Abstrasct
Globally, food waste is a menacing issue, which is primely focused on Sustainable Development Goal (SDG) Target 12.3, aiming to cut down wastage by 2030. Thirty-five percent of the fish caught in fisheries and aquaculture are thrown away as fish waste and by-products. Fish processing waste accounts for about 70% of them, which can be turned into a valuable resource through a circular bioeconomy. The utilization of fish waste can also assuage the pressure on the fishery’s resources and nurture the sustainability of the realm. The valorization of fish waste and by-products is gaining significant attention as a source of nutrition besides being used as fodder or thrown away as waste. The by-products of fish typically include heads (9–12% of the total weight of the fish), bones (9–15%), scales (around 5%), viscera (12–18%), and skin (1–3%). Indeed, fish waste is justified to be a good source of protein (58% dry matter), fat (19%), and minerals. The potential techniques are being utilized to treat fish waste and recover valuable bioactive compounds. Therefore, this chapter underlines the enormous power of fish waste and its by-products.
A Chapter Submitted to Fish Waste to Valuable Products: Recent Applications and Research Update
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bioactive compounds
- By-products
- Circular bioeconomy
- Enzymes
- Fish waste treatment
- Nutrition
- Potential utilization
- Protein
- Sustainability
- Underutilization
1 Introduction
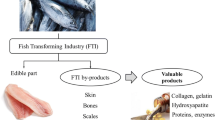
Nowadays, the fish production has tremendously increased all over the world. However, this negatively entails an increased fish waste generation. The fish waste and by-products usually consist of heads, bones, viscera, scales, and skin. The utilization of fish waste is low profitable due to the lack of proper knowledge about the final product or partly thrown away (Guillen et al. 2018). The better fish waste management paves the way for a circular bioeconomy. The circular bioeconomy aims at attaining the environment and terms of resources. The fundamental role of bioeconomy is bio-waste valorization. Fish waste is one of the potential starting materials due to its eminent nutritional composition and gains a promising market value among bio-waste (Shahidi et al. 2019). Hence, commercializing fish waste boosts economic development and reduces environmental issues. The ideology of the composition of fish waste entails the prior valorization and potential usage of fish waste. Therefore, this chapter provides a clear view of fish waste and its composition.
2 Fish Waste and Its Composition
A tremendous quantity of fish waste is generated as the fish processing industry expands, accounting for up to processed fish (70%) and total fish mass (¾) (Rustad et al. 2011). Fish by-products decompose quickly unless properly processed or placed in suitable circumstances due to heavier bacterial load and endogenic enzyme content, which can create severe environmental and food-technical concerns (Chalamaiah et al. 2012).
Stevens et al. (2018) stated that by-products are the leftovers (edible or non-edible) after manufacturing primary products. Heads, skins, frames, trimmings, viscera, and blood are common leftovers of finfish (Table 1).
Underutilization of Fish Waste
Scales, skin, bones, and fins are common fish processing waste. They are normally dumped on land and into the ocean. This practice results in the underutilization of raw materials and has an impact on the utilization of available resources. The disposal of these wastes causes several challenges for the environment (Fig. 1 and Table 2).
3 Techniques Involved in Treating Fish Waste
Fish waste is treated by several methods including physical, chemical, and biological methods. These approaches are being used to treat fish waste and turn leftover or discarded fish into useful by-products.
3.1 Mechanical/Physical Method
The following mechanical/physical methods are used to utilize the fish waste (Table 3).
3.2 Chemical Method
The following chemical methods are used to utilize the fish waste (Table 4).
3.3 Biological/Enzymatic Method
The following biological/enzymatic methods are used to utilize the fish waste (Table 5).
4 Potential Utilization of Fish Waste
4.1 Fish Silage
Fish silage is defined as the process of preserving entire fish by the addition of acid (chemical silage) or by the fermentation with sugar and microbial culture (biological silage). Acid/chemical silage method has been employed on a commercial scale in Denmark for more than 30 years. The fish silage production is popularly being utilized in Southeast Asia countries for the utilization of waste and contributes to the circular economy. Fish silage has an excellent protein value composed of hydrolyzed fish protein and micronutrients. They mostly consist of free amino acids and peptides such as methionine, histidine, glycine, alanine, and tyrosine (Xavier et al. 2017).
The raw material selection (Table 6) plays a pivotal role in arbitrating the quality of the silage. The raw material should be fresh and natural, ideally only after several hours of fish being processed into the by-products. However, the low-quality raw material can be utilized as fertilizer and not for feed preparation.
The main aim of fish silage production is to dually preserve the raw materials and augment the bioavailability of the nutrients. Thus, the fish silage paves an excellent pathway to reduce waste and convert them into a valuable product dually in terms of economy and nutrition. The microbial spoilage in the silage is prevented by lowering the pH, which is done by the addition of acid or in situ acid production. The presence of fermentable sugar initially prevents the deamination of amino acids by bacteria and in the case of biological silage. Later, the lactic acid bacteria predominate suppressing or killing the spoilage bacteria by lowering the pH and producing numerous antibiotic substances called bacteriocins.
The common ways involved in fish silage production are chemical silage and biological silage. In chemical silage, the preservation is carried out either by the addition of organic acids (formic, citric, and propionic acids), inorganic acids (hydrochloric, sulfuric, and phosphoric acid), or a mixture of both. Fish silage prepared using inorganic acid requires neutralization utilizing chalk or high salt levels, which is not nutritionally satisfactory. Therefore, organic acids like formic acid at 2–3% are more suitable for storing in a tropical climate. The biological silage is an alternative process in which a slurry is prepared using minced fish, 5–10% by weight of molasses, and 30% by weight of water to which Lactobacillus plantarum of 18–22 h old culture is inoculated, mixed well and allowed for fermentation process for 72 h. LAB helps in transforming the sugar molecules to lactic acid and resulted with the lowered pH (4.5), which in turn reduces the growth of spoilage bacteria (Xavier et al. 2017).
