Abstract
The inevitable depletion of the nonrenewable resources has given us biofuels as an alternative fuel to be preferably used in the environment. Biofuels are considered as friendly due to neutrality of carbon dioxide and are derived from various sources through biomass conversion. Biofuels such as biodiesel, bioethanol, biogas, and biohydrogen are sustainable and renewable sources. Thus, there is a surge in the biofuel production which is being developed by biochemical processes through enzymes. This chapter summarizes about sources of biofuels, classification of biofuels into its generations, enzymes which can be used for biofuel production, biodiesel production, the pros and cons and the potential opportunities to increase enzyme production technology in a vivid way.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
14.1 Introduction
Biofuels provide a sustainable and renewable source of energy with the advancement of biotechnology. There is an adverse effect due to excessive fossil fuel consumption at an alarming rate which emits out poisonous gases such as carbon dioxide and sulfur dioxide. Thus, biofuel usage is significant as an alternative to nonconventional sources. The demand of biofuel due to high consumption has changed the market scenario. Thus, it is inevitable to increase biofuel production. It changes the compatibility of the environment by balancing with the depletion of the nonconventional sources (Ezeoha et al. 2017). Due to limited sources of fossil fuel, a crisis has developed owing to a surge in energy sector. Thus, there is a need to work on developing the new technologies taking from natural sources and developing alternate energy resources. Biofuels are formed from biomass by thermal and physical processes to generate renewable energy sources (Shaibani et al. 2012; Jahirul et al. 2012). Production of biofuel is a low-cost organic process that is environmentally friendly compared to other conventional fuels. Biofuels are available in three states (solid, liquid, and gas). Biofuel production was achieved from natural substances (Dufey 2006). Edible oil like plants, several varieties of crops, and animal fat serve as a source for first-generation biofuels (Singh et al. 2011). Nonedible substances make the source for second-generation biofuels. Recently, microbial species are known to be third-generation biofuels. The sources of biofuel are (a) bioethanol (first-generation and popular), (b) plant oil and animal fats, (c) yellow horn oil, and (d) chicken fat (Zhang et al. 2020; Dehghan et al. 2019). Bioethanol is made of maize corn, sugarcane, and beetroot through the fermentation process and produces starch as a substrate. Biodiesel fuels are also produced from plant oil and animal fats by using transesterification reactions. The sources for second-generation biofuel are discarded materials like wheat straw, sorghum stalk, and corn stover. The wasted materials’ conversion into biofuels such as bioethanol or biobutanol is done through hydrolysis of enzymes and chemical procedures (Shamsudin et al. 2021). The sources of third-generation biofuel are bacteria, viruses, algae, fungi, protozoa, and archaea. As reported from the previous work (Neto et al. 2019), enlisted algae are also one of the most favorable sources due to availability on used land and pretreated lignocellulose biomass. There are a lot of scientific challenges which are required to be facilitated to develop economically viable enzymes to increase biofuel production. Here, the chapter summarizes about the biofuel production which can be enhanced by using enzymes and different technologies from the feedstock and nonedible sources and a vivid description of biodiesel production as a biofuel through different processes. The biofuel production and consumption by different countries is shown in Fig.14.1.
Comparison of biofuel production (2019) to consumption (2030). (https://www.iea.org/data-and-statistics/charts/biofuel-production-in-2019-compared-to-consumption-in-2030-under-the-sustainable-development-scenario)
14.2 Sources of Biofuel
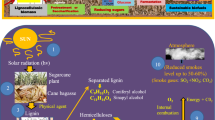
Biofuels are mainly formed from biomasses. There is a usage of different or the same feedstocks for different classes of biofuels. Sugarcane can be taken as an example for first- and second-generation biofuel feedstocks. Various biofuel feedstocks such as crops that produce oil, lignocellulose waste from solid, biomass, bacterium, yeast, algae, and fungi can be utilized as good sources (Ruan et al. 2019). The various renewable resources for biofuel production have been depicted through illustration in Fig. 14.2.
Illustration depicting various renewable resources for biofuel production (Kassim et al. 2016)
14.2.1 Oil Crops
For biofuel production crops such as soybean, sunflower, rapeseed, corn, palm kernel, palm oil, fruits of palm, coconut, copra, canola oil, castor, jatropha plant, sesame, and polenta are mainly used. The biofuel production from these oil crops is dependent on the percentage of weight of the plants. The same crop can generate different percentages of biofuels due to the variation in temperature, use of advanced technology, and enhancement of infrastructure (Ruan et al. 2019; Yusoff et al. 2021) (Table 14.1).
