Abstract
The second messenger cyclic guanosine monophosphate (cGMP) is a key mediator in physiological processes such as vascular tone, and its essential involvement in pathways regulating metabolism has been recognized in recent years. Here, we focus on the fundamental role of cGMP in brown adipose tissue (BAT) differentiation and function. In contrast to white adipose tissue (WAT), which stores energy in the form of lipids, BAT consumes energy stored in lipids to generate heat. This so-called non-shivering thermogenesis takes place in BAT mitochondria, which express the specific uncoupling protein 1 (UCP1). The energy combusting properties of BAT render it a promising target in antiobesity strategies in which BAT could burn the surplus energy that has accumulated in obese and overweight individuals. cGMP is generated by guanylyl cyclases upon activation by nitric oxide or natriuretic peptides. It affects several downstream molecules including cGMP-receptor proteins such as cGMP-dependent protein kinase and is degraded by phosphodiesterases. The cGMP pathway contains several signaling molecules that can increase cGMP signaling, resulting in activation and recruitment of brown adipocytes, and hence can enhance the energy combusting features of BAT. In this review we highlight recent results showing the physiological significance of cGMP signaling in BAT, as well as pharmacological options targeting cGMP signaling that bear a high potential to become BAT-centered therapies for the treatment of obesity.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Brown adipose tissue
- cGMP
- Inducible brown adipocytes
- PDE5 inhibitors
- PKGI
- sGC
- Thermogenesis
- UCP1
- White adipose tissue
1 cGMP Signaling
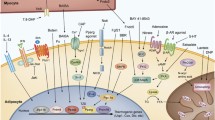
The second messenger cyclic guanosine monophosphate (cGMP) is synthesized from guanosine triphosphate by guanylyl cyclases (GC), which exist as membrane bound (pGC) or soluble forms (sGC). pGCs are activated by the natriuretic peptides (NP) atrial NP (ANP), brain NP (BNP), and C-type NP (CNP) (Potter 2011). The clearance receptor NPR-C also binds NPs but lacks a cyclase domain. Nevertheless, recent evidence suggests that it also contributes to signaling pathways (Anand-Srivastava 2005; Azer et al. 2012). GC-A and GC-B are the two cGMP-synthesizing isoforms expressed in mammals. sGC is a heterodimeric hemeprotein consisting of an α-subunit (α1 or α2) and a heme-containing β-subunit (Derbyshire and Marletta 2012). In the reduced Fe2+ state, sGC is activated by its endogenous ligand nitric oxide (NO). Upon oxidation, which happens during increased oxidative stress, the heme group is oxidized and unresponsive to NO. The gaseous molecule NO is endogenously produced by NO synthases (NOS) of which three isoforms have been identified: endothelial NOS (eNOS), neuronal NOS (nNOS), and inducible NOS (iNOS) (Murad 2006).
The downstream effects of cGMP are mediated by cGMP-dependent protein kinases (PKGI or PKGII), cyclic nucleotide-gated channels (CNG), and phosphodiesterases (PDE) (Francis et al. 2010). PDEs break down cGMP but can also be activated or inhibited by cGMP or its sibling cyclic adenosine phosphate (cAMP), which allows crosstalk between cGMP and cAMP signaling pathways (Francis et al. 2011). PKG is a major transmitter of cGMP signaling in many cells and phosphorylates a variety of substrates, thereby regulating important physiological processes such as blood pressure, learning, platelet aggregation (Schlossmann and Desch 2009), and metabolic control. Here, we review the role of the cGMP pathway in brown adipose tissue (BAT).
2 BAT and the Brown-Beige Debate
2.1 Brown Adipose Tissue
BAT can produce heat from energy stored in lipids by a process termed non-shivering thermogenesis (NST). The main function of BAT is to maintain the body temperature of mammals; hence, it is highly abundant in newborns yet remains active in adults (Cypess et al. 2009, 2013; Saito et al. 2009; van Marken Lichtenbelt et al. 2009; Virtanen et al. 2009). Mitochondria are the powerhouses for NST and are highly abundant in brown adipocytes (BA). Together with the high density of vessels, mitochondria give BAT its brownish color. Uncoupling protein 1 (UCP1) is a special protein in BAT mitochondria that drives NST. It is expressed in the inner mitochondrial membrane and can be activated by free fatty acids (FFA), which are released from lipids stored in multilocular droplets within BA (Cannon and Nedergaard 2004). Lipolysis is initiated by cold exposure leading to activation of sympathetic nerves innervating BAT. Sympathetic nerves release norepinephrine (NE) which activates β-adrenoceptors (AR) on BA. Recent evidence shows sympathetic cotransmission of NE and adenosine, which activates A2A receptors on human and murine BA (Gnad et al. 2014). Activation of Gs-coupled receptors induces cAMP production, which in turn drives FFA liberation (Robidoux et al. 2004). Once activated, UCP1 disrupts the mitochondrial proton gradient resulting in production of heat instead of ATP. Besides cold stimulation, BAT might also be activated by food ingestion resulting in diet-induced thermogenesis (DIT), but this concept is still a matter of debate (Kozak 2010; Cannon and Nedergaard 2011; Vosselman et al. 2013). In addition to β-adrenergic and adenosine receptor signaling pathways, BAT is regulated by hormones such as melanocortin or by thyroid hormones (Vaughan et al. 2011; Lahesmaa et al. 2014).
Activation of BAT results in depletion of the intracellular BA lipid stores, and lipids from other peripheral tissues are transported to BAT. It has been shown that activated BAT takes up 50% of nutrient lipids (Bartelt and Heeren 2012). Thus, BAT functions as a major sink for lipids and dissipates the contained energy. BAT contributes significantly to whole-body energy expenditure (EE), i.e., the amount of energy needed for food digestion, breathing, heat production, and physical activity.
2.2 White Adipose Tissue
In contrast to BAT, the major task of white adipose tissue (WAT) is to store energy in the triacylglycerol form of lipids. WAT takes up lipids through specialized transporters such as CD36 or fatty acid transport proteins or synthesizes lipids through de novo lipogenesis (Lobo et al. 2007; Bartelt et al. 2013). Lipids are stored in big unilocular droplets that fill up white adipocytes (WA). When energy intake persistently exceeds EE, as in the positive energy balance state underlying overweight and obesity, surplus energy will be stored in WAT, from where it can be released in times of energy shortage or negative energy balance. Lipolysis makes FFA available and is driven by hormone-sensitive lipase and perilipins (Large et al. 2004). In addition to its energy-storing properties, WAT functions as an endocrine organ and secretes adipokines such as leptin, TNFα, or interleukins, which can affect metabolism and can increase systemic inflammation. WAT also secretes adipokines with beneficial effects on whole-body metabolism such as adiponectin (Kwon and Pessin 2013; Turer and Scherer 2012). This topic is covered in another chapter of this volume. In humans it was estimated that activation of BAT could result in EE equivalent to approximately 4 kg fat tissue during a year (Virtanen et al. 2009).