Fish Silage Production
Fish silage production is a simple process that does not require sophisticated equipment. However, proper and regular cleaning, checking, and maintenance of the equipment are vital to obtaining good quality silage. The equipment is differentiated from low-cost manual units to higher automated plants. The various components involved are a grinder, pumps, mixing tank, and storage tank. The grinder helps to grind the product to less than 1 mm particle size which ensures the penetration of preservative into the core particle. The pump assists in the proper circulation of the product to expose the particles to acid and enzymes which enables the transformation of fish into silage and the pumps are also used in transferring the silage from one place to another. The mixing tank which is made of acid-resistant material is used for mixing fresh raw material, acid, and antioxidant. The producers use sulfite (e.g., Potassium metabisulphite) to control the oxidation and growth of fungi. The storage tank should be resistant to corrosion and a weekly check of the contents in the storage tank (check of pH) should be carried out to prevent rotting across the tank pockets (Fig. 2).
The protective face shields and safety glasses should be worn throughout the process. Acid-resistant gloves and rubber boots should be worn during handling and mixing of acids with silage. The production of silage requires the knowledge of raw material and silage. This knowledge helps in predicting the behavior of each substance. The substantial physical properties of silage are viscosity, termination of hydrolysis, and heat convection coefficients (Arason 1994).
The optimal temperature between 5 and 40 °C should be maintained which has a good impact on hydrolyzing the proteins. High temperature leads to enzyme inactivation, while low temperature prolongs the process time. The undissolved bones at the bottom should be removed to arrest the pH rise which could lead to a rotting process of silage. Decantation of silage should be carried out to separate the oil. Antioxidants should be added along with acids to maintain the silage's quality. The fish silage could be stored for years without any prominent reduction in its nutritional quality. However, regular stirring and pH maintenance are essential.
The fish silage is rich in nutrients and has higher digestibility due to hydrolyzed proteins. The low acidity level of silage makes it suitable to be directly used as feed for pigs without any prior treatment resulting in higher growth rates. The inclusion of silage in feed ingredients produced stronger pellets thereby reducing the wastage during feeding and dust during transportation (Table 7).
4.2 Animal Feed
Animal feed from food/fish waste has gained increasing attention due to its multiple benefits to the human and environment also reducing the cost of feed production (Westendorf 2000). Fish/fishery waste has proven to possess essential protein, fat, and minerals. Fish waste has a plentiful number of fatty acids (monosaturated acids, oleic acids, and palmitic acid) and a low concentration of harmful substances (such as Hg, Pb, Cd, and As). The utilization of food waste as animal feed (protein source) has been studied and it has also been used as poultry and swine feed.
Fish wastes from white fish (haddock, cod, and plaice) are used to prepare intermediate moisture animal feed (Pig feed). The fish silage has a good amino acid profile which is favorable for monogastric animals, though it has less amount of lysine, methionine, and tryptophan when compared with the fish meal (Kjos et al. 1999). Therefore, they have been directly fed to pigs without any prior mixing and treatment. The pigs fed with fish silage have higher growth rates, good health, and reduced mortality.
Currently, fish waste is being used as a substrate for rearing insects (Hermetia illucens), which is further used as aquafeed ingredients for Asian sea bass (Chaklader et al. 2021).
4.3 Fish Sauce
In the past, the consumption of fermented fishery products has been on-trend. Fish sauce is a clear liquid (brown in color) seasoning that is utilized in Southeast Asia. It is basically produced by a combination of fish and salt (2:1 marine water fish, 3:1—freshwater fish) and allowed to fermentation for a duration of more than 6 months (30–35 °C). The resultant product is rich in flavor-active compounds which are developed during the progressive fermentation process (Zang et al. 2020).
The fish sauce is popular in Southeast Asia having different names depending on the country it has been manufactured (Table 8). For instance: Burma-Ngapi, China-Yeesui.
Thai fish (Nam pla) sauce is very popular in Western countries, specifically in the US when compared to other fish sauces. The fish sauce mostly produced in Thailand has expanded its domestic production to an international level over the last 5 decades.
The two methods involved in fish sauce production are the traditional and other is enzymatic methods. Traditionally, the fish is grounded and placed in jars made up of clay with layers of salt (fish to salt in the ratio of 3:1), this ratio depending on the country (Beddows et al., 1998). Then, they are buried in the ground and left to ferment for several months. The addition of proteolytic enzymes before fermentation is carried out in the enzymatic method. However, the price of proteolytic enzymes is very expensive which becomes a constraint for fish sauce production. In that case, alternative papaya sap which contains papain, a proteolytic enzyme, or young pineapples containing bromelain can be utilized. The enzymatically produced fish sauce has a different color and odor however the same nutritional content as traditional fish sauce. The filtration is done and the filtrate is further ripened under the sun (2–4 weeks) after the fermentation process to get top-grade fish sauce. Ultrafiltration (UF) has currently been used for recovering antioxidant peptides (Najafian and Babji 2019) in fish sauce production, whereas electrodialysis has been carried out for desalination (Ratanasanya et al. 2018).