14.2.2 Lignocellulosic Biomass
Lignocellulose is obtained mainly from a feedstock of agriculture residues, energy crops, and forest residues. Agriculture residues include straw obtained from rice, husk from rice, straw from wheat, straw from sorghum, corn stover, and sugarcane bases. The forest residues consist of wood, chips from wood, branches from wood, and sawdust. Crops consist of switchgrass, miscanthus, cane, grasses, and lignocellulose biomass. In agriculture residues, lignocellulose containing lignin ranges from wt% 7.00 to 36.02, cellulose ranges from wt 30.42 to 49.8, and hemicellulose ranges from wt% 18.00 to 35.00. The residues collected from the forest consist of the compositions of cellulose, hemicellulose, and lignin which are 23.70–59.70 wt%, 13.00–39.00 wt%, and 18.10–34.00 wt% while energy crops are at 28.00–49.00 wt%, 15.00–32.17wt%, and 4.00–25.94 wt%, respectively (Zheng et al. 2014; Tumuluru et al. 2011) (Table 14.2).
14.2.3 Waste from Solid
The municipal waste solids obtained are paper, plastic, sludge from wastewater, waste from food, and manure from an animal source. Solid waste substances produce biofuels that are based on the chemical composition of cellulose (Table 14.3).
14.2.4 Algae
Algae are easily fast-growing simple cellular structure photosynthetic organisms with rich lipid composition. The growth of algae is observed in salts, wastewater, and marginal lands. Algae need carbon dioxide to grow and it is biodegradable without sulfur contents (Murphy et al. 2015). Algae have favorable characteristics for biofuel production. Thus, advanced methodology can be utilized to increase the growth of algae. Algae are classified into two groups: microalgae and macroalgae. The macroalgae contain proteins (5.06–20.93 wt%), carbohydrates (11.60–56.25 wt%), and lipids (6.99–15.70 wt%). Further, microalgae consist of proteins (6.00–71.00 wt%), carbohydrates (4.00–64.00 wt%), and lipids (1.90–40.00 wt%). Usually, microalgae have a higher percentage of carbohydrates and lipids compared to macroalgae (Wei et al. 2013; Jones and Mayfield 2012) (Table 14.4).
14.3 Classification of Biofuels
Biofuel’s classification is mainly based on the nature of their feedstock. Commonly known as conventional sources feedstock materials include sugar, starch, or any alternate vegetable type of oil for the production of first-generation biofuels. Beet sugar and sugarcane fermentation produce ethanol. Biogas, biodiesel, and bioalcohols are the common first-generation fuels. It is widely produced via plant oil transesterification. “Olive green” and “cellulosic ethanol” fuels are the other ways of referring to secondary biofuels. They are mainly produced from lignocellulosic biomass. The feedstock is the second-generation fuel for the production of vegetable oil produced from the waste, residue from the forest, residue from industry, and biomass. Algae help in the production of third-generation fuels. Hence, third-generation fuels are referred to as “algae fuel.” The yield of types of biofuels such as biodiesel, gasoline, butanol, and propanol is approximately ten times higher compared to the second-generation biofuel (Suganya.et al. 2016) (Fig. 14.3).
-
(a)
First-generation fuels consist of vegetable oils and biodiesel derived from agricultural plants. This generation’s biofuels have a negative influence on food security; this may be mitigated by developing feedstock sources that are nonedible which lead to an economic way to produce biofuel (Singh et al. 2020).
-
(b)
Second-generation fuels include bioethanol and biohydrogen as examples and are made from waste from agriculture and crops which are not edible sources.
-
(c)
Third-generation biofuels consist of bioethanol and biobutanol and are generated from a source of marine organisms. Examples include weeds from the sea, microorganisms, and cyanobacteria.
-
(d)
Fourth-generation biofuels include fuels from electro- and solar types from noncultivated land and microorganisms which cause photosynthesis (Singh et al. 2020) (Table 14.5).
Fig. 14.3 Generations of biofuel (Ruan et al. 2019)
Table 14.5 Replacement of petroleum-derived fuels with biofuels
A large amount of feedstock is vital to produce first-generation bioethanol causing competition within fuel and food. Second-generation biofuels used lignocellulosic biomass as the nonedible source. Sugarcane or bagasse which is the lignocellulosic material is used in the generation of bioethanol of second-generation fuels by using thermal and biological treatment. Algae due to their sustenance in a diverse environment are used for pretreatment and hydrolysis. It is observed that there is higher photosynthetic efficiency in algae. The absentia of lignin, causing a reduction in enzymatic hydrolysis, is also one of the factors in higher biofuel production by algae (Bibi et al. 2017).
