clusters and a decrease in defect size. No repair response was observed in mature articular cartilage defects. The alginate culture experiment demonstrated a higher proliferation rate of immature chondrocytes. Addition of recombinant TGFβ1 increased proliferation rate and GAG production of mature chondrocytes. We were not able to further stimulate immature chondrocytes. These results indicate that TGF 1 addition may contribute to induce cartilage repair responses in mature cartilage as observed in immature, developing cartilage.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
20.7. References
Namba, R.S., Meuli, M., Sullivan, K.M., Le, A.X. & Adzick, N.S. Spontaneous repair of superficial defects in articular cartilage in a fetal lamb model. in J Bone Joint Surg Am, Vol. 80 4–10 (1998).
Buckwalter, J.A. & Mankin, H.J. Articular cartilage repair and transplantation. in Arthritis Rheum Vol. 41 1331–42 (1998).
Buckwalter, J.A. Were the Hunter brothers wrong? Can surgical treatment repair articular cartilage? in Iowa Orthop J Vol. 17 1–13 (1997).
Ahsan, T. & Sah, R.L. Biomechanics of integrative cartilage repair. in Osteoarthritis Cartilage Vol. 7 29–40 (1999).
Shapiro, F., Koide, S. & Glimcher, M.J. Cell origin and differentiation in the repair of full-thickness defects of articular cartilage. in J Bone Joint Surg Am Vol. 75 532–53 (1993).
Messner, K. & Gillquist, J. Cartilage repair. A critical review [see comments]. in Acta Orthop Scand Vol. 67 523–9 (1996).
Verwoerd-Verhoef, H.L., ten Koppel, P.G., van Osch, G.J., Meeuwis, C.A. & Verwoerd, C.D. Wound healing of cartilage structures in the head and neck region. in Int J Pediatr Otorhinolaryngol Vol. 43 241–51 (1998).
O’Driscoll, S.W., Keeley, F.W. & Salter, R.B. Durability of regenerated articular cartilage produced by free autogenous periosteal grafts in major full-thickness defects in joint surfaces under the influence of continuous passive motion. A follow-up report at one year. in J Bone Joint Surg [Am] Vol. 70 595–606 (1988).
Amiel, D. et al. Rib perichondrial grafts for the repair of full-thickness articular-cartilage defects. A morphological and biochemical study in rabbits. in J Bone Joint Surg [Am] Vol. 67 911–20 (1985).
Tew, S.R., Kwan, A.P., Hann, A., Thomson, B.M. & Archer, C.W. The reactions of articular cartilage to experimental wounding: role of apoptosis. in Arthritis Rheum Vol. 43 215–25 (2000).
Desjardins, M.R., Hurtig, M.B. & Palmer, N.C. Heterotopic transfer of fresh and cryopreserved autogenous articular cartilage in the horse. in Vet Surg Vol. 20 434–45 (1991).
Wakitani, S. et al. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. in J Bone Joint Surg Am Vol. 76 579–92 (1994).
Giurea, A., DiMicco, M.A., Akeson, W.H. & Sah, R.L. Development-associated differences in integrative cartilage repair: roles of biosynthesis and matrix. in J Orthop Res Vol. 20 1274–81 (2002).
Wei, X. & Messner, K. Age-and injury-dependent concentrations of transforming growth factor-beta 1 and proteoglycan fragments in rabbit knee joint fluid. in Osteoarthritis Cartilage Vol. 6 10–8 (1998).
Guerne, P.A., Blanco, F., Kaelin, A., Desgeorges, A. & Lotz, M. Growth factor responsiveness of human articular chondrocytes in aging and development. in Arthritis Rheum Vol. 38 960–8 (1995).
Barbero, A. et al. Age related changes in human articular chondrocyte yield, proliferation and post-expansion chondrogenic capacity. in Osteoarthritis Cartilage Vol. 12 476–84 (2004).
Rosen, F. et al. Differential effects of aging on human chondrocyte responses to transforming growth factor beta: increased pyrophosphate production and decreased cell proliferation. in Arthritis Rheum Vol. 40 1275–81 (1997).
Kaweblum, M. et al. Histological and radiographic determination of the age of physeal closure of the distal femur, proximal tibia, and proximal fibula of the New Zealand white rabbit. in J Orthop Res Vol. 12 747–9 (1994).
van Osch, G.J., van den Berg, W.B., Hunziker, E.B. & Hauselmann, H.J. Differential effects of IGF-1 and TGF beta-2 on the assembly of proteoglycans in pericellular and territorial matrix by cultured bovine articular chondrocytes. in Osteoarthritis Cartilage Vol. 6 187–95 (1998).