Fish sauce is tremendously affected by the five major factors which include salt, fish, fish and salt ratio, oxygen level, and minor ingredients. The unique flavor and aroma of fish sauce are mainly caused due to the presence of volatile compounds, aspartic acid and glutamic acid (amino acids), nucleotides, peptides, and succinic acid (organic acid). The three major contributory factors of fish sauce aroma are ammoniacal, cheesy, and meaty notes. Fish sauce consists of halophilic bacteria due to the high level of salt content. The major roles of these bacteria are protein degradation and flavor-aroma development.
The quality of fish sauce varies based on the type of processing method and fish species. The minerals found in the fish sauce are potassium, sulfur, phosphorus, magnesium, calcium, and iron, etc. In addition, water-soluble vitamins such as B6, B12, thiamine, niacin, and riboflavin are presented in the sauce.
The chemical composition (pH, nitrogen content, and volatile acids) varies depending on manufacturing conditions. In general, the amino acid content increases due to polypeptide nitrogen breakdown (Table 9).
4.4 Biofuel
The fish waste consists of a good amount of fish oil. The yield generally depends on the fat level of fish species used, almost 50% of the body weight is utilized as waste. It possesses better quality fish oil, which can be utilized for the production of biofuel.
Nowadays, one of the greatest challenges is to maintain the global energy supply security. In that case, biofuel plays a viable role as an alternative replacement for conventional fuels. The biofuels obtained from the wastes are classified into two useful products: biodiesel and biogas (bio-methane). In chemical terms, biodiesel contains alkyl esters (long-chain fatty acids), whereas biogas is composed of methane and carbon dioxide. Biodiesel can be extracted using other methods such as trans-esterification, single-step trans-esterification using an alkaline catalyst, microwave-assisted lipid extraction, separation process, two-stage reaction process, and conventional processes including wet rendering and dry rendering. Biogas is produced during the anaerobic digestion of fish waste. Anaerobic digestion is a process in which various microorganism are involved and undergoes biodegradation. The fats, proteins, and carbohydrates present in a fish waste substrate have biologically degraded to biogas during this condition. Therefore, the yield of biogas depends on the content of the organic compounds. The microbes involved in the process are mostly acetogens and methanogens. The four steps involved in the decomposition process are hydrolysis, acidogenesis, acetogenesis, and methanogenesis.
In the hydrolysis step, the enzymes like amylases, lipases, proteases, cellulases, and hemicelluloses help in converting polysaccharides into small monomers, which paves the way for easier digestion for bacteria. This step is followed by acidogenesis, acetogenesis, and methanogenesis, where the methanogen bacteria convert the acetogenesis products to methane and carbon dioxide. The methane (by-product) is used as biogas. Several varieties of reactors are used in biogas production from fish waste.
Though fish waste has high valued organic carbon for methane production, the presence of a high amount of N2 and NH3 is limited. The anaerobic digestion of fish waste is feasible with co-digestion, in which the fish waste is mixed with slurry or other wastes hence biogas is produced. The limitation of co-digestion is maintaining the balance of macro and micronutrients (Yuvaraj et al. 2019) (Table 10).
4.5 Cosmetics
The regaining of chemical compounds from fish waste is a promising field of study to develop valuable by-products (cosmetics, nutraceuticals, and pharmaceuticals). Chitosan is generally extracted from shrimp and crab shells. However in recent times, the extraction of chitosan from scales is being extensively carried out (Kumari and Rath 2014). The chitosan has the ability to form films and poly oxy salts and possesses optical characteristics that make them attractive for utilization in cosmetic industries. The cationic and humectant properties of chitosan make a wiser application in skin and hair care products. In addition, chitosan is an excellent flocculent and adheres easily to hair and skin. Therefore, it has tremendous usage in the field of cosmetics. Although, the extraction of chitosan and chitin from fish scales has promising results, only limited study has been carried out for the characterization of these compounds.
Collagen obtained from fish waste has good antioxidant properties making it suitable for the cosmetic industry. Collagen peptides, an emerging substance obtained from fish waste prevent skin wrinkles and improve the elasticity of the skin. Moreover, a collagen drink prepared from fish is examined for improvement in the aging of the skin. The results showcased that collagen drink improved the cell viability of ultraviolet A irradiated human skin fibroblast and reduced reactive oxygen species production. In addition, it improved elastin and collagen production (Lin et al. 2020).
Gelatin is a derivative of its parent compound collagen. Gelatin is obtained by the partial hydrolysis of collagen. Gelatin is utilized as a gelling agent in face creams, bath salts, body lotions, and shampoos in the cosmetic industry (Elgadir et al. 2013). It protects against UV radiation by boosting immunity, thereby increasing hydroxyproline in the skin, which makes fish gelatin a novel ingredient for future skin anti-aging products.
4.6 Value-Added Products
4.6.1 Fish Protein Hydrolysate
FPH has prepared from fish/fish waste using the protein hydrolyzation process (the breaking down of proteins). Generally, FPH has a range of improved qualities including better functional and bioactive properties than anti-oxidative or anti-hypertensive activity (He et al. 2013). FPH has recently been employed as cryoprotectants for frozen fish items.
FPH is prepared in two types: liquid and dry. Liquid FPH is a watery combination of hydrolyzed proteins (90% moisture). FPH in the liquid state is exceedingly unstable for prolonged storage and is also difficult to transport. Therefore, dried FPH is preferred since it has a better shelf life than liquid FPH and is also easier to store and transport. However, in liquid FPH, the removal of large volumes of water is a difficult and expensive task, which is one of the problems faced by dried FPH manufacturers. Thus, the FPH has enough protein sources for human consumption. However, the process of dehydration requires a large amount of energy and is thus quite expensive (Petrova et al. 2018).
