Abstract
The vegetation of tropical alpine environments is characterized by giant rosette plants, of which, two species, Senecio keniodendron and Lobelia telekii, were found to have grown on deglaciated terrain below Tyndall Glacier on Mount Kenya. The species are one of the most attractive elements of the landscape, which captures the attention of visiting eco-tourists. Therefore, the plants are of conservation interest, particularly during a period of climate change. In this study, I analyzed the growth dynamics and distribution of the two species. Field surveys were conducted in 2016 and 2018. The rates of mortality and recruitment were found to be closely similar, and therefore, the population numbers were stable. The heights of the surviving plants were found to have increased over the two years. When the heights of Lobelia telekii rosette leaves were >20 cm in 2016, the elongation rates of anthotactic spiral inflorescences tended to increase. The amount of solar radiation affected the distribution of the giant rosette plants, i.e., plants were distributed in areas where total solar radiation levels were high.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords

Vegetation on the upper slopes of Mount Kenya is characterized by Senekio keniodendron and Lobelia telekii. Image was acquired at the head of Teleki Valley, close to Tyndall Glacier
1 Introduction
Giant rosette plants occur on the slopes of the East African mountains. Giant forms of Senecio and Lobelia are abundant and widespread taxa that are endemic to the alpine zone of Mount Kenya (Coe 1967). The vegetation on the volcanic land is characterized by giant rosette species, including Senekio keniodendron (Dendrosenecio keniodendron; SK; Fig. 1) and Lobelia telekii (LT; Fig. 2). These species and other giant rosette plants are typical of high-elevation tropical landscapes with large diurnal temperature fluctuations (Hedberg 1964; Coe 1967; Rehder et al. 1988). These unusual plants are an important natural resource of Mount Kenya, which has been designated a World Heritage site and has received the status of national park in Kenya. Eco-tourism is a major industry in Kenya. As these species attract the interests of tourists, an expanded knowledge of the distribution dynamics and habitats of these unusual plants is expected to contribute to the development of this industry. The data will also improve our understanding of the changes in vegetation associated with recent climate changes. The giant rosette species are relatively long-lived, a trait that enables investigation of the effects of long-term changes in local environments (Young 1994).
In this study, I investigated an early vegetation succession on Mount Kenya in areas that were recently deglaciated. I examined the distribution of SK and LT on this terrain. Vegetation distribution at the foot of a glacier in tropical alpine zones is determined by the timing of deglaciation and a range of other environmental factors such as soil development (Matthews 1992). Mizuno (2005a) showed that giant rosette plants, which are distributed around the glacier, are affected by geomorphological conditions and gravel size. Young (1991, 1994) and Young and Peacock (1992) conducted biological surveys of giant rosette plants. Smith and Young (1982, 1987, 1994) examined the population dynamics of these plants. The studies found that LT occurs mainly on dry rocky slopes at elevations of 3500–5000 m (Young 1994). Smith and Young (1994) showed that changes in the population of SK are related to slope aspect and elevation. However, micro-scale analyses of the relationship between giant rosette plant distribution, vegetation succession, and habitat conditions are yet to be explored. The dynamics of seedlings and the growth period in high-elevation zone are especially unclear. I investigated the distribution dynamics of giant rosette plants through field studies in 2016 and 2018.
2 Research Area
Mount Kenya is an isolated, extinct volcano located on the Equator (0°6′S, 37°18′E). The summit (Batian) is at an elevation of 5199 m. Rocks on the volcanic massif consist of basalt, phonolite, kenytes, agglomerates, trachyte, and syenite (Bhatt 1991). This study was conducted below the snout of Tyndall Glacier (Fig. 3), which is the second largest glacier on the volcano. The snout is at an elevation of 4600 m. The glacier has continually retreated for at least 100 years (Hastenrath 2008).
The Tyndall Glacier retreated at a rate of 0–3 m/year during 1958–1997, the rate of retreat was increased to 7–15 m/year between 1997 and 2011 (Mizuno and Fujita 2014). The recent trends of deglaciation are strongly related to global warming (Mizuno and Fujita 2014). Mizuno and Fujita (2014) also showed that pioneer plant species have advanced with the retreat of the glacier. The movements of some species did not appear to be spatially related to glacial retreat; however, they might be related to an increase in air temperature, soil development, seed dispersal limitation, and the interval of masting (Mizuno and Fujita 2014). Tyndall Glacier has developed glacial moraines during different developmental periods since the last glacial period (Mahaney 1990). For example, Lewis moraine was formed 100 years ago (Fig. 4). Although the abundance of plants was very low on this youngest moraine, several species were able to get established in this location (Mizuno 2005b). The characteristics of vegetation on the deglaciated moraines in this area have been reported by Mizuno and Fujita (2014).