2.3 Inducible Brown Adipocytes and Beige/Brite Adipocytes
BAT consists of constitutive classical BA, which are present at birth (Pfeifer and Hoffmann 2014), along with a second type of BA discovered in humans and mice in the recent years: inducible BA (iBA) or beige/brite (brown in white) cells that appear within WAT depots. iBA share many features with classical BA: they have multilocular lipid droplets, are rich in mitochondria, and express UCP1 highly, allowing them to combust significant amounts of energy in a nonproductive manner (Rosen and Spiegelman 2014; Frontini and Cinti 2010; Wu et al. 2013). iBA are induced by several physiological stimuli such as cold exposure or through treatment with pharmacological compounds such as β3AR and peroxisome proliferator-activated receptor gamma (PPARγ) agonists (Vegiopoulos et al. 2012; Petrovic et al. 2010). Two mechanisms of browning have been identified (Rosenwald and Wolfrum 2014): iBA are formed through a classic differentiation cascade of iBA precursors residing in WAT or through transdifferentiation of WA (Lee et al. 2012; Barbatelli et al. 2010 and references in Rosenwald and Wolfrum 2014). The potential for browning is higher in inguinal WAT than in epididymal WAT and occurs in a mottled pattern in areas of particular adipose depots (Frontini and Cinti 2010; Rosenwald et al. 2013). iBA contribute to whole-body EE and protect mice from diet-induced obesity (Seale et al. 2011; Vegiopoulos et al. 2012).
Importantly, iBA also have been discovered in humans, where they were found in close vicinity to areas that contain classical BAT or within BAT depots (Wu et al. 2012; Jespersen et al. 2013).
iBA can revert to WA (Rosenwald et al. 2013). This process is known as “whitening” and can be observed for iBA and also in the classical BAT of obese mice. Whitening in BAT is characterized by an increase in lipid droplet size, a reduction in vascularization, and a decrease in VEGF expression compared to nonobese mice (Shimizu et al. 2014).
Pink adipocytes add another color to adipose tissue. These special adipocytes arise during pregnancy and lactation in subcutaneous WA of the mammary gland (Giordano et al. 2014). They not only produce milk but they also secrete leptin which might play a role in the regulation of obesity in the offspring (Giordano et al. 2014; Morroni et al. 2004).
In summary, adipose tissue is a highly dynamic tissue, with a white compartment that can expand and contract to match the energy supply and demand of the organism and a brown compartment comprised of both classical and inducible cell types, which shows high plasticity in number and activation of BA. Understanding the signaling pathways that regulate adipose tissue might reveal new targets for antiobesity therapies, which are highly needed in times of the obesity pandemic.
3 Central Nervous System Control of BAT
As with other organ systems in the body, BAT is under both neural and endocrine control. Autonomic regulation of BAT has been recently reviewed (Tupone et al. 2014), and its role in health and disease suggests potential clinical applications through altering BAT activity.
BAT sympathetic nerve system (SNS) activity controlling BAT thermogenic function is modulated by central nervous system neural networks that react to thermal afferent signaling from cutaneous and body core thermal receptor systems and also to changes in the firing of central neurons with intrinsic thermal reactivity. In addition to this basal network providing thermal regulation of BAT thermogenesis, a second system of central networks controlling metabolic aspects of energy homeostasis provides centrally derived neural inputs into peripheral BAT thermogenic action. The role of cGMP signaling in central control of energy balance has been most recently demonstrated through intraventricular application of CNP, which alters food intake by activation of the melanocortin system in mice (Yamada-Goto et al. 2013). In addition, intravenously applied BNP modulated ghrelin, hunger, and satiety in humans (Vila et al. 2012).
Central control of energy balance through activation of BAT also has been demonstrated by several recent cases. First, induction of UCP1 by central delivery of IL-6 requires autonomic sympathetic innervation of BAT and causes body weight reduction in rodents (Li et al. 2002). Second, central signaling originating from the calcitonin gene-related peptide (CGRP) and amylin receptors, which are expressed in areas of the brain regulating energy balance, has been shown to regulate peripheral activity of BAT. Overexpressing RAMP1, one of several accessory proteins controlling the activity of these receptors through modulation of the ligand-binding specificity, decreases body weight, lowers fat mass and circulating leptin, elevates total body EE as measured by increased oxygen consumption, and raises body temperature and sympathetic tone into BAT. As well, the elevated BAT RNA levels of PPARγ coactivator 1 alpha (PGC1α), UCP1, and UCP3 in these mice can be reversed by chronic blockade of sympathetic nervous system signaling (Zhang et al. 2011). Finally, blocking KATP-channel-dependent modulation of catecholaminergic neurons in the locus coeruleus (LC), which alter their firing frequency according to their external glycemic environment, by expressing a variant KATP channel enhances diet-induced obesity in mice through lower EE and BAT sympathetic tone and a decreased ability of centrally administered glucose to stimulate BAT sympathetic nerve activity (Tovar et al. 2013).
4 cGMP and Metabolic Control
The cGMP pathway is involved in the regulation of various physiological functions ranging from regulation of vascular tone, memory and hearing processes, and metabolic control. The crucial role of cyclic nucleotides in metabolism has long been appreciated with a special focus on cAMP which is essential for sympathetic activation of BAT. But the other cyclic nucleotide cGMP also plays a key role in adipose tissue. Here, we focus on the role of cGMP signaling in adipose tissue especially in brown adipose tissue.
