Abstract
Concepts such as biorefinery and green chemistry focus on the usage of biomass, as with the oil value chain. However, it can cause less negative impact on the environment. A biorefinery based on sugarcane (Saccharum spp.) as feedstock is an example, because it can integrate into the same physical space, of processes for obtaining biofuels (ethanol), chemicals (from sugars or ethanol), electricity, and heat.
The use of sugarcane as feedstock for biorefineries is dictated by its potential to supply sugars, ethanol, natural polymers or macromolecules, organic matter, and other compounds and materials. By means of conversion processes (chemical, biochemical, and thermochemical), sugarcane biomass can be transformed into high-value bioproducts to replace petrochemicals, as a bioeconomy model.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The economic development of different biomass chains is part of the agenda for R&D&I in most developed and developing countries, such as Germany, France, USA, Brazil, and South Korea, among others, mobilizing large amounts of public and private effort and resources, and aiming at the optimized utilization of biomass. It is a trend that seeks to add value to supply chains and to reduce negative environmental impacts. Brazil is one of the major biomass producers for food and biofuels, and therefore these efforts are of great importance for the maintenance of a positive and less impactful economic scenario.
Concepts such as biorefinery and green chemistry focus on the usage of biomass, as with the oil value chain. However, it causes less impact on the environment, considering sustainable integrated systems (feedstock-processes-products-residues) according to technical parameters that take into account, among other things, mass and energy balances and lifecycle analysis.
It is possible to observe a great synergy between biorefineries and green chemistry, especially regarding the minimization of waste and negative environmental impacts, as well as promoting a “green economy.” A biorefinery based on sugarcane as feedstock is an example, because it can integrate into the same physical space, of processes for obtaining biofuels (ethanol), chemicals (from sugars or ethanol), electricity, and heat.
We can consider on a scale of economic valuation [1] that fine chemicals and specialties developed from biomass are those with the greatest potential to add value on bioenergetic chains (as sugarcane). This is because of the strategic participation of the chemical industry in the supply of chemical commodities and final products to various economic sectors, such as petrochemical, pharmaceutical, automotive, construction, agroindustry, cosmetics, etc. Biofuels and materials are at a second level of valuation, followed by energy and chemical commodities such as fertilizers and pesticides.
In countries with a large production of sugarcane, such as Brazil, efforts have been made to promote the biomass economic potential, in accordance with the use of renewable sources to develop a sustainable chemistry as well as its use in biorefineries [2, 3]. It is important to mention that the use of residual biomass is crucial to enable the production of biofuels. Figure 1 shows the relationship between bioeconomy and renewable chemistry as a proposal for an innovative design to obtain good results for the usage of sugarcane in biorefineries.
In Brazil, sugarcane-biorefineries have a particularly great potential to share wealth based on a solid industrial leadership in this sector, when allied to technical and scientific development.
2 Sugarcane as a Feedstock for Biorefineries
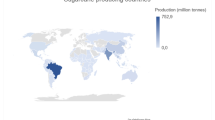
Table 1 describes the major worldwide sugarcane producers. As expected, the major producers are established in the tropical regions, excepted for the USA. We have a forecast of availability in these countries of 1.2 billion tonnes of sugarcane for biorefineries.
In Brazil, sugarcane is practically the only source of feedstock for sugar (sucrose) and its derivatives; in some countries, such as the USA, the major sugar source for ethanol is corn. Nowadays, the Brazilian production of ethanol (anhydrous and hydrated) is 27.5 million m3 and the production of commercial sugar is 37.3 million tonnes [4].
The use of sugarcane (Saccharum spp.) as feedstock for biorefineries is desirable because of its potential to supply the following compounds and materials:
-
Sugars: sucrose (Broth), glucose (derived from cellulose and sucrose inversion), and xylose (derived from hemicellulose)
-
Ethanol: from sucrose fermentation
-
Natural polymers or macromolecules: cellulose, hemicellulose, and lignin, which are present in bagasse and straw (lignocellulosic biomass); they can be converted into hexose, pentose, and phenol sources, respectively
-
Stillage or wastewater rich in organic matter
-
Other compounds, such as higher alcohols and carbon dioxide (CO2) at a high purity content for food and chemical usages
The average mass composition of the sugarcane plant is presented in Table 2.