Extraction of FPH
FPH is produced by various techniques such as autolysis, chemical hydrolysis (acid and alkaline hydrolysis), enzymatic hydrolysis, and bacterial fermentation. Chemical hydrolysis and enzymatic hydrolysis are the most often utilized procedures due to their numerous advantages. The chemical hydrolysis technique is affordable, quick, and yields high protein recovery. Nevertheless, there is limited control over the uniformity of hydrolyzed products with considerable variability in the free amino acid profile due to non-specific peptide bond breaking (Siddik et al. 2021).
Enzymatic hydrolysis is extensively used to manufacture precise hydrolysates that maintain the nutritional content of the source protein in order to make high-grade FPH (Zamora-Sillero et al. 2018). This method requires minimal reaction time and is effective in targeting certain peptide bonds and amino acids with optimal activity under certain circumstances. Furthermore, the final product of enzymatic hydrolysis does not include any leftover organic solvents or hazardous compounds (Najafian and Babji 2012).
Debittering of FPHs: Fish protein hydrolysate is quite bitter in taste. It is plausibly caused by certain peptides present in it. An enzyme (acidic serine carboxypeptidase) isolated from the hepatopancreas of the squid can reduce the bitterness of FPH by eliminating certain amino acids in the carboxy terminal (Komai et al. 2007) (Fig. 3).
Commercial Significance of FPH
FPH is used as a flavor enhancer in dietic foods such as macaroni, meringues, souffles, or bread. However, in the preparation of fish paste, fish soup, and shellfish analogs, which is used as the source of small amino acids and peptides (Muzaddadi et al. 2016). FPH is reported to be an effective growth promoter for different kinds of bacteria, yeasts, and molds due to the high quantity of amino acids, peptones, and other important elements (Guerard et al. 2001). FPH has improved antioxidant, anticancer, immunomodulatory, anticoagulant, anti-obesity, and anti-hypertensive properties and is widely employed in nutraceuticals. Vasotensin, Amizate, Protizen, Seacure, and Molval are commercial nutraceutical products of FPH marketed in several countries (Elavarasan 2019) (Table 11).
4.6.2 Collagen/gelatin
Collagen is the fibrous protein and presents in the extracellular matrix of the body's connective tissues (i.e., bones, skin, tendons, ligaments, and cartilage) (Muller et al. 2003). It is the only protein that is abundant in the animal kingdom. Plants and unicellular creatures lack it, as polysaccharides and cellulose fill the void. Generally, collagen is found in the body walls and cuticles of invertebrates (Silvipriya et al. 2015). Collagen's fundamental structural unit is made up of three polypeptide chains. Each chain is 300 nm long and includes 1050 amino acids twisted around one another in a typical right-handed helical shape. It has a molecular weight of about 290 kDa and a diameter of around 1.5 nm (Silvipriya et al. 2015). In general, collagen is extracted from the skin, bones, scales, fins, and swim bladder of fishes.
Collagen from fish has two α-chain variants such as α1 and α2 (Gómez-Guillén et al. 2002). Changes in the amino acid composition of α chain types, end up with small differences in hydrophobicity. Despite having the same molecular weight (95,000 Da), these chain variants can be separated by SDS-PAGE because of their differing affinity.
Gelatin—A derivative of collagen
Gelatin is eventually obtained by heating the collagen over the transition temperature of the triple-helix structure commonly known as “superhelix.” A three-peptide strand with a helix structure forms the superhelix (Ward and Courts 1977). The collagen molecule's helical shape collapses; subsequently, the molecular chains unfurl and decrease in molecular weight and such changes occur in a very narrow range of temperatures (Rahman et al. 2008). This treatment is required to destabilize the protein's non-covalent bonding and results in sufficient swelling and rupture of inter- and intra-molecular bonds, which leads to collagen solubilization (Gómez-Guillén et al. 2002) and gelatin conversion with improved hydration capacity (Mohtar et al. 2010).
Extraction of gelatin
Commercial significance of gelatin
Gelatin is a significant biodegradable polymer and is extensively used in the food, pharmaceutical, cosmetics, and photographical industries due to its effectiveness and technological capabilities. However, gelatin is not a naturally occurring protein. This heterogeneous protein has outstanding film-forming capability, high flexibility, good processability, effective gas barrier properties, high availability, and cheap cost (Mousazadeh et al. 2021) (Fig. 4).
In the food industry, fish gelatin is a highly adaptable food additive generally employed to increase the stability, consistency, and elasticity of food and is utilized in the confectionery, packaging, and the dairy processing industry. Gelatin is typically advised to be used in foods to lower the carbohydrate content and increase the protein content in diets designed for diabetes people because it has minimal calories.
In the pharmaceutical industry, fish gelatin is widely used for the production of numerous products such as ointments, cosmetics, capsules, tablet coatings, and emulsions. In addition, fish gelatin can be used in the production of microencapsulated foods and other pharmaceutical additives.
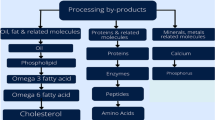
4.6.3 Proteins, Minerals, and Enzymes
Proteins
Muscle proteins are highly nutritious and readily digestible and abundant in fish frames. As a result, rather than being dumped as trash, proteins from fish waste may be recovered using several techniques. Over the years, three techniques have been used to recover protein from fish processing waste. They have aimed to break the peptide bonds from the encrypted mother proteins or isolate the protein using pH changes. Li-Chan (2015) used the strategy to produce peptides and bioactive protein hydrolysates in one of the following three ways, namely chemical, enzymatic, or fermentation techniques. The recovery method used for fish protein is influenced by the product cost, quality, and time (Ananey-Obiri et al. 2019).