4.1 Adipose Tissue
4.1.1 cGMP Downstream Signaling
Initially, though, the expression of PKGI in adipose tissue was questioned (Lutz et al. 2011). In contrast, several other papers described PKGI to be expressed in both BAT and WAT (Haas et al. 2009; Mitschke et al. 2013; Leiss et al. 2014). Importantly, in vivo studies during the last several years have proven the functional importance of cGMP/PKGI signaling for adipose tissue. As the major downstream target of cGMP, PKGI plays a central role in BAT, most obviously in mice lacking PKGI. These mice show dramatically reduced thermogenesis compared to WT littermates. BAT of PKGI−/− mice has dramatically reduced protein levels of the thermogenic key protein UCP1 and shows significantly decreased amounts of mitochondrial DNA (Haas et al. 2009). Molecular analysis of BA differentiated from the stromal vascular fraction of these PKGI−/− mice revealed that BA differentiation is dependent on functional cGMP signaling. Furthermore, abrogation of the cGMP pathway leads to downregulation of BA key features such as expression of thermogenic markers UCP1 and PGC1α and abundance of mitochondria. The adipogenic pathway in PKGI-deficient BA is impaired as well: lipid incorporation and adipogenic marker expression are significantly reduced in PKGI−/− cells compared to WT cells. Importantly, with the use of PKGI−/− BA, a crosstalk between cGMP and insulin signaling was revealed that explained the great impact of PKGI deficiency on BA differentiation and function. In BA, Ras homolog gene family member A (RhoA), activates Rho-associated protein kinase (ROCK) which in turn inhibits the insulin receptor substrate 1 (IRS-1). IRS-1 initiates a signaling cascade that transmits insulin binding to its receptor into mitochondrial biogenesis and adipogenic differentiation. PKGI inhibits RhoA and thereby releases the inhibitory effect of RhoA/ROCK on insulin signaling. Hence, insulin signaling in PKGI−/− BA is hampered resulting in the observed defects in differentiation and function (Haas et al. 2009).
In contrast, global overexpression of PKGI results in increased mitochondrial DNA content and upregulation of PGC1α and UCP1 in BAT (Miyashita et al. 2009). This seems to result in increased thermogenesis as PKGI transgenic mice display a higher body temperature. Importantly, PKGI transgenic mice are leaner than WT littermates and protected against diet-induced obesity which is accompanied by improved insulin sensitivity (Miyashita et al. 2009; Nikolic et al. 2009).
In addition to BA, PKGI also plays an important role in WA. Ablation of PKGI in primary murine adipocytes represses adipogenesis resulting in decreased lipid storage and reduced expression of the adipogenic markers PPARγ and adipocyte protein 2 (aP2) (Mitschke et al. 2013). Similar to the situation in BA, mitochondrial markers UCP1 and cytochrome c are also reduced in PKGI−/− WA. In addition to its role in adipogenesis and mitochondrial biogenesis, the cGMP pathway also influences the endocrine function of WA. Overexpression of PKG in WA results in a reduction of pro-inflammatory cytokines such as monocyte chemoattractant protein 1 (MCP1) and IL-6 and increases the antidiabetic and anti-inflammatory adipokine adiponectin (Adipoq) (Caselli 2014; Mitschke et al. 2013). Taken together, the studies on PKGI−/− mice and cells clearly show that the cGMP pathway is an essential enhancer of BA differentiation. Moreover, the elegant studies on PKG transgenic mice (Miyashita et al. 2009) and on adipocytes incubated with cGMP (Haas et al. 2009) show that cGMP activation results in “super” BAT.
Vasodilator-stimulated phosphoprotein (VASP) is phosphorylated by PKGI and plays an important and unexpected role in BAT through regulation of cGMP signaling. As PKGI−/− BA show dysfunctional differentiation and function, the same was anticipated for BA lacking VASP. Surprisingly, VASP−/− BA show increased adipogenesis and have an upregulated thermogenic program resulting in enhanced BA function: Cellular respiration and lipolysis are significantly higher in VASP−/− cells than in WT cells (Jennissen et al. 2011). In WT cells, VASP blocks Rac-mediated activation of sGC-gene transcription in a negative feedback loop (Jennissen et al. 2011). In VASP−/− cells, sGC promoter activity is upregulated resulting in increased cGMP signaling. This pathway is also active in WA. VASP−/− mice are leaner and have increased EE. Importantly, activation of the cGMP pathway through ablation of the inhibitory effect of VASP on sGC expression results in browning of WAT in VASP−/−. Taken together, the results obtained from VASP−/− cells and mice demonstrate the central role of cGMP in regulating BA differentiation and function and in inducing and recruiting BA.
Apart from PKGI and VASP, PDEs that break down cGMP levels also have been shown to regulate BAT function. PDE5 is the major PDE for the breakdown of cGMP and is expressed in adipose tissue (Moro et al. 2007). Inhibition of PDE5 by sildenafil in WA resulted in upregulation of the BA markers UCP1 and PGC1α, indicating browning of WA (Mitschke et al. 2013). In addition, an antidiabetic potential was observed in a WA cell line that showed increased basal and insulin-stimulated glucose uptake after incubation with sildenafil (Zhang et al. 2010).
4.1.2 cGMP Upstream Signaling
Concerning the upstream regulation of cGMP signaling, a major focus was on NOS and NO. Exogenously applied NO results in the upregulation of PGC1α and UCP1, mitochondrial DNA, and the mitochondrial markers nuclear respiratory factor 1 (Nrf1) and mitochondrial transcription factor A (mtTFA) (Nisoli et al. 1998, 2003). Inhibition of the NO receptor sGC with ODQ abrogates these effects (Nisoli et al. 1998, 2003). In BAT of mice deficient for eNOS, the major NOS in BAT (Haas et al. 2009), mitochondrial content is reduced and lipid droplets are enlarged pointing to whitening (Nisoli et al. 2003). eNOS−/− mice gain more weight than WT littermates and have reduced EE (Nisoli et al. 2003). Overexpression of sGC results in improved BAT function and WAT browning (Jennissen et al. 2011). Apart from NO/sGC, also NPs induce cGMP production in BAT and have been shown to be involved in the induction of iBA and increased BA function. In a human BA cell line, ANP and BNP increased thermogenic marker expression and cellular respiration (Bordicchia et al. 2012). Enhancing NP signaling by deletion of the clearance receptor NPR-C results in reduction of adipose tissue mass in mice (Bordicchia et al. 2012).
5 cGMP and Mitochondrial Biology
The relationship between cGMP signaling and mitochondrial biology is complex. The stimulatory role of NO/sGC/cGMP signaling toward mitochondrial biogenesis can be measured in cellular lineages beyond BA, such as 3T3-L1, U937, and HeLa cells, is mediated by the transcriptional coactivator PGC1α (Nisoli et al. 2003), and can be exploited to protect and restore the function of highly respiring tissues such as the kidney (Whitaker et al. 2013). The NO/sGMP pathway stimulation of mitochondria extends even outside the animal kingdom to enhance mitochondrial respiration in plants such as Arabidopsis (Wang et al. 2010). Beyond the NO/sGC/cGMP input to mitochondrial biogenesis, NO inhibits mitochondrial respiration by competing with O2 at cytochrome oxidase, and reactive nitrogen derivatives of NO irreversibly inhibit multiple sites on cytochrome oxidase. As well, NO/sGC/cGMP-mediated vasodilation drives mitochondria respiration through increased circulatory delivery of substrate and oxygen, and reactive nitrogen derivatives of NO can activate the mitochondrial permeability transition pore, leading to apoptosis or necrosis (Brown 2007).