Bagasse, as the main lignocellulosic biomass, has the following average mass composition [6]:
-
Cellulose: 41.7 wt%
-
Hemicellulose: 34 wt%
-
Lignin: 12.6 wt%
Sucrose, which is a disaccharide originating from fructose and glucose monosaccharides, is used for ethanol production by fermentation and for commercial sugar production through separation and crystallization. However, one could obtain other molecules of higher value from sucrose, glucose and xylose, constituents of cellulose and hemicellulose, respectively. Bagasse is frequently used for animal feeding and production of bioelectricity by cogeneration in Brazil; the sugarcane mills then are self-sufficient in electricity [7]. The use of bagasse and straw for second generation (2G) ethanol production is a topic that has a large amount of published literature. However, commercial production requires certain barriers to be overcome, such as reducing the cost of development of enzymes and yeasts that ferment the pentoses from hemicellulose [8]. Stillage has been used both in generating biogas and in soil fertigation. However, frequent monitoring of its application is required because of the high content of ions and organic matter, which can alter the physicochemical properties of the soil, with subsequent leaching of ions (NO3 −, PO4 3−, K+, etc.) and groundwater pollution [9].
Table 3 shows the main sugarcane components and derivatives with economic potential for biorefineries. Ethanol stands out as the main product because it can be obtained from three of the four sugarcane components (sucrose, bagasse, and straw). Furthermore, it can be used to produce other derivatives for renewable chemistry, such as “green” plastics. Figure 2 shows the product tree obtained from a sugarcane-biorefinery.
3 Perspectives for Sugarcane Usages in Biorefineries
A biorefinery incorporates the technologies and processes used for the conversion of biomass into five types of products: energy, chemical commodities or bulk chemicals, biofuels, materials, and fine chemicals. The technologies are compiled into processes, which are divided into biochemical (fermentation and enzymatic catalysis), chemical (chemo-catalytic), and thermochemical (combustion, gasification, and pyrolysis).
Table 4 presents a description of target products with high added value that can be obtained from sugarcane by means of different processes. In this table, only building blocks and end-use products can be observed. They were defined according to the characteristics of Brazilian biomass, data from national and international scientific literature, and demand from the national chemical industry and related industries. Building blocks are molecules from which a number of other chemical products of economic interest originate. Synthesis intermediates are those compounds used in small quantities as fine chemical reagents and in the production of drugs, pesticide, and cosmetics, among others. Chemical specialties must be considered, which are restricted to certain specific purposes.
Even with the laudable efforts of institutions such as the DOE-NREL (US Department of Energy – National Renewable Energy Laboratory) in R&D, product survey, and potential routes, most “green” compounds (or bioproducts) have not reached the stage of industrial maturity. A strong exception is succinic acid: considered as one of the main opportunities for renewable chemicals because of its possible application as building blocks [3, 10, 11] industrial production has started [12]. However, it is necessary to evaluate each molecule according to the market scenario, and two major aspects that must be considered are (1) commercial balance for chemicals (deficit or surplus) and (2) the need for synthesis intermediates, mainly for drugs, which may become more interesting than the search for building blocks, usually seen on the international scenario.
The current technological scenario, taken from the information shown in Tables 2 and 3, suggests a production of a wide range of products – as covered in Fig. 2 – in a model similar to an oil refinery. Furthermore, the American Chemical Society published in 2014 a revision of compounds from DOE-NREL’s forecasts considering building block molecules for biobased polymers [11], adding information to Table 4:
-
3-Hydroxypropionic acid from bacterial fermentation of glucose from sucrose – likely in 10 years for commercial step
-
Sorbitol from hydrogenation of glucose from sucrose – likely in 10 years
-
Levulinic acid from acid-catalyzed dehydration of sugars (hexose or pentose) – maybe likely in 10 years
The main processes to be applied in a sugarcane-biorefinery are described in the following.
