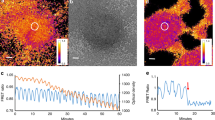
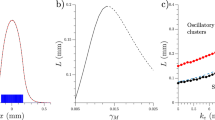
Abstract
For many years it has been known that developing cells of Dictyostelium discoideum show periodic surges as they aggregate. When it was discovered that the cells were responding chemotactically to cAMP gradients produced within the populations, experiments were carried out that demonstrated similar periodic changes in the concentration of extracellular cAMP. Moreover, homogenous populations of developed cells held in suspension could be shown to respond to cAMP by changes in cell shape. Such suspensions showed spontaneous oscillations in light scattering as well as cAMP levels as the result of entrainment of the cells. The molecular components necessary for the pulsatile release of cAMP were uncovered by analyzing the behavior of a large number of strains with defined mutations isolated from saturation mutagenic screens. Subsequent genetic and biochemical studies established the connections between a dozen proteins essential for spontaneous oscillations. Computer simulations of a molecular circuit based on these results showed that it is able to account for the temporal and quantitative aspects of the oscillatory system. The circuit also appears to be coupled to the construction and dismantling of the actin/myosin cortical layer that ensures that pseudopods are restricted to the anterior of cells during chemotaxis and that the cells do not back-track when the natural wave is behind them. Since the same molecular clock controls both signal production and signal response, these behaviors are always kept strictly in phase.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Dyachok O, Isakov Y, Sagetorp J et al. Oscillations of cyclic AMP in hormone-stimulated insulin-secreting beta-cells. Nature 2006; 439:349–352.
Navarro C, Saeed S, Murdock C et al. Regulation of cyclic adenosine 3′, 5′-monophosphate signaling and pulsatile neurosecretion by Gi-coupled plasma membrane estrogen receptors in immortalized gonadotrophin-releasing hormone neurons. Mol Endocrinol 2003; 17:1792–1804.
Wetsel W, Valenca M, Merchenthaler I et al. Intrinsic pulsatile secretory activity of immortalized luteinizing hormone-releasing hormone-secreting neurons. Proc Natl Acad Sci 1992; 89:4149–4153.
Baillie G, MacKenzie S, Houslay M. Phorbol 12-myristate 13-acetate triggers the protein kinase A-mediated phosphorylation and activation of the PDE4D5 cAMP phosphodiesterase in human aortic smooth muscle cells through a route involving extracellular signal regulated kinase (ERK). Mol Pharmacol 2001; 60:1100–1111.
Konijn TM, van de Meene JGC, Bonner JT et al. The acrasin activity of adenosine-3′, 5′-cyclic phosphate. Proc Natl Acad Sci USA 1967; 58:1152–1154.
Loomis WF. Role of PKA in the timing of developmental events in Dictyostelium cells. Microbiol Mol Biol Rev 1998; 62:684.
Laub MT, Loomis WF. A molecular network that produces spontaneous oscillations in excitable cells of Dictyostelium. Mol Biol Cell 1998; 9:3521–3532.
Maeda M, Lu SJ, Shaulsky G et al. Periodic signaling controlled by an oscillatory circuit that includes protein kinases ERK2 and PKA. Science 2004; 304:875–878.
Martiel JL, Goldbeter A. A model based on receptor desensitization for cyclic AMP signaling in Dictyostelium cells. Biophys J 1987; 52:807–828.
Parent CA, Devreotes PN. Molecular genetics of signal transduction in Dictyostelium. Annu Rev Biochem 1996; 65:411–440.
Loomis WF, Smith DW. Molecular phylogeny of Dictyostelium dscoideum by protein sequence comparison. Proc Natl Acad Sci USA 1990; 87:9093–9097.
Song J, Olsen R, Loomis WF et al. Comparing the Dictyostelium and Entamoeba genomes reveals an ancient split in the Conosa lineage. PLoS Comput Biol 2005; 1:579–584.
Iranfar N, Fuller D, Loomis WF. Genome-wide expression analyses of gene regulation during early development of Dictyostelium discoideum. Euk Cell 2003; 2:664–670.
Kumagai A, Hadwiger JA, Pupillo M et al. Molecular genetic analysis of two Galpha protein subunits in Dictyostelium. J Biol Chem 1991; 266:1220–1228.
Segall JE, Kuspa A, Shaulsky G et al. A MAP kinase necessary for receptor-mediated activation of adenylyl cyclase in Dictyostelium. J Cell Biol 1995; 128:405–413.
Levine H, Aranson I, Tsimring L et al. Positive genetic feedback governs cAMP spiral wave formation in Dictyostelium. Proc Natl Acad Sci USA 1996; 93:6382–6386.
Aubry L, Maeda M, Insall R et al. The Dictyostelium mitogen-activated protein kinase ERK2 is regulated by ras and cAMP-dependent protein kinase (PKA) and mediates PKA function. J Biol Chem 1997; 272:3883–3886.