As noted above, cGMP plays an important role in the biology of classical and inducible BAT, stimulating BA differentiation and mitochondrial biogenesis as well as healthy expansion and browning of WAT (Mitschke et al. 2013; Haas et al. 2009). These data have been extended to human WAT (De Toni et al. 2011) upon the observation that human adipose tissue contains an intact signaling system of NO/sGC/cGMP/PKGI, as well as PDE5, the main cGMP catabolizing enzyme. Treating cultured human omental adipose tissue explants with the PDE5 inhibitor vardenafil increased PPARγ, PGC1α, and mitochondrial DNA levels, suggesting a beneficial effect on energy metabolism in human white adipose tissue (De Toni et al. 2011).
Given the ancestral relationship between BAT and the skeletal muscle (Seale et al. 2008), it is not surprising that cGMP signaling promotes mitochondrial biogenesis in the skeletal muscle in a manner analogous to that in BAT. In vitro, cGMP rescues mitochondrial dysfunction induced by glucose and insulin in myocytes (Mitsuishi et al. 2008). In C2C12 myotubular cells with mitochondrial dysfunction generated by high-glucose and high-insulin treatment, cGMP treatment elevated genes involved in oxidative phosphorylation and ROS reduction, resulting in increased mitochondrial biogenesis and ATP production with no increase in ROS levels. Likewise, in cultured rat L6 myotubes, NO/cGMP-dependent mitochondrial biogenesis correlates with coupled respiration and content of ATP, with the latter not generated from glycolysis (Nisoli et al. 2004). In the same report, the gastrocnemius muscle from eNOS−/− mice exhibited reduced mitochondrial content, smaller mitochondria, and a decrease in the number of mitochondria in the subsarcolemmal region of the gastrocnemius muscle. These results are consistent with findings from other investigators demonstrating NOS expression in the sarcolemma, sarcoplasmic reticulum, and mitochondria regions, suggesting a paracrine nature of NO/cGMP signaling in the skeletal muscle that contributes to increased cGMP-dependent mitochondrial functioning (Buchwalow et al. 2005). Lastly, studies with transgenic mice have demonstrated that chronic in vivo activation of natriuretic peptides/cGMP/cGMP-dependent protein kinase cascades promotes increased muscle mitochondrial content and fat oxidation through elevated PPARδ and PGC1α content, which work to lower glucose intolerance and body weight (Miyashita et al. 2009).
As BAT and skeletal muscle together comprise a large fraction of the total EE in lean individuals, augmenting the metabolic actions of these tissues, particularly those of BAT, represents a promising therapeutic strategy for the treatment of obesity and its associated comorbidities.
6 BAT-Centered Antiobesity Therapies
The global obesity epidemic continues to expand, with prevalence of disease and its comorbidities now increasing in both developed and developing nations. The comorbidities and associated economic burdens bring substantial and growing healthcare and financial consequences throughout the globe (Imes and Burke 2014). Weight loss strategies for the treatment of obesity escalate from lifestyle approaches including diet, physical activity, and behavior change therapies to pharmacological agents approved as adjuncts to lifestyle modification to bariatric surgery, demonstrated to be the most effective and persistent treatment for individuals with severe obesity or moderate obesity complicated by comorbid conditions refractory to other approaches (Kushner 2014). Current pharmacotherapy for obesity has been recently reviewed and will not be detailed here (Gadde 2014). With increases in obesity prevalence in most of the developed world continuing, and the largest increases now being seen in developing countries, new antiobesity therapies – including pharmacotherapies – are needed.
Activation and recruitment of BAT represents a promising strategy for obesity treatment in humans, as BAT is the primary source of sympathetically activated adaptive thermogenesis during cold exposure thus modulating whole-body EE and adiposity, with an inverse relationship between BAT activity and adiposity implying that the energy-dissipating activity of BAT defends against body fat accumulation and potentially glucose intolerance (Contreras et al. 2014). As noted earlier, both autonomic sympathetic nervous system activity and endocrine factors have been demonstrated to affect BAT activity and thus provide guidance toward novel therapeutic strategies (Lidell et al. 2014).
A number of BAT-centered antiobesity potential strategies have proven unsuccessful, for a variety of reasons. β3-adrenoceptor agonists are efficacious antiobesity and insulin-sensitizing agents acting in WAT, BAT, and muscles. However, the selectivity needed to avoid β1/2-adrenoceptor-mediated side effects has been shown to be challenging (Arch 2002). Analogs of fibroblast growth factor 21 (FGF21), which act in part through BAT activation, have been advanced into early clinical trials recently but have provided only modest metabolic improvements in patients (Gaich et al. 2013). A PGC1α-dependent myokine irisin, cleaved from the membrane protein FNDC5, generated significant excitement due to its ability to stimulate browning of WAT and thermogenesis (Bostrom et al. 2012), though recently a number of published and unpublished reports suggest a more complicated biology with less clear therapeutic implications that had been originally hoped. Finally, increased T3 production through elevating peripheral deiodinase 2 (Dio2) levels, such as can be achieved by agonism of the bile acid receptor TGR5, would be predicted to increase EE through increased thermogenesis and thus decrease body weight (Mullur et al. 2014). However, few pharmacological TGR5 programs have advanced into clinical development due to frequent observations of gallbladder fibrosis in nonclinical species.
Given its importance in classical and inducible BA and the potential relevance of these cells in weight management, modulating cGMP signaling may represent an important goal for an antiobesity pharmacotherapy. Drugs that target the cGMP pathway have already proven to be safe and are approved for clinical use.
Among these are PDE5 inhibitors that have been used to treat erectile dysfunction and pulmonary hypertension for more than 15 years (Huang and Lie 2013). PDE5 inhibitors have been shown to induce browning in mice. A short-term treatment with the PDE5 inhibitor sildenafil for only 7 days significantly increased UCP1 and PGC1α expression and induced iBA in WATi of WT mice (Mitschke et al. 2013). Importantly, this short time of treatment shows that molecular changes take place relatively early and indicated that a long-term treatment might have an influence on whole-body energy metabolism. Interestingly, when mice on a high-fat diet were treated with sildenafil, weight gain was significantly lower and EE was significantly higher than in untreated obese mice (Ayala et al. 2007). Unfortunately, WAT of these mice was not analyzed in this study. Taken together, the two studies point to a potential of PDE5 inhibitors to induce browning and thereby induce weight loss in mice. Nevertheless, data from humans are still missing.