Contour map of the terrain below the snout of Tyndall Glacier, Mount Kenya. The margins of Tyndall Glacier and geomorphological features are described in Mizuno and Fujita (2014). (Contour map generated by the AW3D DEM model)
Data from the Centre for Training and Integrated Research in ASAL Development (CETRAD) indicate that the average rainfall from 1978 to 2019 was 1493.5 mm/year at Naro Moru meteorological station (Altitude: 3050 m). The maximum rainfall recorded was 2030.3 mm in 1981, and the minimum rainfall recorded was 939.2 mm in 2000. During 2016, 2017 and 2018, when the study was conducted, the average annual precipitation was 1194.2 mm, 1304.0 mm, and 1914.4 mm, respectively. Therefore, we can suggest that precipitation near the study site was lesser than the average in 2016 and was higher than the average in 2018.
3 Research Methods
The field survey was conducted in August 2016 and August 2018. The research area was located inside a ridge of Lewis moraine on a trim line (Fig. 4). The positions of all individual types of giant rosette species of SK and LT that are taller than 3 cm were mapped in 2016. In this study, the growth and death of all existing individuals of the two species in 2018 were analyzed, and the appearance of new species was recorded. The distribution maps of SK and LT were plotted using these data. A contour map was generated with a 5 m-mesh using the ALOS World 3D topographic data digital surface model (AW3D; NTT DATA, RESTEC included JAXA).
I analyzed the relationship between the distribution of giant rosette plants and elevation, slope angle, and slope direction using AW3D data. Solar radiation (W m–2 day−1) was calculated using AW3D and the r.sun package in GRASS GIS. The procedure was influenced by the shadow of the summit, and not by the amount of cloud cover. Therefore, the calculated values were ideal for this study.
4 Results
4.1 Distributions of Giant Rosette Plants
Table 1 lists the changes in the number of SK and LT plants in 2016 and 2018. The number of SK and LT plants in 2018 were 171 and 217, respectively. During the period 2016–2018, 14 individuals of SK species died and 15 were recruited; and 31 individuals of LT died and 34 were recruited. Hence, the population densities were stable over this two-year period.
The average height of SK was 21.0 cm (maximum: 112 cm). The average height of LT was 17.7 cm (maximum: 199 cm). Figures 5 and 6 show the distributions of SK and LT, respectively. Both species were distributed around Tyndall Tarn. The distribution area was approximately 400 m distant from the glacier. The two species were mostly distributed on the north-western slope and on the southern side of the tarn. They were less frequent in the northern sector of the tarn, which was near to the glacier, and on the eastern side of the tarn. Dead individuals and new recruits were sparsely distributed.
4.2 Growth of Senekio Keniodendron and Lobelia Telekii
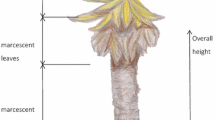
The heights of SK and LT changed over two years. The average increase in the height of SK and LT was 3.7 cm and 8.7 cm, respectively; the corresponding maximum height was 22 cm and 124 cm. SK and LT possessed similar growth patterns; however, their growth processes differed. The growth of LT was caused by the elongation of anthotaxis (Fig. 7).
Figure 8 is a scatter plot showing the differences in the height of LT between 2016 and 2018 (newly recruited plants and dead individuals are not included in the plot). It can be observed that the size of almost all individuals was increased, but the height of certain species was reduced in 2018. Height losses could be attributed to the foraging animals, such as tree hyraxes. When the heights of rosette leaves were >20 cm in 2016, the elongation rate of anthotaxis tended to increase (Fig. 8); when the heights were <20 cm in 2016, anthotaxis did not develop.
4.3 Environmental Factors
The distribution pattern of both species was related to elevation. Figure 9 shows the number of live, dead, and newly recruited individuals in relation to changes in elevation. Generally, the number tended to increase at lower elevations and decrease at higher elevations. The distribution was patchy, even at lower elevations. This patchiness was related to local topography. However, the relationship between elevation and the number of dead and newly recruited individuals was not clear.
Both species generally occurred on flat terrain with a slope of 10°; however, certain species were also found on the relatively steep slopes of approximately 30°. Most individuals occurred on slopes with a southeast aspect.
Figure 10 shows the density plot of number of individuals in relation to solar radiation. The two curves in the figure are (i) the probability density function of SK and LT, and (ii) the probability density function of random points generated by GIS. SK and LT tended to occur in areas with elevated solar radiation. Kolmogorov–Smirnov tests showed that the distributions of both species were significantly different (p < 0.01), indicating that their abundance was related to the amount of solar radiation. Although most plants occurred in strongly illuminated sites, a few individuals did not; hence, solar radiation might not be the only factor influencing the distribution pattern. Seed dispersal ranges and other factors such as differences in distributed gravel size might have been also influential.