14.4 Enzymes Used in Biofuel Production
The biofuel production mediated by biocatalyst enzyme improves efficiency, minimizes the environmental impacts, and increases the quality of biofuel production. Moreover, the biocatalyst uses unrefined feedstock, which includes waste oil and free fatty acids. The advantage of using biocatalyst enzymes is a) Optimum temperature b) water-based solvents. In biodiesel manufacture, lipase and phospholipase are the key constituents. Free fatty acids (FFA) and triacylglycerol are converted by lipase to fatty acid methyl esters—the key product in biodiesel. Phospholipids are converted to diacylglycerol by phospholipase thus becoming substrate for the lipase. For fermentation, cellulases are vital for cellulose digestion into glucose (https://www.biofuelsdigest.com/bdigest/2016/06/06/catalysts-and-enzymes-in-biofuel-production/). The potential efficiency of enzymes, viz., amylases, lipases, cellulases, xylanases, proteases, and monooxygenase, is further searched extensively for the production of biofuel production (Chandra and Chowdhary 2015). However, due to higher costs, several challenges are faced to use enzymes as biocatalysts in biofuel synthesis. Thus, to lower the cost and extending the life of the enzymes for utilization, immobilized form can be enabled through the solid substrate for multiple usages. In the process of utilizing immobilized enzymes, processes such as operational stability, enzyme depletion, and inhibition by reactants/products are critical to consider. In the literature survey, it is reported higher catalytic activity is observed in immobilized biocatalysts rather than free catalysts. Processes such as multiple separations, purification, and posttreatment of contaminants and wastewater will decrease if reusable immobilized biocatalysts are used which further decreases the cost and increases continuous biodiesel production (Zhao et al. 2015; Chandra and Chowdhary 2015; Chowdhary et al. 2020; Chowdhary and Raj 2020). The usage of membrane bioreactors for enzymatic processing is effective as it allows the continuous separation of products thus critical in inhibition of the enzyme. Such example is the introduction of gene-splicing enzymes to increase the production of biofuel. Genes of assorted enzymes are cloned. The utilization of recombinant DNA technology and protein engineering will minimize the cost of the enzyme (Yadav et al. 2021).
14.5 Biodiesel Production
Biodiesel can be obtained from vegetables, microemulsions, pyrolysis, and oil transesterification. Catalyzed and non-catalyzed reaction methods of transesterification of oils are used for the production of better quality biodiesel. Usually, base-catalyzed, acid-catalyzed, and enzyme-catalyzed transesterifications are done. Non-catalyzed reactions without alcohol are done at the critical condition with high temperature and pressure above its critical values (Olkiewicz et al. 2016). The transesterification process is selected due to minimum waste generation, cost-efficiency, and high productivity. Lipase-catalyzed transesterification converts feedstock into biodiesel at low temperatures. The enzyme can be reused by the immobilization technique (Fig. 14.4).
Schematic representation of biodiesel production (https://afdc.energy.gov/fuels/biodiesel_production.html)
14.5.1 Biodiesel Production from Waste Cooking Oil
Three benefits obtained were (a) cost-effectiveness, (b) eco-friendliness, and (c) waste management.
Heterogeneous catalyst is significant in biodiesel production as nano-sized calcium oxide can be used at laboratory scale and the parameters measured at optimum levels are the ratio of methanol, dose of catalyst, and temperature for the reaction Zhang et al. (2003).
-
A.
Transesterification: The reaction occurs in the presence of alcohol with catalyst (Shah et al. 2004).

The process is done as follows:
-
(a)
Placing a 300–350 ml flask on a plate heated for a certain time.
-
(b)
The plate should be well-equipped with a magnetic stirrer and a sensor controlling the temperature.
-
(c)
Cooking oil (waste) was heated to optimum temperature before the addition of catalyst and methanol.
-
(d)
The amount of methanol to oil ratio was calculated and then added to the reactor.
-
(e)
The calcium oxide catalyst was added between wt% 0.5 and 5%, and later the reaction mixture was set from 30 °C to 70 °C in 5-sec intervals. The reaction continued with continuous stirring for the desired duration.
-
B.
Factors Affecting the Transesterification of Enzyme
-
(a)
Lipid Source. Lipases are excellent as biocatalysts as triglyceride variety substrates are used during biodiesel production. Vegetable oils, viz., sunflower, rice bran, soybean, and rapeseed, has been transesterified in systems. Waste fats, tallow, and triglyceride can also be used as a few alternative sources. In previous studies, Watanabe et al. have shown a comparison of the effectiveness of transesterification and identified types such as raw soybean oil, purely refined and degummed. Candida antarctica lipase activity was observed during the production of biodiesel from degummed and refined oil. It was also suggested during transesterification reaction that refined oil has a good rate of conversion than raw oil (Watanabe et al. 2001).
-
(b)
Acyl Acceptor. Acyl acceptors are classified as straight, primary, secondary, and branched chains. Lipase acts as catalysts similar to esters which are employed in transesterification. Ethanol (CH3CH2OH) and methanol (CH3OH) are used for the industrial production of biodiesel. The performance of lower enzymes can inhibit methanol. Thus, adding alcohol, acyl acceptor, and engineered solvents are the solutions which can be done to overcome the problem. Usage of methyl acetate or ethyl acetate for the prevention of lipase inactivation is done. Ethanol can be used as an acyl acceptor with derivatives such as fatty acid ethyl esters (FAEEs), thus ethanol as an acyl acceptor can be an effective alternative for biodiesel production. Wang et al. (2009) lipase stability and operation under optimal conditions were studied which suggested optimal transesterification conditions. It was observed that oils from plant and fats of animal yield high methyl ester, viz., rapeseed oil 95%, soybean oil 91%, tea seed oil 92%, cottonseed oil 98%, and lard 95% (Wang et al. 2009).