The potential for browning also extends to PDE3 and PDE4 which regulate cAMP inhibition. After addition of the PDE3 inhibitor cilostamide and the PDE4 inhibitor rolipram, UCP1 expression increased in BAT of mice and in in vitro differentiated BA (Kraynik et al. 2013).
cGMP levels can be elevated not only by PDE5 inhibition but also via activation of pGC by NP. Transgenic overexpression of BNP in mice challenged with a high-fat diet results in decreased WAT mass; increases body temperature, mitochondrial biogenesis, and UCP1 and PGC1α expression in BAT; and induces mitochondrial biogenesis in the muscle (Miyashita et al. 2009). Furthermore, short-term infusion of BNP for 7 days in mice results in significantly upregulated EE and increased thermogenic marker gene expression in BAT and WAT (Bordicchia et al. 2012). In humans and isolated human WA, NPs induce lipolysis which can provide fuel in the form of FFA for oxidative tissues such as BAT and the muscle (see references in Schlueter et al. 2014). Interestingly, a connection between weight loss and ANP and BNP has been shown in type 2 diabetes patients that were treated with the antidiabetic drug liraglutide (Li et al. 2014). Weight loss in this study was positively correlated with an increase in plasma ANP levels. Furthermore, BNP levels were increased after liraglutide-induced weight loss. However, the significant renal and cardiovascular actions of natriuretic peptides in reducing arterial blood pressure as well as sodium reabsorption could potentially limit their application to modulating biological processes beyond hemodynamic regulation.
Recently, bone morphogenic protein 7 (BMP7) and BMP8B have been shown to have effects on BAT development and energy balance and thus may represent a novel approach to BAT-mediated antiobesity therapy (Saini et al. 2014; Whittle et al. 2012).
Finally, the canonical ventromedial nucleus of the hypothalamus (VMH) AMP-activated protein kinase (AMPK)-SNS-BAT axis has long been regarded as an attractive hypothalamic and peripheral obesity target, particularly through its direct and indirect actions in BAT (Klaus et al. 2012), but the challenges of developing a drug-like, selective kinase activator have precluded advancement into significant clinical development.
7 BAT-Centered Antidiabetic Therapies
Obesity is accompanied by many comorbidities such as type 2 diabetes, certain types of cancer, and cardiovascular diseases (Cao 2010). The prevalence of type 2 diabetes is increasing in the recent years and, just like obesity, is a major burden to the global health system (Hu 2011). BAT has been shown to have antidiabetic functions. Recently it has been shown in humans that activated BAT not only increases EE but importantly improves plasma glucose oxidation and insulin sensitivity (Chondronikola et al. 2014). As well, increases in BAT activity through acute cold exposure and increases in BAT volume through long-term exposure can modulate insulin sensitivity in humans (Lee et al. 2014), an important observation given the persistent need for insulin sensitizers as disease-modifying antidiabetic drugs (Larson 2014). This suggests the aforementioned BAT-mediated antiobesity strategies may also be indicated to address the global diabetes epidemic, a critical point since modern drug discovery has not resulted in agents that substantially reverse the molecular or cellular origins of either insulin resistance or loss of functional beta-cell mass. Diabetic pathophysiology can become disabling and even life threatening because it is concentrated in the circulatory and nervous systems, thereby resulting in heart disease; chronic kidney disease, and kidney failure; peripheral artery disease; neuropathies leading to ulcerations, infections, and amputation; and retinopathies triggering loss of vision. A recent encouraging report suggests that diabetic nephropathy might be beneficially addressed with PDE5 inhibitors that reduce glomerular hypertension and other hemodynamic aspects of diabetic nephropathy, as well as metabolic and inflammatory aspects of disease (Thompson 2013).
8 Conclusions
The cGMP second messenger system plays a crucial role in the functions of BAT and WAT, in the induction of iBA/beige adipocytes, and in the interplay between these cell types and tissue depots important for maintaining energetic balance in mammals, including humans. As mitochondria are the major source of energy in the mammalian cells, it is not surprising that the cGMP pathway plays a critical role in the activities of this organelle, though the relationship between cGMP signaling and mitochondrial biology is surprisingly complex. The cGMP pathway contributes to brain pathways underlying central metabolic control, as well as functioning downstream of the neural and endocrine inputs into these tissues. Modulating the cGMP pathway could lead to increased EE through BAT activation and recruitment. Hence, the cGMP pathway has a high therapeutic potential to be used in antiobesity therapies as both classical and inducible BAT can increase EE and thereby might be protective against obesity and could possibly induce weight loss. Furthermore, cGMP signaling could be used to induce antidiabetic effects. Pharmacological or genetic enhancement of cGMP pathways has shown beneficial effects in models of obesity and diabetes. These effects might be translated into the clinics with the use of already available drugs targeting the cGMP pathway such as PDE5 inhibitors.