Shaulsky G, Fuller D, Loomis WF. A cAMP-phosphodiesterase controls PKA-dependent differentiation. Development 1998; 125:691–699.
Sawai S, thomason PA, Cox EC. An autoregulatory circuit for long-range self-organization in Dictyostelium cell populations. Nature 2005; 433:323–326.
Gerisch G, Hess B. Cyclic-AMP-controlled oscillations in suspended Dictyostelium cells: Their relation to morphogenetic cell interactions. Proc Natl Acad Sci USA 1974; 71:2118–2122.
Gerisch G, Malchow D, Roos W et al. Oscillations of cyclic nucleotide concentrations in relation to the excitability of Dictyostelium cells. J exp Biol 1979; 81:33–47.
Winfree A. Phase control of neural pacemakers. Science 1977; 197:761–763.
Mohanty S, Lee S, Yadava N et al. Regulated protein degradation controls PKA function and cell-type differentiation in Dictyostelium. Genes Devel 2001; 15:1435–1448.
Boeckeler K, Adley K, Xu X et al. The neuroprotective agent, valproic, acid, regulates the mitogen-activated protein kinase pathway through modulation of protein kinase A signalling in Dictyostelium discoideum. Eur J Cell Biol 2006; 85:1097–1057.
Brzostowski JA, Parent CA, Kimmel AR. A Galpha-dependent pathway that antagonizes multiple chemoattractant responses that regulate directional cell movement. Genes Devel 2004; 18:805–815.
Comer F, Parent CA. Phosphoinositide 3-kinase activity controls the chemoattractant-mediated activation and adaptation of adenylyl cyclase. Mol Biol Cell 2006; 17:357–366.
Devreotes PN, Zigmond SH. Chemotaxis in eukaryotic cells: A focus on leukocytes and Dictyostelium. Annu Rev Cell Biol 1988; 4:649–686.
Soll DR, Wessels D, Heid PJ et al. A contextual framework for characterizing motility and chemotaxis mutants in Dictyostelium discoideum. J Muscle Res Cell Motil 2002; 23:659–672.
Wessels D, Voss E, Von Bergen N et al. A computer-assisted system for reconstructing and interpreting the dynamic three-dimensional relationships of the outer surface, nucleus and pseudopods of crawling cells. Cell Motil Cytoskel 1998; 41:225–246.
Wessels DJ, Zhang H, Reynolds J et al. The internal phosphodiesterase RegA is essential for the suppression of lateral pseudopods during Dictyostelium chemotaxis. Mol Biol Cell 2000; 11:2803–2820.
Stepanovic V, Wessels D, Daniels K et al. Intracellular role of adenylyl cyclase in regulation of lateral pseudopod formation during Dictyostelium chemotaxis. Euk Cell 2005; 4:775–786.
Zhang H, Heid PJ, Wessels D et al. Constitutively active protein kinase A disrupts motility and chemotaxis in Dictyostelium discoideum. Euk Cell 2003; 2:62–75.
Yumura S, Yoshida M, Betapudi V et al. Multiple myosin II heavy chain kinases: Roles in filament assembly control and proper cytokinesis in Dictyostelium. Mol Biol Cell 2005; 16:4256–4266.
Heid PJ, Wessels D, Daniels KJ et al. The role of myosin heavy chain phosphorylation in Dictyostelium motility, chemotaxis and F-actin localization. J Cell Sci 2004; 117:4819–4835.
Behrens MM, Juliani MH, Maia JCC. Periodic changes in the cAMP-dependent protein kinase activity ratio in Dictyostelium discoideum. Biochem Internat 1986; 13:221–226.
Janetopoulos C, Jin T, Devreotes P. Receptor-mediated activation of heterotrimeric G-proteins in living cells. Science 2001; 291:2408–2411.
Geiger J, Wessels D, Soll DR. Human polymorphonuclear leukocytes respond to wave of chemoattractant, like Dictyostelium. Cell Motil Cytoskel 2003; 56:27–44.
Song L, Nadkarni S, Bodeker H et al. Dictyostelium discoideum chemotaxis: Threshold for directed motion. Eur J Cell Biol 2006; 85:981–989.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Landes Bioscience and Springer Science+Business Media
About this chapter
Cite this chapter
Loomis, W.F. (2008). cAMP Oscillations during Aggregation of Dictyostelium . In: Maroto, M., Monk, N.A.M. (eds) Cellular Oscillatory Mechanisms. Advances in Experimental Medicine and Biology, vol 641. Springer, New York, NY. https://doi.org/10.1007/978-0-387-09794-7_3
Download citation
DOI: https://doi.org/10.1007/978-0-387-09794-7_3
Publisher Name: Springer, New York, NY
Print ISBN: 978-0-387-09793-0
Online ISBN: 978-0-387-09794-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)