Enzymatic Hydrolysis: The most prevalent technique of recovering protein from seafood processing waste is usually by enzymatic or proteolytic hydrolysis. This process is carried out naturally with proteolytic enzymes or expedited and regulated by exogenous enzymes.
Chemical Hydrolysis: The chemical approach of recovering fish protein entails breaking down the proteins into peptides. In general, this process of recovering proteins is quite easy and affordable. However, it is a challenging process to manage due to the undefined peptide breakage during hydrolysis. In acid hydrolysis, HCl or H2SO4 is used to hydrolyze the proteins at high temperatures and also at high pressure (Kristinsson and Rasco 2000).
Fermentation Hydrolysis: Microorganisms are used in fermentation to break down fish proteins into amino acids and peptides. A variety of microbes have been found and employed to extract fish protein. The functionality of FPHs recovered by this technique might vary due to changes in microbes used in the fermentation culture (Daliri et al. 2017). In this method, protein hydrolysates have been produced by using lactic acid bacteria (LAB) Pediococcus acidilactici NCIM5368 (Chakka et al. 2015) and Enterococcus faecium NCIM5335 (Balakrishnan et al. 2011).
Minerals
Fish bones have generally been separated after the removal of muscle proteins from the frames. Minerals such as calcium, hydroxyapatite, and phosphorus are found in 60–70% of fish bones (Kim and Mendis 2006). Generally, a lower amount of calcium is present in most people's diets. Therefore, the consumption of smaller fishes can help to increase the level of calcium in the human body. The fish bones obtained from the processing industry can be used to improve calcium levels in our body. Bones should be converted into edible form before being used as a fortified meal (Ishikawa et al. 1990). Fish bones are an excellent source of Ca10(PO4)6(OH)2 (hydroxyapatite) that can be used for the medical application. In the past, bone fractures are repaired using autografts, xenografts, and allografts. However, these materials are proven to be inefficient owing to mechanical instability and incompatibility.
Enzymes
Fishery waste such as head, liver, viscera, and shell are abundant in enzymes such as proteases, alkaline phosphatase, chitinase, and hyaluronidase. These enzymes are mostly abundant in the intestines, followed by pancreatic tissues, pyloric ceca, hepatopancreas, shell, and other waste components (Sriket 2014). It must be essential in relatively large quantities and acceptable quality for enzyme recovery. The commercially viable enzyme separation techniques from crude waste extracts are ultrafiltration, precipitation by salts and polyacrylic acids, isoelectric solubilization/precipitation, pH shift, overcooled acetone extraction, flocculation and membrane filtration (Benjakul et al. 2010).
Proteases are the digestive enzymes usually found in the digestive system of marine animals. It can be classified as endopeptidases or exopeptidases based on the hydrolysis of a protein molecule. Other digestive enzymes including trypsin, pepsin, gastricsin, chymotrypsin, elastase, carboxypeptidase, collagenase, and carboxyl esterase are also found in marine animals.
Lipases are a diverse group of enzymes that catalyze the hydrolysis of ester bonds in various substrates including phospholipids, triglycerides, vitamin esters, and cholesteryl esters (Venugopal 2016). Hydrolytic enzymes known as chitinases are capable of dissolving the glycosidic linkages found in chitin polymers. True chitinases are divided into chitinase and chitobiase and these enzymes are commonly found in species that must either remodel their own chitin or digest chitinous material consumed as food. Animals that feed on worms and arthropods require chitinases as their primary food source to digest their diet (Kim and Dewapriya 2014).
Other enzymes: Hyaluronidase, an animal tissue constituent that breaks hyaluronate to improve tissue permeability is recovered from shrimp shells which found in shallow tropical seas. Alkaline phosphatase, nucleoside phosphorylase, 5′-nucleotidase, urease, xanthine oxidase, trimethylamine oxide (TMAO) reductase, and TMAO-demethylase are enzymes that have been used in seafood quality evaluation (Benjakul et al. 2010) (Table 12).
4.6.4 Bioactive Compounds
Fish is a vital source of nutraceuticals due to its unique combination of bioactive compounds such as long-chain PUFAs (DHA and EPA), ω-3 PUFAs, peptides, protein hydrolysates, minerals, amino acids, vitamins, collagen, gelatin, fish bone, fish oil and fat-soluble vitamins (Kundam et al. 2018). Numerous nutraceuticals are used for preventative reasons in several sectors of medicine due to the high therapeutic value of seafood by-products. They can be used to reduce complications or treat a variety of disorders, including cardiovascular disease, cancer, rickets, viral infections, hypertension, dermatologic issues, particularly during pregnancy, and parasite infections (Siddiqui et al. 2015). In addition to these advantages, fish-derived nutraceuticals have anti-inflammatory, anticoagulant, and antioxidant properties (Tables 13 and 14).
-
Intestine waste of fish species such as mackerel, black halibut, spiny dogfish, arctic char, salmon, fried calamari, sardine, and anchovies are rich in bioactive components nutraceuticals such as ω-3 and ω-6 fatty acids. It has a myriad of medical applications particularly therapeutic and clinical applications such as improving insulin sensitivity, anti-inflammatory agent, reducing the risk of cardiovascular problems, increasing vascular adhesion molecule-1 expression, and improving eyesight (Ashraf et al., 2020).