References
Anand-Srivastava MB (2005) Natriuretic peptide receptor-C signaling and regulation. Peptides 26:1044–1059, Epub 2005 Apr 8
Arch JR (2002) beta(3)-Adrenoceptor agonists: potential, pitfalls and progress. Eur J Pharmacol 440:99–107
Ayala JE, Bracy DP, Julien BM, Rottman JN, Fueger PT, Wasserman DH (2007) Chronic treatment with sildenafil improves energy balance and insulin action in high fat-fed conscious mice. Diabetes 56:1025–1033, Epub 2007 Jan 17
Azer J, Hua R, Vella K, Rose RA (2012) Natriuretic peptides regulate heart rate and sinoatrial node function by activating multiple natriuretic peptide receptors. J Mol Cell Cardiol 53:715–724
Barbatelli G, Murano I, Madsen L, Hao Q, Jimenez M, Kristiansen K, Giacobino JP, De Matteis R, Cinti S (2010) The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol Endocrinol Metab 298:30
Bartelt A, Heeren J (2012) The holy grail of metabolic disease: brown adipose tissue. Curr Opin Lipidol 23:190–195
Bartelt A, Weigelt C, Cherradi ML, Niemeier A, Todter K, Heeren J, Scheja L (2013) Effects of adipocyte lipoprotein lipase on de novo lipogenesis and white adipose tissue browning. Biochim Biophys Acta 1831:934–942
Bordicchia M, Liu D, Amri EZ, Ailhaud G, Dessi-Fulgheri P, Zhang C, Takahashi N, Sarzani R, Collins S (2012) Cardiac natriuretic peptides act via p38 MAPK to induce the brown fat thermogenic program in mouse and human adipocytes. J Clin Invest 122:1022–1036
Bostrom P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, Rasbach KA, Bostrom EA, Choi JH, Long JZ, Kajimura S, Zingaretti MC, Vind BF, Tu H, Cinti S, Hojlund K, Gygi SP, Spiegelman BM (2012) A PGC1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 481:463–468
Brown GC (2007) Nitric oxide and mitochondria. Front Biosci 12:1024–1033
Buchwalow IB, Minin EA, Samoilova VE, Boecker W, Wellner M, Schmitz W, Neumann J, Punkt K (2005) Compartmentalization of NO signaling cascade in skeletal muscles. Biochem Biophys Res Commun 330:615–621
Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev 84:277–359
Cannon B, Nedergaard J (2011) Nonshivering thermogenesis and its adequate measurement in metabolic studies. J Exp Biol 214:242–253
Cao Y (2010) Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat Rev Drug Discov 9:107–115
Caselli C (2014) Role of adiponectin system in insulin resistance. Mol Genet Metab 7192:00292–00293
Chondronikola M, Volpi E, Borsheim E, Porter C, Annamalai P, Enerback S, Lidell ME, Saraf MK, Labbe SM, Hurren NM, Yfanti C, Chao T, Andersen CR, Cesani F, Hawkins H, Sidossis LS (2014) Brown adipose tissue improves whole body glucose homeostasis and insulin sensitivity in humans. Diabetes 63:4089–4099
Contreras C, Gonzalez F, Ferno J, Dieguez C, Rahmouni K, Nogueiras R, Lopez M (2014) The brain and brown fat. Ann Med 10:1–19
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng YH, Doria A, Kolodny GM, Kahn CR (2009) Identification and importance of brown adipose tissue in adult humans. N Engl J Med 360:1509–1517
Cypess AM, White AP, Vernochet C, Schulz TJ, Xue R, Sass CA, Huang TL, Roberts-Toler C, Weiner LS, Sze C, Chacko AT, Deschamps LN, Herder LM, Truchan N, Glasgow AL, Holman AR, Gavrila A, Hasselgren PO, Mori MA, Molla M, Tseng YH (2013) Anatomical localization, gene expression profiling and functional characterization of adult human neck brown fat. Nat Med 19:635–639
De Toni L, Strapazzon G, Gianesello L, Caretta N, Pilon C, Bruttocao A, Foresta C (2011) Effects of type 5-phosphodiesterase inhibition on energy metabolism and mitochondrial biogenesis in human adipose tissue ex vivo. J Endocrinol Invest 34:738–741
Derbyshire ER, Marletta MA (2012) Structure and regulation of soluble guanylate cyclase. Annu Rev Biochem 81:533–559
Francis SH, Busch JL, Corbin JD, Sibley D (2010) cGMP-dependent protein kinases and cGMP phosphodiesterases in nitric oxide and cGMP action. Pharmacol Rev 62:525–563
Francis SH, Blount MA, Corbin JD (2011) Mammalian cyclic nucleotide phosphodiesterases: molecular mechanisms and physiological functions. Physiol Rev 91:651–690
Frontini A, Cinti S (2010) Distribution and development of brown adipocytes in the murine and human adipose organ. Cell 11:253–256
Gadde KM (2014) Current pharmacotherapy for obesity: extrapolation of clinical trials data to practice. Expert Opin Pharmacother 15:809–822
Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, Kharitonenkov A, Bumol T, Schilske HK, Moller DE (2013) The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab 18:333–340
Giordano A, Smorlesi A, Frontini A, Barbatelli G, Cinti S (2014) White, brown and pink adipocytes: the extraordinary plasticity of the adipose organ. Eur J Endocrinol 170:13–0945
Gnad T, Scheibler S, von Kugelgen I, Scheele C, Kilic A, Glode A, Hoffmann LS, Reverte-Salisa L, Horn P, Mutlu S, El-Tayeb A, Kranz M, Deuther-Conrad W, Brust P, Lidell ME, Betz MJ, Enerback S, Schrader J, Yegutkin GG, Muller CE, Pfeifer A (2014) Adenosine activates brown adipose tissue and recruits beige adipocytes via A receptors. Nature 516:395–399
Haas B, Mayer P, Jennissen K, Scholz D, Diaz MB, Bloch W, Herzig S, Fassler R, Pfeifer A (2009) Protein kinase G controls brown fat cell differentiation and mitochondrial biogenesis. Sci Signal 2:ra78
Hu FB (2011) Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care 34:1249–1257
Huang SA, Lie JD (2013) Phosphodiesterase-5 (PDE5) inhibitors in the management of erectile dysfunction. P T 38:407–419
Imes CC, Burke LE (2014) The obesity epidemic: the United States as a cautionary tale for the rest of the world. Curr Epidemiol Rep 1:82–88
Jennissen K, Haas B, Kunz W, Pfeifer A (2011) cGMP and cAMP differentially regulate differentiation and function of brown adipocytes. BMC Pharmacol 11:P37
Jespersen NZ, Larsen TJ, Peijs L, Daugaard S, Homoe P, Loft A, de Jong J, Mathur N, Cannon B, Nedergaard J, Pedersen BK, Moller K, Scheele C (2013) A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans. Cell Metab 17:798–805