Kim, H.A. & Song, Y.W. Apoptotic chondrocyte death in rheumatoid arthritis. in Arthritis Rheum Vol. 42 1528–37 (1999).
Bos, P.K., DeGroot, J., Budde, M., Verhaar, J.A. & van Osch, G.J. Specific enzymatic treatment of bovine and human articular cartilage: implications for integrative cartilage repair. in Arthritis Rheum Vol. 46 976–85 (2002).
Kim, Y.J., Sah, R.L., Doong, J.Y. & Grodzinsky, A.J. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. in Anal Biochem Vol. 174 168–76 (1988).
Farndale, R.W., Sayers, C.A. & Barrett, A.J. A direct spectrophotometric microassay for sulfated glycosaminoglycans in cartilage cultures. in Connect Tissue Res Vol. 9 247–8 (1982).
Hunziker, E.B. & Quinn, T.M. Surgical removal of articular cartilage leads to loss of chondrocytes from cartilage bordering the wound edge. in J Bone Joint Surg Am Vol. 85-ASuppl 2 85–92 (2003).
Adolphe, M. et al. Effects of donor’s age on growth kinetics of rabbit articular chondrocytes in culture. in Mech Ageing Dev Vol. 23 191–8 (1983).
Evans, C.H. & Georgescu, H.I. Observations on the senescence of cells derived from articular cartilage. in Mech Ageing Dev Vol. 22 179–91 (1983).
Bos, P.K., van Osch, G.J., Frenz, D.A., Verhaar, J.A. & Verwoerd-Verhoef, H.L. Growth factor expression in cartilage wound healing: temporal and spatial immunolocalization in a rabbit auricular cartilage wound model. in Osteoarthritis Cartilage Vol. 9 382–9 (2001).
Moos, V., Fickert, S., Muller, B., Weber, U. & Sieper, J. Immunohistological analysis of cytokine expression in human osteoarthritic and healthy cartilage [In Process Citation]. in J Rheumatol Vol. 26 870–9 (1999).
Dahlgren, L.A., Mohammed, H.O. & Nixon, A.J. Temporal expression of growth factors and matrix molecules in healing tendon lesions. in J Orthop Res Vol. 23 84–92 (2005).
Komi-Kuramochi, A. et al. Expression of fibroblast growth factors and their receptors during full-thickness skin wound healing in young and aged mice. in J Endocrinol Vol. 186 273–89 (2005).
Yamamoto, T. et al. Fibroblast growth factor-2 promotes the repair of partial thickness defects of articular cartilage in immature rabbits but not in mature rabbits. in Osteoarthritis Cartilage Vol. 12 636–41 (2004).
Hunziker, E.B., Driesang, I.M. & Morris, E.A. Chondrogenesis in cartilage repair is induced by members of the transforming growth factor-beta superfamily. in Clin Orthop Relat Res S171–81 (2001).
Elford, P.R. et al. Induction of swelling, synovial hyperplasia and cartilage proteoglycan loss upon intra-articular injection of transforming growth factor beta-2 in the rabbit. in Cytokine Vol. 4 232–8 (1992).
van Beuningen, H.M., Glansbeek, H.L., van der Kraan, P.M. & van den Berg, W.B. Differential effects of local application of BMP-2 or TGF-beta 1 on both articular cartilage composition and osteophyte formation. in Osteoarthritis Cartilage Vol. 6 306–17 (1998).
van Beuningen, H.M., Glansbeek, H.L., van der Kraan, P.M. & van den Berg, W.B. Osteoarthritis-like changes in the murine knee joint resulting from intra-articular transforming growth factor-beta injections. in Osteoarthritis Cartilage Vol. 8 25–33 (2000).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer Science+Business Media, LLC
About this paper
Cite this paper
Bos, P.K., Verhaar, J.A.N., van Osch, G.J.V.M. (2006). Age-Related Differences in Articular Cartilage Wound Healing: A Potential Role for Transforming Growth Factor β1 in Adult Cartilage Repair. In: Fisher, J.P. (eds) Tissue Engineering. Advances in Experimental Medicine and Biology, vol 585. Springer, Boston, MA. https://doi.org/10.1007/978-0-387-34133-0_20
Download citation
DOI: https://doi.org/10.1007/978-0-387-34133-0_20
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-32664-1
Online ISBN: 978-0-387-34133-0
eBook Packages: EngineeringEngineering (R0)