-
(c)
Organic Solvent. Hydrophilic alcohols and triglyceride hydrophobicity mutual solubility prevent denaturation of enzymes. Transesterification of substrates by lipase with triglycerides, acceptors of acyl groups, can be used in the production of biodiesel. Examples of organic solvents are hexane, cyclohexane, heptane, petroleum ether, isooctane, acetone, and chloroform. Waste fat is converted into biodiesel and produces glycerol as a by-product. A previous study by Pollardo et al. in 2018 observed organic solvent potentiality in C. Antarctica lipase B in the production of biodiesel. They tested animal waste fats and enzyme reversibility by using organic and nonorganic solvents. Further, it was investigated that the enzyme activity was affected by organic solvents (Pollardo et al. 2018).
-
(d)
Temperature. Lipase activity may lose if enzymatic transesterification is done at low temperature. From previous studies, for biodiesel synthesis, optimal temperature ranges from 30 °C to 55 °C. The temperature variability was shown in several studies. Further, in such a case study by Iso et al. (2001), it is reported that lipase sourced from P. fluorescens was used for ethyl oleate synthesis where there was a parallel increase in temperature rate of transesterification reaction (Iso et al. 2001).
-
(e)
Water Content. Although biodiesel production is based on lipase-catalyzed reaction, water has a significant influence on the stability and catalytic activity and thus offers various roles. Enzyme undergoes a conformational change depending on the interaction of the oil-water interface. But excess addition of water to the lipases stimulates the competing hydrolysis reaction to decrease yields in transesterification. In several studies it is reported that there is addition of water content of 5% increased transesterification to 98% previous to that of 70% when water was not added. It was observed in studies of Kaieda et al. (2001) the significance of the effect of water content tested in free lipases catalyzed by P. cepacia, C. rugosa, and P. fluorescens. Without water, there was no significant change in reaction. Biodiesel has more affinity toward moisture content in contrary to petroleum diesel. In fuels the water content can be classified as free, soluble, and emulsion rated (Kaieda et al. 2001).
-
(a)
14.5.2 Biodiesel Production from Edible Oils
Biodiesel production from edible oils such as soybean is significant. Soybean mainly contains fatty acids such as palmitic, linolenic, oleic, linoleic, and stearic in equal amounts. Biodiesel from soybean oil has a high biodegradability, higher flashpoint, higher lubricity, and lower toxicity.
14.5.3 Biodiesel Production from Nonedible Sources
Nonedible sources used are jatropha, Pongamia, and castor oil. The tropical plant Jatropha curcas has been observed to be good due to the usual growth observed in low-to-high rainfall areas. Shah et al. studied on lipases such as Candida rugosa, Chromobacterium viscosum, and porcine pancreas (Shah et al. 2004). They investigated the lipases of jatropha oil in a solvent-free system to produce biodiesel by transesterification. It is reported Chromobacterium viscosum gave a promising result. Lipase immobilization on Celite 545 increases biodiesel production to 71% at 40 °C within 8 hours. Karmee and Chadha (2005) studied crude oil transesterification of Pongamia pinnata with methanol and potassium hydroxide as catalysts. For biodiesel production, the highest conversion was achieved using a 1:10 molar ratio of oil to methanol at 60 °C (Karmee and Chadha 2005).
14.5.4 Biodiesel Production from Algae
Microalga with efficient photosynthetic capability than land crops is used as a good source to produce biodiesel. Algae oil of 20%–80% is transformed into fuel types such as kerosene and biodiesel. The species used for the generation of biodiesel are Tribonema, Ulothrix, and Euglena. Algae are considered to be safer, noncompetitive, and rapidly growing organisms. Energy content at 80% (Chisti 2013) and lipid content at 30% (Lam and Lee 2012) are present in algae.