-
Chitin and chitosan are recovered from the crustacean. It accelerates the healing of wounds, lowers blood cholesterol, acts as an anti-ulcer and anti-aging agent, and is used in cosmetics and ophthalmology.
-
Chondroitin recovered from shark cartilage reduces the risk of osteoarthritis and supports the nutritional supplement.
-
Glucosamine recovered from shellfish shells acts as an anti-inflammatory agent, reduces the risk of osteoarthritis, and supports the dietary supplement.
-
Body walls and cuticles of invertebrates are rich in collagen. It reduces the risk of osteoarthritis, reduces hypertension, plays a vital role in tissue engineering, and acts as an antioxidant, anti-hypertensive, and anti-skin aging.
-
Hyaluronidase is an endo-enzyme that is extracted from shellfish waste and has enormous therapeutic applications.
-
Squalene extracted from shark liver reduces the risk of cardiovascular problems and acts as an antioxidant, anti-bacterial, antifungal, and anti-cancerous agent.
-
Squalamine extracted from the liver, gallbladder, and intestine waste of sharks is effective against bacteria and fungi and plays a vital role in tumor angiogenesis activities.
-
Astaxanthin, β-carotene, lutein, and Zeaxanthin are rich in freshwater fish, red fishes, and other fishes. It has antioxidant and anti-atherogenic properties, kills cancer cells, reduces the risk of neurological, cardiovascular problems, and psoriasis, and also plays a vital role in cosmetics.
5 Conclusion
The success of any food realm depends on sensible waste management. Fish being a highly perishable product needs more attention for waste management in the fishery industry. Fish wastes are rich in a number of novel components which include bioactive compounds, peptides, enzymes, etc. The effective utilization of the resourceful fish waste and its by-products enables the cut-off in dumping the wastes in aquatic environments which leads to eutrophication. Fish waste valorization plays a major role in achieving one of the Sustainable Development Goals’ 2030 – ‘zero discards.’ It also paves the way to attain a sustainable circular economy by eminent utilization of other discarded fish wastes and by-products in the food chain. To date, there are several technologies to convert fish waste into valuable resources used in a myriad of industries paving the way to attaining a green approach in the fisheries industry. In a nutshell, the conversion of fish wastes into high-value products is promising and the intervention of food industries can bring a way forward in the future, in terms of product diversification, resource mobilization, and sustainable growth.
References
Ahmad M, Benjakul S (2010) Extraction and characterisation of pepsin-solubilised collagen from the skin of unicorn leatherjacket (Aluterus monocerous). Food Chem 120(3):817–824
Akter S, Rahman MA, Bhowmik S, Haidar MI, Alam AN (2016) Assessment of fishery wastes and suitability of its utilization in the manufacture of fish glue. Am J Food Nutrition 6:77–81
Ananey-Obiri D, Matthews LG, Tahergorabi R (2019) Proteins from fish processing by-products. In: Proteins: Sustainable source, processing and applications. Academic Press, pp 163–191
Aoki H, Ahsan MN, Matsuo K, Hagiwara T, Watabe S (2004) Partial purification of proteases that are generated by processing of the Northern shrimp Pandalus borealis and which can tenderize beef. Int J Food Sci Technol 39(5):471–480
Arason S (1994) Production of fish silage. In: Fisheries processing. Springer, Boston, MA, pp 244–272
Ashraf SA, Adnan M, Patel M, Siddiqui AJ, Sachidanandan M, Snoussi M, Hadi S (2020) Fish-based bioactives as potent nutraceuticals: exploring the therapeutic perspective of sustainable food from the sea. Mar Drugs 18(5):265
Balachandran KK (2001) Post-harvest technology of fish and fish products. Daya Books
Balakrishnan B, Prasad B, Rai AK, Velappan SP, Subbanna MN, Narayan B (2011) In vitro antioxidant and antibacterial properties of hydrolysed proteins of delimed tannery fleshings: comparison of acid hydrolysis and fermentation methods. Biodegradation 22(2):287–295
Beddows CG (1998) Fermented fish and fish products. In: Microbiology of fermented foods. Springer, Boston, MA, pp 416–440
Bekhit AEA (2011) Fish Roe: Fermentation. Encyclopedia of biotechnology in agriculture and food
Benjakul S, Klomklao S, Simpson BK (2010) 10 Enzymes in fish processing. Enzymes Food Technol 211
Chakka AK, Elias M, Jini R, Sakhare PZ, Bhaskar N (2015) In-vitro antioxidant and antibacterial properties of fermentatively and enzymatically prepared chicken liver protein hydrolysates. J Food Sci Technol 52(12):8059–8067
Chaklader MR, Howieson J, Foysal MJ, Fotedar R (2021) Transformation of fish waste protein to Hermetia illucens protein improves the efficacy of poultry by-products in the culture of juvenile barramundi, Lates calcarifer. Sci Total Environ 796:149045
Chalamaiah M, Hemalatha R, Jyothirmayi T (2012) Fish protein hydrolysates: proximate composition, amino acid composition, antioxidant activities and applications: a review. Food Chem 135(4):3020–3038
Chandrasekaran, M. ed., 2012. Valorization of food processing by-products. CRC press
