Abstract
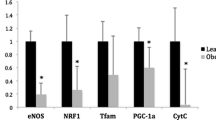
Objective: An excess of adipose tissue (AT) in obese individuals is linked to increased cardiovascular risk and mitochondria have been shown to be defective in the muscle and AT of patients with metabolic disorders such as obesity and Type 2 diabetes. Nitric oxide (NO) generated by endothelial NO synthase (eNOS) plays a role in mitochondrial biogenesis through cyclic-GMP (cGMP). AT harbors the whole molecular signaling pathway of NO, together with type 5-phosphodiesterase (PDE-5), the main cGMP catabolising enzyme. Aim: Our aim was to evaluate the effect of the modulation of NO pathway, through PDE-5 inhibition, on energy metabolism and mitochondria biogenesis in human omental AT. Methods and measurements: Cultured human omental AT was stimulated with PDE-5 inhibitor, vardenafil, at different concentration for 24 and 72 h. Analysis of the expression of both key-regulator genes of adipocyte metabolism and mitochondria- biogenesis markers was performed. Results: We found an increased gene expression of peroxisome proliferator-activated receptor-γ (PPAR-γ), adiponectin, and proliferator- activated receptor gamma coactivator- 1 α (PGC- 1α) after a 24- h stimulation with vardenafil at the lowest concentration employed compared to controls (p<0.05). After 72 h of stimulation, a significant increase of mitochondrial DNA was found compared to control samples (p<0.05). Conclusion: Our data suggest that PDE-5 inhibition could have an impact on mitochondrial content of human AT suggesting a positive effect on energy metabolism and adding new elements in the comprehension of AT pathophysiology.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Weiss R, Caprio S. The metabolic consequences of childhood obesity. Best Pract Res Clin Endocrinol Metab 2005, 19: 405–19.
Patti ME, Corvera S. The role of mitochondria in the pathogenesis of type 2 diabetes. Endocr Rev 2010, 31: 364–95.
Choo HJ, Kim JH, Kwon OB, et al. Mitochondria are impaired in the adipocytes of type 2 diabetic mice. Diabetologia 2006, 49: 784–91.
Lee S, Kim S, Sun X, Lee JH, Cho H. Cell cycle-dependent mitochondrial biogenesis and dynamics in mammalian cells. Biochem Biophys Res Commun 2007, 357: 111–7.
Nisoli E, Clementi E, Paolucci C, et al. Mitochondrial biogenesis in mammals: the role of endogenous nitric oxide. Science 2003, 299: 896–9.
Nisoli E, Falcone S, Tonello C, et al. Mitochondrial biogenesis by NO yields functionally active mitochondria in mammals. Proc Natl Acad Sci U S A 2004, 101: 16507–12.
Engeli S, Janke J, Gorzelniak K, et al. Regulation of the nitric oxide system in human adipose tissue. J Lipid Res 2004, 45: 1640–8.
Rybalkin SD, Yan C, Bornfeldt KE, Beavo JA. Cyclic GMP Phosphodiesterases and Regulation of Smooth Muscle Function. Circ Res 2003, 93: 280–91.
Enoksson S, Degerman E, Hagström-Toft E, Large V, Arner P. Various phosphodiesterase subtypes mediate the in vivo antilipolytic effect of insulin on adipose tissue and skeletal muscle in man. Diabetologia 1998, 41: 560–8.
Zhang X, Ji J, Yan G, et al. Sildenafil promotes adipogenesis through a PKG pathway. Biochem Biophys Res Commun 2010, 396: 1054–9.
Moro C, Klimcakova E, Lafontan M, Berlan M, Galitzky J. Phospho-diesterase-5A and neutral endopeptidase activities in human adipocytes do not control atrial natriuretic peptide-mediated lipolysis. Br J Pharmacol 2007, 152: 1102–10.
Selemidis S, Dusting GJ, Peshavariya H, Kemp-Harper BK, Drummond GR. Nitric oxide suppresses NADPH oxidase-dependent superoxide production by S-nitrosylation in human endothelial cells. Cardiovasc Res 2007, 75: 349–58.
Malena A, Loro M, Di Re M, Holt IJ, Vergani L. Inhibition of mitochondrial fission favours mutant over wild-type mitochondrial DNA. Hum Mol Gen 2009, 18: 3407–16.
Foresta C, Caretta N, Corona G, et al. Clinical and metabolic evaluation of subjects with erectile dysfunction: a review with a proposal flowchart. Int J Androl 2009, 32: 198–211.
Tontonoz P, Spiegelman BM. Fat and Beyond: The Diverse Biology of PPARgamma. Annu Rev Biochem 2008, 77: 289–312.
Goldstein BJ, Scalia RG, Ma XL. Protective vascular and myocardial effects of adiponectin. Nat Clin Pract Cardiovasc Med 2009, 6: 27–35.
Koh EH, Park JY, Park HS, et al. Essential role of mitochondrial function in adiponectin synthesis in adipocytes. Diabetes 2007, 56: 2973–81.
Koh EH, Kim M, Ranjan KC, et al. eNOS plays a major role in adiponectin synthesis in adipocytes. Am J Physiol Endocrinol Metab 2010, 298: E846–53.
Jahnke VE, Sabido O, Freyssenet D. Control of mitochondrial biogenesis, ROS level, and cytosolic Ca2+ concentration during the cell cycle and the onset of differentiation in L6E9 myoblasts. Am J Physiol Cell Physiol 2009, 296: C1185–94.
Muzaffar S, Shukla N, Angelici GD, Jeremy JY. Nitroaspirins and morpholinosydnonimine, but not aspirin, inhibit the formation of superoxide and the expression of gp91phox induced by endotoxin and cytokines in pig pulmonary artery vascular smooth muscle cells and endothelial cells. Circulation 2004, 110: 1140–7.
Turrens JF. Mitochondrial formation of reactive oxygen species. J Physiol 2003, 552: 335–44.
Zoraghi R, Francis SH, Corbin JD. Critical amino acids in phosphodiesterase-5 catalytic site that provide for high-affinity interaction with cyclic guanosine monophosphate and inhibitors. Biochemistry 2007, 46: 13554–63.
Tedesco L, Valerio A, Cervino C, et al. Cannabinoid type 1 receptor blockade promotes mitochondrial biogenesis through endothelial nitric oxide synthase expression in white adipocytes. Diabetes 2008, 57: 2028–36.
Miyashita K, Itoh H, Tsujimoto H, et al. Natriuretic peptides/cGMP/cGMP-dependent protein kinase cascades promote muscle mitochondrial biogenesis and prevent obesity. Diabetes 2009, 58: 2880–92.
Engeli S, Janke J, Gorzelniak K, et al. Regulation of the nitric oxide system in human adipose tissue. J Lipid Res 2004, 45: 1640–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Toni, L., Strapazzon, G., Gianesello, L. et al. Effects of type 5-phosphodiesterase inhibition on energy metabolism and mitochondrial biogenesis in human adipose tissue ex vivo . J Endocrinol Invest 34, 738–741 (2011). https://doi.org/10.1007/BF03346724
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346724