14.6 Enzymatic Hydrolysis
Plant dry matter is made of lignocellulosic matter which contains 60–90% cellulose and hemicellulose (contribute to biofuel), and greater than 5% of lignin varies with different samples of biomass (Marriott et al. 2016). In the production of biofuels, enzymatic hydrolysis plays an important role in converting lignocellulosic biomass into simpler fermentable sugars. Enzymatic hydrolysis is known as the process of breaking down a chemical by inserting a water molecule across a bond aided by an enzyme. Lignin’s presence causes toxicity to lignin derivatives and adsorption of hydrolytic enzymes within lignocelluloses ultimately obstructing biomass hydrolysis. After lignin elimination, there is an increase in biomass digestibility (Chang and Holtzapple 2000). The pretreatment methods significantly increase the efficiency of enzymatic hydrolysis (Hendriks and Zeeman 2009; Savla et al. 2020). Enzymatic hydrolysis occurs due to highly stable cellulose microfibers and coated polysaccharides which is difficult to degrade. So, the solutions of both dilute and concentrated acids are added (Agbor et al. 2011). Various enzymes such as cellulases, xylanases, hemicellulase, endoxylanases, beta-xylosydases, cellobiohydrolase, endoglucanase, etc. are used according to different substrates. Cellulases hydrolyze at glycosidic ß (1, 4) linkages and convert cellulose chains into simple sugars. So, they can be fermented by bacteria or yeast. Cellulases are extensively used enzymes. They are produced by species of bacteria and fungus that are aerophilic or anaerobic and work on an optimum range of temperature (Balat 2011). The fungi and their strains such as Trichoderma viride, Trichoderma reesei, Trichoderma longibrachiatum, and Penicillium are the commercial sources of cellulases (Bothwell et al. 1993).
The factors affecting enzyme hydrolysis are
-
1.
pH
-
2.
Temperature
-
3.
Process time
-
4.
Porosity
-
5.
Degree of crystal structure
-
6.
Particle size of lignocellulosic biomass
-
7.
Pore volume of lignocellulosic biomass and accessible surface area
14.6.1 Comparison of Efficiency of Enzymes in Biofuel Production
Lipase, xylanase, β-glucosidase, cellobiase, and cellulase are mostly researched enzymes for biofuel production processes (Singhvi and Kim 2020), of which cellulases are mostly used and they are derived from strains of bacteria (Bacillus, Cellulomonas, Thermomonospora, Caldicellulosiruptor, Erwinia, and Clostridium) or fungi (Trichoderma, Penicillium, and Aspergillus). Cellulase is a combination enzyme mainly (a) exoglucanase, (b) endoglucanase, and (c) β-glucosidase. They act together for cellulose hydrolysis (Satyamurthy et al. 2011; Binod et al. 2019). The last β-glucosidase mainly causes degradation of lignocelluloses and governs the rate of total conversion. When compared to bacteria, fungal strains produce greater amounts of cellulases. So commercially used cellulases are primarily expressed in fungi. Even in terms of the ability to digest, fungal strains are preferred. Biomass hydrolysis is limited by the lignin level in lignocellulosic materials (De Souza 2013). Strains of fungus such as soft-rot, brown-rot, and white-rot fungi are commonly employed, and their activity varies on the kind of lignocellulosic materials. These fungi can disrupt lignin because they can produce laccases, peroxides of lignin, manganese, and other enzymes of lignin degradation (Victoria et al. 2017). The fungi Trichoderma reesei and Aspergillus niger are to be taken as industrially significant microorganisms for producing a high specific class of cellulases and hemicellulases, respectively (Stricker et al. 2008a, b), but the yield of β-glucosidases collected from T. reesei is low due to its recovery process (Stricker et al. 2008a, b). A. saccharolyticus was capable of producing a greater titer of β-glucosidases in contrast to A. niger and thus increases commercial β-glucosidases production (Sørensen et al. 2012). Enzymes immobilized on carriers such as magnetic nanoparticles (MNPs) Si, Ni, and AgNPs had greater activity, thermal stability, and pH stability than free enzymes and can be reused with 60–70% efficiency (Kim et al. 2018). Compared to free enzymes, cellulases of A. niger immobilized on MNPs cyclodextrin produce larger quantities of simple sugars, with 85% of immobilized enzymes recovered for subsequent hydrolysis. From Trichoderma reesei, cellulase is staged on chitosan-associated MNPs. It is used as a preservative as it sustains 80% hydrolytic activity even after 15 cycles of hydrolysis (Huang et al. 2015). Further, during the process, the ideal circumstances of several factors such as temperature, pH, and enzyme concentration improve the efficiency of the process. The concept of a variety of strains coculturing shows efficiency for higher production of enzymes (Fusco et al. 2018). In a case study by Zhao et al., the benefits of coculturing T. reesei mixed with the culture of A. niger by genetic modification were reported. This method produced high potent cellulase with the increased hydrolysis yielding 89.35%, and it used the least amount of cellulose to produce 1g of glucose (Hu et al. 2011; Zhao et al. 2018) (Table 14.6).
14.7 Potential Opportunities to Increase Enzyme Production Technology
Enzymes are biocatalysts that are essential for all processes of life and mainly due to their ability to transform specific chemical transformations. Thus, strategies for enzyme engineering are an important concern. The applications are varied such as organic synthesis (Clouthier and Pelletier 2012), biofuel production (Himmel et al. 2007), and bioprocess engineering (Panke and Wubbolts 2002). Figure 14.5 gives a detailed view of the current scenario.