Klaus S, Keipert S, Rossmeisl M, Kopecky J (2012) Augmenting energy expenditure by mitochondrial uncoupling: a role of AMP-activated protein kinase. Genes Nutr 7:369–386
Kozak LP (2010) Brown fat and the myth of diet-induced thermogenesis. Cell Metab 11:263–267
Kraynik SM, Miyaoka RS, Beavo JA (2013) PDE3 and PDE4 isozyme-selective inhibitors are both required for synergistic activation of brown adipose tissue. Mol Pharmacol 83:1155–1165
Kushner RF (2014) Weight loss strategies for treatment of obesity. Prog Cardiovasc Dis 56:465–472
Kwon H, Pessin JE (2013) Adipokines mediate inflammation and insulin resistance. Front Endocrinol 4:71
Lahesmaa M, Orava J, Schalin-Jantti C, Soinio M, Hannukainen JC, Noponen T, Kirjavainen A, Iida H, Kudomi N, Enerback S, Virtanen KA, Nuutila P (2014) Hyperthyroidism increases brown fat metabolism in humans. J Clin Endocrinol Metab 99:2013–2312
Large V, Peroni O, Letexier D, Ray H, Beylot M (2004) Metabolism of lipids in human white adipocyte. Diabetes Metab 30:294–309
Larson CJ (2014) The persistent need for insulin sensitizers and other disease-modifying anti-diabetic drugs. Expert Rev Endocrinol Metab 9:1–3
Lee YH, Petkova AP, Mottillo EP, Granneman JG (2012) In vivo identification of bipotential adipocyte progenitors recruited by beta3-adrenoceptor activation and high-fat feeding. Cell Metab 15:480–491
Lee P, Smith S, Linderman J, Courville AB, Brychta RJ, Dieckmann W, Werner CD, Chen KY, Celi FS (2014) Temperature-acclimated brown adipose tissue modulates insulin sensitivity in humans. Diabetes 63:3686–3698
Leiss V, Illison J, Domes K, Hofmann F, Lukowski R (2014) Expression of cGMP-dependent protein kinase type I in mature white adipocytes. Biochem Biophys Res Commun 452:151–156
Li G, Klein RL, Matheny M, King MA, Meyer EM, Scarpace PJ (2002) Induction of uncoupling protein 1 by central interleukin-6 gene delivery is dependent on sympathetic innervation of brown adipose tissue and underlies one mechanism of body weight reduction in rats. Neuroscience 115:879–889
Li CJ, Yu Q, Yu P, Yu TL, Zhang QM, Lu S, Yu DM (2014) Changes in liraglutide-induced body composition are related to modifications in plasma cardiac natriuretic peptides levels in obese type 2 diabetic patients. Cardiovasc Diabetol 13:1475–2840
Lidell ME, Betz MJ, Enerback S (2014) Brown adipose tissue and its therapeutic potential. J Intern Med 276:364–377
Lobo S, Wiczer BM, Smith AJ, Hall AM, Bernlohr DA (2007) Fatty acid metabolism in adipocytes: functional analysis of fatty acid transport proteins 1 and 4. J Lipid Res 48:609–620, Epub 2006 Dec 11
Lutz SZ, Hennige AM, Feil S, Peter A, Gerling A, Machann J, Krober SM, Rath M, Schurmann A, Weigert C, Haring HU, Feil R (2011) Genetic ablation of cGMP-dependent protein kinase type I causes liver inflammation and fasting hyperglycemia. Diabetes 60:1566–1576
Mitschke MM, Hoffmann LS, Gnad T, Scholz D, Kruithoff K, Mayer P, Haas B, Sassmann A, Pfeifer A, Kilic A (2013) Increased cGMP promotes healthy expansion and browning of white adipose tissue. FASEB J 27:1621–1630
Mitsuishi M, Miyashita K, Itoh H (2008) cGMP rescues mitochondrial dysfunction induced by glucose and insulin in myocytes. Biochem Biophys Res Commun 367:840–845
Miyashita K, Itoh H, Tsujimoto H, Tamura N, Fukunaga Y, Sone M, Yamahara K, Taura D, Inuzuka M, Sonoyama T, Nakao K (2009) Natriuretic peptides/cGMP/cGMP-dependent protein kinase cascades promote muscle mitochondrial biogenesis and prevent obesity. Diabetes 58:2880–2892
Moro C, Klimcakova E, Lafontan M, Berlan M, Galitzky J (2007) Phosphodiesterase-5A and neutral endopeptidase activities in human adipocytes do not control atrial natriuretic peptide-mediated lipolysis. Br J Pharmacol 152:1102–1110, Epub 2007 Oct 1
Morroni M, Giordano A, Zingaretti MC, Boiani R, De Matteis R, Kahn BB, Nisoli E, Tonello C, Pisoschi C, Luchetti MM, Marelli M, Cinti S (2004) Reversible transdifferentiation of secretory epithelial cells into adipocytes in the mammary gland. Proc Natl Acad Sci U S A 101:16801–16806, Epub 2004 Nov 19
Mullur R, Liu YY, Brent GA (2014) Thyroid hormone regulation of metabolism. Physiol Rev 94:355–382
Murad F (2006) Shattuck Lecture. Nitric oxide and cyclic GMP in cell signaling and drug development. N Engl J Med 355:2003–2011
Nikolic DM, Li Y, Liu S, Wang S (2009) Overexpression of constitutively active PKG-I protects female, but not male mice from diet-induced obesity. Obesity 19:784–791
Nisoli E, Clementi E, Tonello C, Sciorati C, Briscini L, Carruba MO (1998) Effects of nitric oxide on proliferation and differentiation of rat brown adipocytes in primary cultures. Br J Pharmacol 125:888–894
Nisoli E, Clementi E, Paolucci C, Cozzi V, Tonello C, Sciorati C, Bracale R, Valerio A, Francolini M, Moncada S, Carruba MO (2003) Mitochondrial biogenesis in mammals: the role of endogenous nitric oxide. Science 299:896–899
Nisoli E, Falcone S, Tonello C, Cozzi V, Palomba L, Fiorani M, Pisconti A, Brunelli S, Cardile A, Francolini M, Cantoni O, Carruba MO, Moncada S, Clementi E (2004) Mitochondrial biogenesis by NO yields functionally active mitochondria in mammals. Proc Natl Acad Sci U S A 101:16507–16512
Petrovic N, Walden TB, Shabalina IG, Timmons JA, Cannon B, Nedergaard J (2010) Chronic peroxisome proliferator-activated receptor gamma (PPARgamma) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J Biol Chem 285:7153–7164
Pfeifer A, Hoffmann LS (2014) Brown, beige, and white: the new color code of fat and its pharmacological implications. Annu Rev Pharmacol Toxicol 55:207–227
Potter LR (2011) Guanylyl cyclase structure, function and regulation. Cell Signal 23:1921–1926
Robidoux J, Martin TL, Collins S (2004) Beta-adrenergic receptors and regulation of energy expenditure: a family affair. Annu Rev Pharmacol Toxicol 44:297–323
Rosen ED, Spiegelman BM (2014) What we talk about when we talk about fat. Cell 156:20–44