3.1 Chemical Processes
Chemical processes are those based exclusively on chemical reactions (forming or breaking). In Fig. 2 it is possible to observe that organic synthesis is very relevant for biomass conversion. Usually a biomass component is extracted and purified and, subsequently, used as starting material in a synthetic route. Catalysts are often used to increase the yield of product of interest and to decrease the reaction time – the chemical processes are chemo-catalytic. This can be seen in several aspects of green chemistry, such as the use of renewable feedstock, catalysts, and the reduction of waste production [13].
In the case of utilization of cellulose and hemicellulose from lignocellulosic waste (bagasse and straw), first these polymers and their constituent sugars must be obtained, especially glucose (hexose) and xylose (pentose), respectively, to obtain products of industrial interest, such as ethanol 2G [14]. With lignin, the breaking of its molecular structure is initially attempted to release mainly phenolic compounds which may be tested, for example, as monomers for various routes of preparation. Obtaining building blocks and synthesis intermediates is the usual approach used by R&D projects to add greater value in bioenergetic chains in a biorefinery [3, 10, 15]. Building blocks, such as furfural and xylitol (derived from xylose constituent of hemicellulose) and 5-hydroxymethylfurfural (derived from glucose), among others, can add great value to carbohydrates, which can be extended to derivatives of lignin [1, 7, 10, 11, 14], as reported in Table 3.
It is worth commenting on the development and use of catalysts for these processes, given their importance for improving yields and selectivities (enantioselectivity, regioselectivity, and stereoselectivity). Zeolites have been used in the cracking of lignins [16]. The metals (soluble and insoluble salts and complexes) have been applied to heterogeneous catalysis (Ni, Pd/C, Ru/C, Co-Mo, Ni-Mo, Ru/Al2O3, etc.) to reduce lignins [10, 16] and metal complexes of V, Mn, Co, Pd, Fe, Re, and Cu acting as homogeneous and heterogeneous catalysts for cellulose oxidation, among other reactions [17].
3.2 Biochemical Processes
The biochemical processes in Fig. 2 are fermentation for the production of first generation ethanol (1G) and other chemicals, such as alcohols and other organic acids, and anaerobic digestion for production of biogas and biofertilizer (mineralized fraction). Enzymatic catalysis contributes to increase the rate of metabolic reactions involved in biochemical conversion routes.
Biochemical processes have major operational similarity to chemical processes with respect to the stages of compositional analysis and characterization of the feedstock, pre-treatment (whenever necessary), structural identification, and study of industrial potential. However, the main feature of these processes relates to the use of microorganisms (fungi, bacteria and yeast), which have the biochemical mechanisms that allow the synthesis of organic chemicals, such as ethanol. For example, production of ethanol (1G and 2G) by the yeast Saccharomyces cerevisiae by means fermentation of sucrose also produces carbon dioxide (CO2) [8], a carbon dioxide with a high purity degree and heat for industrial purposes.
Anaerobic fermentation of organic matter present in the vinasse mainly produces methane (biogas) and carbon dioxide, as well as mineral residue rich in inorganic salts of nitrogen, sulfur, and carbon (biofertilizer) [18]. In this case, bacterial consortia are used, formed by Acinetobacter, Arthrobacter, Bacillus, and Brevbacterium, among others [19].
The bacteria Lactobacillus delbrueckii can be used for the production of lactic acid via fermentation of glucose and Anaerobiospirillum succiniciproducens for the production of succinic acid through fermentation of pentoses and hexoses [10]. However, despite the high potential for production of a large amount of chemicals, the slow rate of reaction (kinetic of conversion) and the difficulty in separating final products (downstream step) can, in some cases, limit the use of bioprocesses in sugarcane-biorefineries for products other than ethanol.
On the other hand, enzymes, such as cellulase, β-glucosidase, and xylanase, are widely used in the hydrolysis of cellulose and hemicellulose to release glucose and xylose, respectively [8].