Increase of enzyme production, requires protein engineering as one of the areas where we can primarily focus. Due to the demand in the market, investments should increase in the biotechnological research sector for development. There should be infrastructure setup with a large scale-up of innovative methods of enzyme production and formulation, evaluation, development, and validation of technologies for commercialization. Development based on techniques such as microorganism culturing, recombinant DNA, and immobilization with several nanoparticles should be upgraded for the huge production of enzymes for biofuel production. Further, awareness in enzyme application can increase the demand in consumers which can eventually lead to enzyme production. In a developed nation like the USA, the government has adopted initiatives for biofuel usage as an alternative fuel. Production of biofuels such as biodiesel, cellulosic biofuels, and advanced biofuels is promoted. DuPont has a robust position in the market of enzymes for the production of ethanol. Similarly, companies such as Genencor, AB Enzymes, Shin-Nihon, ADM, Iogen, and Enmex are the leading enzyme manufacturers for biofuel production (Li et al. 2012).
14.8 Conclusion
With strong growth and prospects in the market, it is mandatory to focus on biofuel production using enzymes. For this we can look for novel biocatalysts, upgrading enzyme properties, innovations to generate various enzyme processes, genetic engineering approaches, downstream processing for the formulation of enzymes, enzyme manipulation, and immobilizations which are some of the techniques which can be followed. In this regard, the essentiality of biofuel production has been discussed with biofuel sources, its classification and the enzymes, methods are involved in biofuel production. The parameters of enzymes should be screened and altered to increase the biofuel productivity with updated knowledge on metagenomic analysis for discovery of enzymes, a cell-free system in enzymatic engineering, de novo designing of biocatalysts and bio-based technologies should be applied for higher biofuel production.
References
Agbor VB, Cicek N et al (2011) Biomass pretreatment: fundamentals toward application. Biotechnol Adv 29(6):675–685
Balat M (2011) Production of bioethanol from lignocellulosic materials via the biochemical pathway: a review. Energy Convers Manage 52(2):858–875
Bibi R, Ahmad Z, Imran M et al (2017) Algal bioethanol production technology: a trend towards sustainable development. Renew Sust Energ Rev 71:976–985
Binod P, Gnansounou E, Sindhu R et al (2019) Enzymes for second generation biofuels: recent developments and future perspectives. Bioresour Technol 5:317–325
Bothwell MK, Walke LP, Wilson DB et al (1993) Synergism between pure Thermomonospora fusca and Trichoderma reesei cellulases. Biomass Bioenerg 4(4):293–299
Chandra R, Chowdhary P (2015) Properties of bacterial laccases and their application in bioremediation of industrial wastes. Environ Sci Process Impacts 17:326–342
Chang VS, Holtzapple MT (2000) Fundamental factors affecting biomass enzymatic reactivity. In: Finkelstein M, Davison BH (eds) Twenty -first symposium on biotechnology for fuels and chemicals. Applied biochemistry and biotechnology. Humana Press, Totowa, pp 5–38. https://doi.org/10.1007/978-1-4612-1392-5_1
Chisti Y (2013) Constraints to commercialization of algal fuels. J Biotechnol 167(3):201–214
Chowdhary P, Raj A (eds) (2020) Contaminants and clean technologies. CRC Press
Chowdhary P, Raj A, Verma D, Yusuf A (2020) Microorganisms for sustainable environment and health. Elsevier
Clouthier CM, Pelletier JN (2012) Expanding the organic toolbox: a guide to integrating biocatalysis in synthesis. Chem Soc Rev 41(4):1585–1605
Dehghan L, Golmakani MT, Hosseini SMH (2019) Optimization of microwave-assisted accelerated transesterification of inedible olive oil for biodiesel production. Renew Energy 138:915–922
Demirbas A, Bafail A et al (2016) Biodiesel production from non-edible plant oils. Energy Explor Exploit 34(2):290–318
De Souza WR (2013) Microbial degradation of lignocellulosic biomass. In: Chandel A, Da Silva SS (eds) Sustainable degradation of lignocellulosic biomass-techniques, applications and commercialization. IntTech, New York, pp 207–247
Dufey A (2006) Biofuels production, trade and sustainable development: emerging issues. International Institute for Environment and Development, London, pp 1–57
Ezeoha SL, Anyanwu CN, Nwakaire JN (2017) The prospects, impacts, and research challenges of enhanced cellulosic ethanol production: a review. Niger J Technol 36(1):267–275