Daliri EBM, Lee BH, Oh DH (2017) Current perspectives on antihypertensive probiotics. Probiotics Antimicrob Proteins 9(2):91–101
Elavarasan K (2019) Health benefits and potential applications of fish protein hydrolysate. ICAR-Central Institute of Fisheries Technology
Elgadir AM, Mirghani ME, Adam A (2013) Fish gelatin and its applications in selected pharmaceutical aspects as alternative source to pork gelatin. J Food Agric Environ 11:73–79
Falch E, Rustad T, Jonsdottir R, Shaw NB, Dumay J, Berge JP, Arason S, Kerry JP, Sandbakk M, Aursand M (2006) Geographical and seasonal differences in lipid composition and relative weight of by-products from gadiform species. J Food Compos Anal 19(6–7):727–736
FAO (2013) Fisheries and aquaculture information and statistics service. Food and Agricultural Organization, Rome, Italy http://www.fao.org/figis/servlet/TabSelector?tb_ds = Production and tb_mode = Table and tb_act = Select and tb_grp = Country
Gildberg A (1993) Enzymic processing of marine raw materials. Process Biochem 28(1):1–15
Gildberg AR (2004) Enzymes and bioactive peptides from fish waste related to fish silage, fish feed and fish sauce production. J Aquat Food Prod Technol 13(2):3–11
Gómez-Guillén MC, Turnay J, Fernández-Dıaz MD, Ulmo N, Lizarbe MA, Montero P (2002) Structural and physical properties of gelatin extracted from different marine species: a comparative study. Food Hydrocolloids 16(1):25–34
Gopakumar K (2002) Textbook of fish processing technology
Govidan TK (1982) Fish processing technology. CIFT, Kochi
Guerard F, Dufosse L, De La Broise D, Binet A (2001) Enzymatic hydrolysis of proteins from yellowfin tuna (Thunnus albacares) wastes using Alcalase. J Mol Catal B Enzym 11(4–6):1051–1059
Guillen J, Holmes SJ, Carvalho N, Casey J, Dörner H, Gibin M, Mannini A, Vasilakopoulos P, Zanzi A (2018) A review of the European Union landing obligation focusing on its implications for fisheries and the environment. Sustainability 10(4):900
He S, Franco C, Zhang W (2013) Functions, applications and production of protein hydrolysates from fish processing co-products (FPCP). Food Res Int 50(1):289–297
Hertrampf JW, Piedad-Pascual F (2000) Fish solubles (de-hydrated). In: Handbook on ingredients for aquaculture feeds. Springer, Dordrecht, pp 211–216
Ishikawa M, Kato M, Mihori T, Watanabe H, Sakai Y (1990) Effect of vapor pressure on the rate of softening of fish bone by super-heated steam cooking. Nion Suisan Gakkaishi 56(10):1687–1691
Javeed A, Mahendrakar NS (1995) Effect of different levels of molasses and salt on acid production and volume of fermenting mass during ensiling of tropical freshwater fish viscera. J Food Sci Technol 32(2):115–118
Karim AA, Bhat R (2009) Fish gelatin: properties, challenges, and prospects as an alternative to mammalian gelatins. Food Hydrocolloids 23(3):563–576
Kim SK, Dewapriya P (2014) Enzymes from fish processing waste materials and their commercial applications. In: Seafood processing by-products. Springer, New York, NY, pp. 183–196
Kim SK, Mendis E (2006) Bioactive compounds from marine processing byproducts–a review. Food Res Int 39(4):383–393
Kjos NP, Skrede A, Øverland M (1999) Effects of dietary fish silage and fish fat on growth performance and sensory quality of growing-finishing pigs. Can J Anim Sci 79(2):139–147
Komai T, Kawabata C, Tojo H, Gocho S, Ichishima E (2007) Purification of serine carboxypeptidase from the hepatopancreas of Japanese common squid Todarodes pacificus and its application for elimination of bitterness from bitter peptides. Fish Sci 73(2):404–411
Kristinsson HG, Rasco BA (2000) Fish protein hydrolysates: production, biochemical, and functional properties. Crit Rev Food Sci Nutr 40(1):43–81
Kumari S, Rath PK (2014) Extraction and characterization of chitin and chitosan from (Labeo rohita) fish scales. Proc Mater Sci 6:482–489
Kundam DN, Acham IO, Girgih AT (2018) Bioactive compounds in fish and their health benefits. Asian Food Sci. J 4(4):1–14
Li-Chan EC (2015) Bioactive peptides and protein hydrolysates: research trends and challenges for application as nutraceuticals and functional food ingredients. Curr Opin Food Sci 1:28–37
Lin P, Hua N, Hsu YC, Kan KW, Chen JH, Lin YH, Lin YH, Kuan CM (2020) Oral collagen drink for antiaging: antioxidation, facilitation of the increase of collagen synthesis, and improvement of protein folding and DNA repair in human skin whfibroblasts. Oxidat Med Cell Longevity
Lopetcharat K, Choi YJ, Park JW, Daeschel MA (2001) Fish sauce products and manufacturing: a review. Food Rev Intl 17(1):65–88
Melendo JA, Beltrán JA, Roncalés P (1997) Tenderization of squid (Loligo vulgaris and Illex coindetii) with bromelain and a bovine spleen lysosomal-enriched extract. Food Res Int 30(5):335–341
Mohtar NF, Perera C, Quek SY (2010) Optimisation of gelatine extraction from hoki (Macruronus novaezelandiae) skins and measurement of gel strength and SDS–PAGE. Food Chem 122(1):307–313
Mousazadeh S, Ehsani A, Kia EM, Ghasempour Z (2021) Zinc oxide nanoparticles and periodate oxidation in developing pH-sensitive packaging film based on modified gelatin. Food Packag Shelf Life 28:100654