3.3 Thermochemical Processes
As for chemical and biochemical processes, the steps of compositional analysis and characterization of feedstocks and the structural identification and study of industrial potential are common. Nevertheless, the main characteristic of these processes is the use of thermal energy that leads to combustion, gasification, or pyrolysis. Through the combustion, it generates thermal energy (heat) [20]; through the gasification it generates synthesis gas (syngas), which is mainly composed of carbon monoxide (CO) and hydrogen (H2), to be used in organic synthesis of various molecules for the chemical industry [21] by Fisher–Tropsch reaction [22]. Fast pyrolysis produces bio-oil (rich in polar and non-polar organic compounds) and bio-char (a soil fertilizer). Cogeneration is a combined thermal process in which electrical energy is produced. Biomass combustion generates heat, which heats the water, generating steam, moving the turbines for the production of bioelectricity, which can either be used by the biorefinery itself or be sold to the electric grid.
As seen in Table 4, gasification is a process that can add value to the biomass waste. Syngas and fast pyrolysis can produce renewable chemicals to substitute for several petrochemicals.
4 Challenges Involved
As already mentioned, many of the target products shown in this chapter are subjects of extensive literature, mainly in the areas of organic chemistry, biotechnology, and chemical engineering. However, in some cases, there are no processes established for industrial production, which points scientific, technical, and economic challenges, separate or combined, which need to be overcome. It is important to consider, however, that the worldwide marketing of chemicals involves values around USD 100 billion per year, of which about 3% relates to bioproducts or biomass derivatives. It is estimated that the total market share could reach 25% by the year 2025 [23]. These figures give an idea of the possibilities and risks involved. For specialties and fine chemicals, the current participation of renewable – about 25% in both segments – could reach a market share of 50%, whereas for polymers, the current 10% could reach 20%, also in 2025 [24].
4.1 Technical Challenges
The technical challenges involve technological developments or improvements that allow the scale-up of processes developed in the laboratory such as separation methods, process optimization, energy efficiency, among others; a clear example of these observations is 2G ethanol.
Failure to overcome this kind of challenge can derail the production of certain bioproducts, as a value-added molecule, which can show great market potential in its R&D stage. Therefore, a well-planned R&D stage must have proper technological support to enable the laboratory scale to move on to an industrial scale.
4.2 Economic Challenges
One of the main economic challenges concerns attracting and allocating funds in R&D&I projects and, subsequently, in demonstration of technology projects. The rise and the possible decline of said “green” chemicals needs to be considered in the budget planning of development projects or for renewable compounds production, as previous international scenarios related to the chemical industry draw attention to external market factors [11, 24]. Nowadays, shale gas could be a threat for renewable chemicals based on its lower cost.
Nevertheless, the existing demand for ethanol 1G and the beginning of ethanol 2G production provides a favorable economic environment for the sugarcane-biorefinery.
5 Conclusion
This chapter attempts to show the great economic potential of sugarcane-biorefineries. Building blocks and synthesis intermediates are good examples of potential bioproducts in a medium and large horizon, integrated into ethanol, sugar, and bioenergy production. Energy and biofuel are very important in sugarcane industrial exploitation, in which the conceptual model is the petrochemical refinery.
When evaluating products mentioned as potential in other regions of the world, it should be noted that they do not always reflect local needs, justifying the use of own information obtained in studies conducted to adapt better for national scenarios, technical and scientific planning, and investment resources.
Chemical, biochemical, and thermochemical processes are fundamental to exploiting the full economic potential of sugarcane. However, strong actions are still required to overcome several challenges, comprising mainly pre-treatments, synthesis routes, catalysts, and microorganisms. However, is very important to establish an integrated sugarcane-biorefinery to achieve the real economic potential for the sugarcane biomass.
Scientific, technical, and economic challenges must be overcome by the government and private sector. It should allow sugarcane-biorefineries to turn into a viable alternative to the replacement of the productive chain based on oil – a non-renewable feedstock – leading to a positive environmental, social, and economic impact as a model of the bioeconomy based on renewable biomass.