Firoz S (2017) A review: advantages and disadvantages of biodiesel. Int Res J Eng Technol 4(11):530–533
Frondel M, Peters J (2007) Biodiesel: a new oildorado? Energy Policy 35(3):1675–1684
Fusco FA, Fiorentino G, Pedone E et al (2018) Biochemical characterization of a novel thermostable β-glucosidase from Dictyoglomus turgidum. Int J Biol Macromol 113:783–791
Hendriks ATWM, Zeeman G (2009) Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour Technol 100(1):10–18
Himmel ME, Ding SY, Johnson DK et al (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315(5813):804–807. https://www.biofuelsdigest.com/bdigest/2016/06/06/catalysts-and-enzymes-in-biofuel-production/, https://www.grandviewresearch.com/industry-analysis/enzymes-industry
Huang PJ, Chang KL, Hsieh JF, Chen ST (2015) Catalysis of rice straw hydrolysis by the combination of immobilized cellulase from Aspergillus niger on β-cyclodextrin-Fe3O4 nanoparticles and ionic liquid. BioMed Res Int 2015:1–9
Hu HL, Van den Brink J, Gruben BS et al (2011) Improved enzyme production by co-cultivation of Aspergillus niger and Aspergillus oryzae and with other fungi. Int Biodeterior Biodegradation 65(1):248–252
IEA, Biofuel production in 2019 compared to consumption in 2030 under the Sustainable Development Scenario, IEA, Paris. https://www.iea.org/data-and-statistics/charts/biofuel-production-in-2019-compared-to-consumption-in-2030-under-the-sustainable-development-scenario
Iso M, Chen B, Eguchi M et al (2001) Production of biodiesel fuel from triglycerides and alcohol using immobilized lipase. J Mol Catal B Enzym 16(1):53–58
Jahirul MI, Rasul MG et al (2012) Biofuels production through biomass pyrolysis—a technological review. Energies 5(12):4952–5001
Jones CS, Mayfield SP (2012) Algae biofuels: versatility for the future of bioenergy. Curr Opin Chem Biol 23(3):346–351
Kaieda M, Samukawa T, Kondo A et al (2001) Effect of methanol and water contents on production of biodiesel fuel from plant oil catalyzed by various lipases in a solvent-free system. J Biosci Bioeng 91(1):12–15
Karmee SK, Chadha A (2005) Preparation of biodiesel from crude oil of Pongamia pinnata. Bioresour Technol 96(13):1425–1429
Kassim MA, Khalil Abdul HPS, Serri NA, Kassim MHM, Syakir MH, Sri Aprila NA, Dungani R (2016) Irradiation pretreatment of tropical biomass and biofiber for biofuel production. In: Waldemar AM (ed) Radiation effects in materials. Intech, pp 329–356
Kim KH, Lee OK, Lee EY (2018) Nano-immobilized biocatalysts for biodiesel production from renewable and sustainable resources. Catalysts 8(2):68
Lam MK, Lee KT (2012) Microalgae biofuels: a critical review of issues, problems and the way forward. Biotechnol Adv 30(3):673–690
Li S, Yang X, Yang S et al (2012) Technology prospecting on enzymes: application, marketing and engineering. Comput Struct Biotechnol J 2(3):e201209017
Marriott PE, Gómez LD et al (2016) Unlocking the potential of lignocellulosic biomass through plant science. New Phytol 209(4):1366–1381
Murphy JD, Drosg B, Allen E et al (2015) A Perspective on algal biomass: Energy from BiogasIEABioenergy http://task37.ieabioenergy.com/files/daten-redaktion/download/Technical%20Brochures/AD_of_Algae_ebook_end.pdf
Neto JM, Komesu A, da Silva Martins LH, Gonçalves VO, De Oliveira JAR, Rai M (2019) Third generation biofuels: an overview. In: Kuila A (ed) Sustainable biofuel and biomass: advances and impacts, 1st edn. Apple Academic Press, Boca Raton, pp 283–298
Olkiewicz M, Torres CM et al (2016) Scale-up and economic analysis of biodiesel production from municipal primary sewage sludge. Bioresour Technol 214:122–131
Oliveira FJ, Santana DD et al (2017) Generation, characterization and reuse of solid wastes from a biodiesel production plant. Waste Manag 61:87–95
Panke S, Wubbolts MG (2002) Enzyme technology and bioprocess engineering. Curr Opin Biotechnol 13(2):111–116
Pollardo AA, Lee HS et al (2018) Solvent effect on the enzymatic production of biodiesel from waste animal fat. J Clean Prod 185(4):382–388
Ruan R, Zhang Y, Chen P et al (2019) Biofuels: introduction. In: Pandey A, Larroche C, Dussap CG, Gnansounou E, Khanal SK, Ricke S (eds) Biomass biofuels biochemicals biofuels: alternative feedstocks and conversion processes for the production of liquid and gaseous biofuels, 2nd edn. Academic Press, pp 3–43
Satyamurthy P, Jain P et al (2011) Preparation and characterization of cellulose nanowhiskers from cotton fibres by controlled microbial hydrolysis. Carbohydr Polym 83(1):122–129