Rosenwald M, Wolfrum C (2014) The origin and definition of brite versus white and classical brown adipocytes. Adipocyte 3:4–9
Rosenwald M, Perdikari A, Rulicke T, Wolfrum C (2013) Bi-directional interconversion of brite and white adipocytes. Nat Cell Biol 15:659–667
Saini S, Duraisamy AJ, Bayen S, Vats P, Singh SB (2014) Role of BMP7 in appetite regulation, adipogenesis, and energy expenditure. Endocrine 48(2):405–409
Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T, Nio-Kobayashi J, Iwanaga T, Miyagawa M, Kameya T, Nakada K, Kawai Y, Tsujisaki M (2009) High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 58:1526–1531
Schlossmann J, Desch M (2009) cGK substrates. Handb Exp Pharmacol 163–193. doi:10.1007/978-3-540-68964-5_9
Schlueter N, de Sterke A, Willmes DM, Spranger J, Jordan J, Birkenfeld AL (2014) Metabolic actions of natriuretic peptides and therapeutic potential in the metabolic syndrome. Pharmacol Ther 144:12–27
Seale P, Bjork B, Yang W, Kajimura S, Chin S, Kuang S, Scime A, Devarakonda S, Conroe HM, Erdjument-Bromage H, Tempst P, Rudnicki MA, Beier DR, Spiegelman BM (2008) PRDM16 controls a brown fat/skeletal muscle switch. Nature 454:961–967
Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J, Cohen P, Cinti S, Spiegelman BM (2011) Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 121:96–105
Shimizu I, Aprahamian T, Kikuchi R, Shimizu A, Papanicolaou KN, MacLauchlan S, Maruyama S, Walsh K (2014) Vascular rarefaction mediates whitening of brown fat in obesity. J Clin Invest 124:2099–2112
Thompson CS (2013) Diabetic nephropathy: treatment with phosphodiesterase type 5 inhibitors. World J Diabetes 4:124–129. doi:10.4239/wjd.v4.i4.124
Tovar S, Paeger L, Hess S, Morgan DA, Hausen AC, Bronneke HS, Hampel B, Ackermann PJ, Evers N, Buning H, Wunderlich FT, Rahmouni K, Kloppenburg P, Bruning JC (2013) K(ATP)-channel-dependent regulation of catecholaminergic neurons controls BAT sympathetic nerve activity and energy homeostasis. Cell Metab 18:445–455
Tupone D, Madden CJ, Morrison SF (2014) Autonomic regulation of brown adipose tissue thermogenesis in health and disease: potential clinical applications for altering BAT thermogenesis. Front Neurosci 8:14. doi:10.3389/fnins.2014.00014, eCollection 2014
Turer AT, Scherer PE (2012) Adiponectin: mechanistic insights and clinical implications. Diabetologia 55:2319–2326
van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, Bouvy ND, Schrauwen P, Teule GJ (2009) Cold-activated brown adipose tissue in healthy men. N Engl J Med 360:1500–1508
Vaughan CH, Shrestha YB, Bartness TJ (2011) Characterization of a novel melanocortin receptor-containing node in the SNS outflow circuitry to brown adipose tissue involved in thermogenesis. Brain Res 1411:17–27
Vegiopoulos A, Muller-Decker K, Strzoda D, Schmitt I, Chichelnitskiy E, Ostertag A, Berriel Diaz M, Rozman J, Hrabe de Angelis M, Nusing RM, Meyer CW, Wahli W, Klingenspor M, Herzig S (2012) Cyclooxygenase-2 controls energy homeostasis in mice by de novo recruitment of brown adipocytes. Science 328:1158–1161
Vila G, Grimm G, Resl M, Heinisch B, Einwallner E, Esterbauer H, Dieplinger B, Mueller T, Luger A, Clodi M (2012) B-type natriuretic peptide modulates ghrelin, hunger, and satiety in healthy men. Diabetes 61:2592–2596
Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, Niemi T, Taittonen M, Laine J, Savisto NJ, Enerback S, Nuutila P (2009) Functional brown adipose tissue in healthy adults. N Engl J Med 360:1518–1525
Vosselman MJ, Brans B, van der Lans AA, Wierts R, van Baak MA, Mottaghy FM, Schrauwen P, van Marken Lichtenbelt WD (2013) Brown adipose tissue activity after a high-calorie meal in humans. Am J Clin Nutr 98:57–64
Wang X, Li J, Liu J, He W, Bi Y (2010) Nitric oxide increases mitochondrial respiration in a cGMP-dependent manner in the callus from Arabidopsis thaliana. Nitric Oxide 23:242–250
Whitaker RM, Wills LP, Stallons LJ, Schnellmann RG (2013) cGMP-selective phosphodiesterase inhibitors stimulate mitochondrial biogenesis and promote recovery from acute kidney injury. J Pharmacol Exp Ther 347:626–634
Whittle AJ, Carobbio S, Martins L, Slawik M, Hondares E, Vazquez MJ, Morgan D, Csikasz RI, Gallego R, Rodriguez-Cuenca S, Dale M, Virtue S, Villarroya F, Cannon B, Rahmouni K, Lopez M, Vidal-Puig A (2012) BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell 149:871–885
Wu J, Bostrom P, Sparks LM, Ye L, Choi JH, Giang AH, Khandekar M, Virtanen KA, Nuutila P, Schaart G, Huang K, Tu H, van Marken Lichtenbelt WD, Hoeks J, Enerback S, Schrauwen P, Spiegelman BM (2012) Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 150:366–376
Wu J, Cohen P, Spiegelman BM (2013) Adaptive thermogenesis in adipocytes: is beige the new brown? Genes Dev 27:234–250
Yamada-Goto N, Katsuura G, Ebihara K, Inuzuka M, Ochi Y, Yamashita Y, Kusakabe T, Yasoda A, Satoh-Asahara N, Ariyasu H, Hosoda K, Nakao K (2013) Intracerebroventricular administration of C-type natriuretic peptide suppresses food intake via activation of the melanocortin system in mice. Diabetes 62:1500–1504
Zhang X, Ji J, Yan G, Wu J, Sun X, Shen J, Jiang H, Wang H (2010) Sildenafil promotes adipogenesis through a PKG pathway. Biochem Biophys Res Commun 396:1054–1059
Zhang Z, Liu X, Morgan DA, Kuburas A, Thedens DR, Russo AF, Rahmouni K (2011) Neuronal receptor activity-modifying protein 1 promotes energy expenditure in mice. Diabetes 60:1063–1071
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Hoffmann, L.S., Larson, C.J., Pfeifer, A. (2015). cGMP and Brown Adipose Tissue. In: Herzig, S. (eds) Metabolic Control. Handbook of Experimental Pharmacology, vol 233. Springer, Cham. https://doi.org/10.1007/164_2015_3
Download citation
DOI: https://doi.org/10.1007/164_2015_3
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-29804-7
Online ISBN: 978-3-319-29806-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)