Density plot showing the number of Senekio keniodendron (1) and Lobelia telekii (2) in relation to solar radiation levels. The curves represent plots of (i) the probability density function of Senekio keniodendron and Lobelia telekii, and (ii) the probability density function of 2000 random points generated by GIS software
Figure 11 is a box plot showing the relationship between the status of each individual (dead/alive/newly recruited) and solar radiation. Dead individuals tended to occur in areas with reduced solar radiation. On the other hand, the new recruits occurred primarily in areas with strong solar radiation. Hence, solar radiation was closely related to the status of each individual. Dead individuals were found mostly at high elevations, where the slope was steep and the amount of solar radiation was limited. The new recruits occurred mostly at low elevation, where radiation was high and slope was gentle. A multi-year investigation of seedling dynamics showed that giant rosette species present in the research site have expanded their distribution in areas with favorable environmental conditions.
5 Conclusion
I studied the growth dynamics and distributions of SK and LT over two years using data collected during field surveys conducted in August 2016 and August 2018. The number of SK and LT did not change markedly over the two years. Growth processes differed between the species. The floral inflorescences of LT increased in length when the height of the rosette leaves was >20 cm. The species also had similar habitat preferences; i.e., they tended to occur at low elevations on terrains that were exposed for 60 years after deglaciation.
The preferential occurrence of individuals at low elevation was related to the amount of solar radiation. Similarly, the status of plant individuals (dead, alive, newly recruited) was related to the amount of solar radiation. Young (1984) indicated that increased solar irradiation increases floral development rates in LT; however, this research focused only on slope aspect. The present work demonstrated a quantitative relationship between plant status and solar radiation and showed that the species’ distributions were affected by factors other than soil moisture (Young 1994) and gravel size (Mizuno 2005b).
Nevertheless, apart from light, plant status and distribution were found to be related to the following environmental factors: soil moisture, ground temperature, and snow melt. The influence of these additional factors warrants further study.
References
Bhatt N (1991) The geology of Mount Kenya. In: Allen I (ed) Guide to Mount Kenya and Kilimanjaro. The Mountain Club of Kenya, Nairobi
Coe MJ (1967) The alpine climate. In: The ecology of the alpine zone of Mount Kenya. Monographiae Biologicae, vol 17. Springer, Dordrecht
Hastenrath S (2008) Recession of equatorial glaciers: a photo documentation. Sundog Publishing, Madison, US
Hedberg O (1964) Features of afroalpine plant ecology. Acta Phytogeogr Suecica 49:1–144
Mahaney WC (1990) Ice on the equator: quaternary geology of Mount Kenya. Wm Caxton Press, Sister Bay, Wisconsin
Matthews JA (1992) The ecology of recently deglaciated terrain: a geoecological approach to glacier forelands and primary succession. Cambridge University Press, Cambridge
Mizuno K (2005a) Vegetation succession in relation to glacial fluctuation in the high mountains of Africa. Afr Study Monogr Suppl Issue 30:195–212
Mizuno K (2005b) Glacial fluctuation and vegetation succession on Tyndall Glacier Mt. Kenya. Mt Res Dev 25:68–75
Mizuno K, Fujita T (2014) Vegetation succession on Mt. Kenya in relation to glacial fluctuation and global warming. J Veg Sci 25:559–570
Rehder H, Beck E, Kokwaro JO (1988) The afroalpine plant communities of Mt. Kenya (Kenya). Phytocoenologia 16:433–463
Smith AP, Young TP (1982) The cost of reproduction in Senecio keniodendron, a giant rosette species of Mt. Kenya. Oecologia 55:243–247
Smith AP, Young TP (1987) Tropical alpine plant ecology. Annu Rev Ecol Evol Syst 18:137–158
Smith AP, Young TP (1994) Population biology of Senecio keniodendron (Asteraceae)—An afroalpine giant rosette plant. In: Rundel PW, Smith AP, Meinzer FC (eds) Tropical alpine environments: plant form and function. Cambridge University Press, Cambridge
Young TP (1984) Solar irradiation increases floral development rates in afro-alpine Lobelia telekii. Biotropica 16:243–245
Young TP (1991) The ecology, flora and fauna of Mount Kenya and Kilimanjaro. In: Allan I (ed) Guide to Mount Kenya and Kilimanjaro. The Mountain Club of Kenya, Nairobi
Young TP (1994) Population biology of Mount Kenya lobelias. In: Rundel PW, Smith AP, Meinzer FC (eds) Tropical alpine environments: plant form and function. Cambridge University Press, Cambridge
Young TP, Peacock MM (1992) Giant senecios and alpine vegetation of Mount Kenya. J Ecol 80:141–148
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Teshirogi, K. (2022). Geoecology II: Growth Dynamics and Distribution of Giant Rosette Plants on Recently Deglaciated Terrain Below the Tyndall Glacier on Mount Kenya. In: Mizuno, K., Otani, Y. (eds) Glaciers, Nature, Water, and Local Community in Mount Kenya. International Perspectives in Geography, vol 17. Springer, Singapore. https://doi.org/10.1007/978-981-16-7853-0_7
Download citation
DOI: https://doi.org/10.1007/978-981-16-7853-0_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-7852-3
Online ISBN: 978-981-16-7853-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)