Muller WE (2003) The origin of metazoan complexity: Porifera as integrated animals. Integr Comp Biol 43(1):3–10
Muzaddadi AU, Devatkal S, Oberoi HS (2016) Seafood enzymes and their application in food processing. In: Agro-industrial wastes as feedstock for enzyme production. Academic Press, pp. 201–232
Myrnes B, Johansen A (1994) Recovery of lysozyme from scallop waste. Prep Biochem Biotechnol 24(1):69–80
Najafian L, Babji AS (2012) A review of fish-derived antioxidant and antimicrobial peptides: their production, assessment, and applications. Peptides 33(1):178–185
Najafian L, Babji AS (2019) Purification and identification of antioxidant peptides from fermented fish sauce (Budu). J Aquat Food Prod Technol 28(1):14–24
Naung SHH, Hpa-an M (2019) Foliar fertilizer production from waste fish
Pedziwiatr P (2017) Aquaculture waste management. Acta Innov 22:20–29
Petrova I, Tolstorebrov I, Eikevik TM (2018) Production of fish protein hydrolysates step by step: technological aspects, equipment used, major energy costs and methods of their minimizing. Int Aquatic Res 10(3):223–241
Raa J, Gildberg A, Olley JN (1982) Fish silage: a review. Crit Rev Food Sci Nutr 16(4):383–419
Raghunath MR, Gopakumar K (2002) Trends in production and utilization of fish silage. J Food Sci Technol (Mysore) 39(2):103–110
Rahman MS, Al-Saidi GS, Guizani N (2008) Thermal characterisation of gelatin extracted from yellowfin tuna skin and commercial mammalian gelatin. Food Chem 108(2):472–481
Ratanasanya S, Chindapan N, Polvichai J, Sirinaovakul B, Devahastin S (2018) Particle swarm optimization as alternative tool to sensory evaluation to produce high-quality low-sodium fish sauce via electrodialysis. J Food Eng 228:84–90
Rustad T, Storrø I, Slizyte R (2011) Possibilities for the utilisation of marine by-products. Int J Food Sci Technol 46(10):2001–2014
Sachindra NM, Mahendrakar NS (2014) Fish processing byproducts (Quality Assessment And Applications). Studium Press
Shahidi F, Varatharajan V, Peng H, Senadheera R (2019) Utilization of marine by-products for the recovery of value-added products. J Food Bioactives 6
Siddik MA, Howieson J, Fotedar R, Partridge GJ (2021) Enzymatic fish protein hydrolysates in finfish aquaculture: a review. Rev Aquac 13(1):406–430
Siddiqui AJ, Bhardwaj J, Goyal M, Prakash K, Soni A, Tiwari V, Puri SK (2015) Assessment of real-time method to detect liver parasite burden under different experimental conditions in mice infected with Plasmodium yoelii sporozoites. Microb Pathog 89:35–42
Silvipriya KS, Kumar KK, Bhat AR, Kumar BD, John A (2015) Collagen: Animal sources and biomedical application. J Appl Pharmaceut Sci 5(3):123–127
Sriket C (2014) Proteases in fish and shellfish: Role on muscle softening and prevention. Int Food Res J 21(2)
Stevens JR, Newton RW, Tlusty M, Little DC (2018) The rise of aquaculture by-products: Increasing food production, value, and sustainability through strategic utilisation. Mar Policy 90:115–124
Synowiecki J, Al-Khateeb NA (2003) Production, properties, and some new applications of chitin and its derivatives
Varun TK, Senani S, Jayapal N, Chikkerur J, Roy S, Tekulapally VB, Gautam M, Kumar N (2017) Extraction of chitosan and its oligomers from shrimp shell waste, their characterization and antimicrobial effect. Veterinary World 10(2):170
Venugopal V (2016) Enzymes from seafood processing waste and their applications in seafood processing. Adv Food Nutr Res 78:47–69
Ward AG, Courts A (1977) Science and technology of gelatin. Academic press
Westendorf ML (2000) Food waste as animal feed: an introduction. Food waste to animal feed, pp 3–16
Wheaton FW, Lawson TB (1985) Processing aquatic food products. John Wiley and Sons, New York, USA
Xavier MKA, Geethalekshmi V, Senapati SR, Mathew PT, Joseph AC, Ramachandran Nair KG (2017) Valorization of squid processing waste as animal feed ingredient by acid ensilaging process. Waste Biomass Valoriz 8(6):2009–2015
Yuvaraj D, Bharathiraja B, Rithika J, Dhanasree S, Ezhilarasi V, Lavanya A, Praveenkumar R (2019) Production of biofuels from fish wastes: an overview. Biofuels 10(3):301–307
Zamora-Sillero J, Ramos P, Monserrat JM, Prentice C (2018) Evaluation of the antioxidant activity in vitro and in hippocampal HT-22 cells system of protein hydrolysates of common carp (Cyprinus carpio) by-product. J Aquat Food Prod Technol 27(1):21–34
Zang J, Xu Y, Xia W, Regenstein JM (2020) Quality, functionality, and microbiology of fermented fish: a review. Crit Rev Food Sci Nutr 60(7):1228–1242
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Murugan, G., Ahilan, K., Prakasam, V.P.A., Malreddy, J., Benjakul, S., Nagarajan, M. (2024). Fish Waste Composition and Classification. In: Maqsood, S., Naseer, M.N., Benjakul, S., Zaidi, A.A. (eds) Fish Waste to Valuable Products. Sustainable Materials and Technology. Springer, Singapore. https://doi.org/10.1007/978-981-99-8593-7_1
Download citation
DOI: https://doi.org/10.1007/978-981-99-8593-7_1
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-8592-0
Online ISBN: 978-981-99-8593-7
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)