Savla N, Shinde A, Sonawane K, Mekuto L, Chowdhary P, Pandit S (2020) Microbial hydrogen production: fundamentals to application. Microorgan Sustain Environ Health 21:343
Shah S, Sharma S, Gupta MN (2004) Biodiesel preparation by lipase-catalyzed transesterification of Jatropha oil. Energy Fuel 18(1):154–159
Shaibani N, Yaghmaei S et al (2012) Ethanol production from sugarcane bagasse by means of on-site produced and commercial enzymes; a comparative study. Period Polytech Chem Eng 56(2):91–96
Shamsudin MI, Tan LS, Tsuji T (2021) Enzyme immobilization technology in biofuel production: a review. IOP Conf Ser Mater Sci Eng 1051(1):012056
Singh A, Olsen SI, Nigam PS (2011) A viable technology to generate third-generation biofuel. J Chem Technol Biotechnol 86(11):1349–1353
Singh V, Singh N, Tabassum N, Mishra V (2020) Microbial system: an emerging application in the bioenergy production. In: Srivastava N, Srivastava M, Mishra PK, Gupta VK (eds) Microbial strategies for techno-economic biofuel production. Springer, Singapore, pp 249–264
Singhvi M, Kim BS (2020) Current developments in lignocellulosic biomass conversion into biofuels using nanobiotechnology approach. Energies 13(20):5300
Sørensen A, Ahring BK et al (2012) Identifying and characterizing the most significant β-glucosidase of the novel species Aspergillus saccharolyticus. Can J Microbiol 58(9):1035–1046
Stricker AR, Mach RL, De Graaff LH (2008a) Regulation of transcription of cellulases-and hemicellulases-encoding genes in Aspergillus niger and Hypocrea jecorina (Trichoderma reesei). Appl Microbiol Biotechnol 78(2):211–220
Stricker SH, Steenpass L et al (2008b) Silencing and transcriptional properties of the imprinted Airn ncRNA are independent of the endogenous promoter. EMBO J27(23):3116–3128
Suganya T, Varman M et al (2016) Macroalgae and microalgae as a potential source for commercial applications along with biofuels production: a biorefinery approach. Renew Sust Energ Rev 55(C):909–941
Tumuluru JS, Wright CT, Hess JR, Kenney KL (2011) A review of biomass densification systems to develop uniform feedstock commodities for bioenergy application. Biofuels Bioprod Biorefin 5(6):683–707
Vassilev SV, Baxter D et al (2010) An overview of the chemical composition of biomass. Fuel 89(5):913–933
Victoria J, Odaneth A, Lali A (2017) Importance of cellulase cocktails favoring hydrolysis of cellulose. Prep Biochem Biotechnol 47(6):547–553
Wang SL, Lin YT et al (2009) Purification and characterization of extracellular lipases from Pseudomonas monteilii TKU009 by the use of soybeans as the substrate. J Ind Microbiol Biotechnol 36(1):65–73
Watanabe Y, Shimada Y et al (2001) Enzymatic conversion of waste edible oil to biodiesel fuel in a fixed-bed bioreactor. J Am Oil Chem Soc 78:703e7
Wei N, Quarterman J, Jin YS (2013) Marine macroalgae: an untapped resource for producing fuels and chemicals. Trends Biotechnol 31(2):70–77
Yadav AK, Surabhi P, Tripathi AD, Paul V (2021) Role of enzymes in biofuel production. In: Srivastava N (ed) Bioenergy research: evaluating strategies for commercialization and sustainability. Wiley, pp 1–18
Yusoff MHM, Nazir MH, Zahid I et al (2021) A comprehensive review on oil extraction and biodiesel production technologies. Sustain For 13(2):788. https://doi.org/10.3390/su13020788
Zhang H, Liu T, Zhu Y et al (2020) Lipases immobilized on the modified polyporous magnetic cellulose support as an efficient and recyclable catalyst for biodiesel production from Yellow horn seed oil. Renew Energy 145:1246–1254
Zhang Y, Dube MA et al (2003) Biodiesel production from waste cooking oil: 1 Process design and technological assessment. Bioresour Technol 89(1):1–16
Zhao C, Deng L, Fang H (2018) Mixed culture of recombinant Trichoderma reesei and Aspergillus niger for cellulase production to increase the cellulose degrading capability. Biomass Bioenergy 112:93–98
Zhao X, Qi F, Yuan C et al (2015) Lipase-catalyzed process for biodiesel production: Enzyme immobilization, process simulation and optimization. Renew Sust Energ Rev 44:182–197
Zheng Y, Zhao J et al (2014) Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog Energy Combust Sci 42:35–53
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Vipparla, C. et al. (2022). Enzyme Technology in Biofuel Production. In: Chowdhary, P., Pandit, S., Khanna, N. (eds) Bio-Clean Energy Technologies Volume 2. Clean Energy Production Technologies. Springer, Singapore. https://doi.org/10.1007/978-981-16-8094-6_14
Download citation
DOI: https://doi.org/10.1007/978-981-16-8094-6_14
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-8093-9
Online ISBN: 978-981-16-8094-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)