References
Vaz S Jr (2014) Perspectives for the Brazilian residual biomass in renewable chemistry. Pure Appl Chem 86:833–842. doi:10.1515/pac-2013-0917
Centro de Gestão e Estudos Estratégicos (2010) Química verde no Brasil: 2010–2030. CGEE, Brasília, 438 pp
United States Department of Energy (2004) Top value added chemicals from biomass: results of screening for potential candidates from sugars and synthesis gas. US-DOE, Springfield, p 76
Brazilian Sugarcane Industry Association (2014) Unicadata. Available on: http://www.unicadata.com.br/. Accessed 14 Dec 2015
Food and Agriculture Organization of the United Nations (2013) Faostat. Available on: http://faostat3.fao.org/home/E. Accessed 14 Dec 2015
Dinardo-Miranda LL, Vasconcelos ACM, Landell MGA (eds) (2008) Cana-de-açúcar. Instituto Agronômico, Campinas, 882 pp
Vaz SJ (2014) A renewable chemistry linked to the Brazilian biofuel production. Chem Biol Technol Agric 1:13. doi:10.1186/s40538-014-0013-1
Sarkar N, Ghosh SK, Bannerjee S, Aikat K (2012) Bioethanol production from agricultural wastes: an overview. Renew Energy 37:19–27. doi:10.1016/j.renene.2011.06.045
Da Silva MAS, Griebeler NP, Borges LC (2007) Uso de vinhaça e impactos nas propriedades do solo e lençol freático. Revista Brasileira de Engenharia Agrícola e Ambiental 11:108-114. doi:10.1590/S1415-43662007000100014
Bozell JJ, Petersen GR (2010) Technology development for the production of biobased products from biorefinery carbohydrates - the US Department of Energy’s Top 10 revisited. Green Chem 12:539–554. doi:10.1039/B922014C
Bomgardner MM (2014) Biobased polymers. Chem Eng News 92:10–14
BioAmber (2015) Products. Available on: http://www.bio-amber.com/bioamber/en/products#succinic_acid. Accessed 14 Dec 2015
Anastas PT, Warner JC (1998) Green chemistry: theory and practice. Oxford University Press, New York, p 30
Kamm B, Gruber PR, Kamm M (2006) Biorefineries: industrial processes and products: status quo and future directions. Wiley-VCH, Weinheim, 406 pp
United States Department of Energy (2007) Top value added chemicals from biomass: results of screening for potential candidates from biorefinery lignin. US-DOE, Springfield, p 79
Zakzeski J, Bruijnincx PCA, Jongerius AL, Weckhuysen BM (2010) The catalytic valorization of lignin for the production of renewable chemicals. Chem Rev 110:3552–3599. doi:10.1021/cr900354u
Collinson SR, Thielemans W (2010) New materials focusing on starch, cellulose and lignin. Coord Chem Rev 254:1854–1870. doi:10.1016/j.ccr.2010.04.007
Salomon KR, Lora EES (2009) Estimate of the electric energy generating potential for different sources of biogas in Brazil. Biomass Bioenergy 33:1101–1107. doi:10.1016/j.biombioe.2009.03.001
Cutright TJ (2002) Biotechnology principles. In: Ghassemi A (ed) Handbook of pollution and waste minimization. Marcel Dekker, New York, pp 189–232
Nussbaumer T (2003) Combustion and co-combustion of biomass: fundamentals, technologies, and primary measures for emission reduction. Energy Fuels 17:1510–1521. doi:10.1021/ef030031q
Akay G, Jordan CA (2011) Gasification of fuel cane bagasse in a downdraft gasifier: influence of lignocellulosic composition and fuel particle size on syngas composition and yield. Energy Fuels 25:2274–2283. doi:10.1021/ef101494w
Gökalp I, Lebas E (2004) Alternative fuels for industrial gas turbines (AFTUR). Appl Therm Eng 24:1655–1663. doi:10.1016/j.applthermaleng.2003.10.035
Vijayendran BJ (2010) Bio products from bio refineries – trends, challenges and opportunities. J Bus Chem 7:109–115
Biotechnology Industry Organization (2010) Biobased chemicals and products: a new driver for green jobs. Available on: http://www.bio.org/articles/biobased-chemicals-and-products-new-driver-green-jobs. Accessed 14 Dec 2015
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Vaz, S. (2017). Sugarcane-Biorefinery. In: Wagemann, K., Tippkötter, N. (eds) Biorefineries. Advances in Biochemical Engineering/Biotechnology, vol 166. Springer, Cham. https://doi.org/10.1007/10_2016_70
Download citation
DOI: https://doi.org/10.1007/10_2016_70
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-97117-9
Online ISBN: 978-3-319-97119-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)