Abstract
Arctium lappa (family, Asteraceae), commonly called Burdock, owing to its diverse volatile and nonvolatile metabolites is known for a variety of therapeutic and pharmacological effects. These secondary metabolites consist of phytosterols, terpenes/terpenoids, hydrocarbons, flavonoids, fatty acids, carboxylic derivatives, lignans, fatty acids, acetylenic compounds, polysaccharides, aldehydes, methoxypyrazines, carboxylic and fatty acids, monoterpenes, and sesquiterpenes. Burdock has also shown multifaceted pharmacological actions that include antidiabetic, antioxidant, hepatoprotective, anticancer, gastroprotective, antibacterial, antiallergic, antimicrobial, antiviral, and anti-inflammatory. This chapter aims to provide a comprehensive overview of the chemistry and biological activities of the secondary metabolites found in A. lappa and its species.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
10.1 Introduction
Arctium lappa, commonly known as greater “burdock,” “gobo,” “edible burdock,” or “beggar’s button,” is an Eurasian species belonging to family Asteraceae. The plant has been originally cultivated in Asia and Europe, but now it is being cultivated in different climates and countries as well. It has become an invasive weed of high-nitrogen-content soils that are mostly found in countries and regions like North America, Australia, and others regions. The plant is regarded as a nutritive and healthy food in Chinese societies. It has been used for its therapeutic value in countries of Europe, North America, and Asia for hundreds of years (Tabassum et al. 2018). The genus name has been derived from a Greek word “arcteion” which means “bear,” alluding to the plant habitus which is characterized by marked hairiness. Among various species, A. lappa is most common and widespread, besides other species as A. minus and A. tomentosum. This shrub grows nearly up to 1 m in height and its young roots usually develop branches that can reach nearly 45–50 and 3–6 cm in depth and diameter, respectively. The shape of the roots is cylindrical, with slightly thin brown skin. The interior of the plant varies from white to yellowish-white that usually depends on the age of the plant (Barceloux 2008). The period from planting to reap varies from 8 to 12 months with a per hectare yield of 8–40 tons. Burdock roots, leaves, and seeds are used as therapeutic elements in traditional medicine, predominantly in the form of tea. Because of presence of a significant content of chlorogenic acid, the parts of plant have bitter and astringent taste (Chan et al. 2010; Burgmans et al. 1992).
In Chinese traditional medicine system, A. lappa is commonly known as “Niu Bang Zi” and is believed to be a healthy and nutritious food in Chinese societies. In folk medicine, seeds of A. lappa are crushed to form a combination that provides relief against common cold, tonsillitis, throat pain, measles, and arthritis. Burdock root is also used to treat ulcers, eczema, rheumatism, gout, psoriasis, and acne. In Chinese traditional system, dried burdock is used as a diaphoretic, diuretic, and blood-purifying agent. It is believed to purify blood by removing dangerous toxins. The extract from different parts of A. lappa has been considered beneficial for health, as it helps to improve the body’s defense system and improves metabolic activities (Liu et al. 2012).
A. lappa and its species are characterized by hemicryptophyte plants that have erect taproot system and stout stems. The leaves are held sporadically as dentate, tomentose, alternate and cordate. The stem is usually strong, upright, grooved, usually branched, and reddish in color. Inflorescences (a cluster of flowers) is formed by corymbose or solitary conical-ovoid to orbicular capitula armed with involucres that are made up of bracts ending with curved apices. Receptacles are made up of many hard scales. Florets are hermaphrodite, white or purple in color. Pollination is mainly carried out by insects, generally belonging to Lepidoptera (The Scientific Foundation for Herbal Medicinal Products 2003). Figure 10.1 shows the photographic images of some species of Arctium at flowering stage, and Fig. 10.2 shows the photographic image of Arctium root.
A. lappa species (Wang et al. 2019)
Burdock root (with copy right permission license number: 499428779638) (Chan et al. 2010)
10.2 Phytochemistry of A. lappa
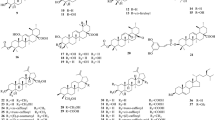
A. lappa has shown diverse pharmacological effects owing to the presence of diverse volatile and nonvolatile secondary metabolites like fatty acids, terpenes, flavonoids, lignans, acetylenic compounds, hydrocarbons, polysaccharides, phytosterols, terpenoids, aldehydes, carboxylic acids, fatty acids, monoterpenes, and sesquiterpenes (Swamy 2019). So far, over 200 nonvolatile compounds have been isolated and identified from this genus. With the advancement of technology different modern analytical techniques like high-performance liquid chromatograph (HPLC), thin layer chromatography (TLC), nuclear magnetic resonance (NMR), mass spectrometry (MS), infrared (IR) spectrometry, etc., more active ingredients of this plant have been isolated over the last 10 years (Park et al. 2007). The details of chemical constituents, occurrence in different plant parts, viz. seeds, leaves, fruits, or roots, and the modern qualitative analytical techniques used for their determinations are briefly summarized in Table 10.1, whereas their detailed description is given in below section. The chemical structures of some of the nonvolatile compounds from Arctium and its species are shown in Fig. 10.3.
10.2.1 Lignans
Main bioactive lignans that are found in A. lappa include arctigenin (a dietary phytoestrogen) and its glycoside arctiin, which are mostly present in seeds, fruits, roots, and leaves (An et al. 2003; Ming et al. 2004; Liu et al. 2012). Apart from lignans, these plant parts are also rich in low levels of sesquilignans and dilignans. Lappaol A and B were the first sesquilignans isolated and characterized from the seeds of A. lappa (Ichihara et al. 1976). In the subsequent years more sesquilignans, namely Lappaol C, D, and E, and two dilignans, namely Lappaol F and H were structurally determined from the seeds of A. lappa. Boldizsár and colleagues in 2010 used simple high performance liquid chromatography analytical technique to identify the presence of active constituent arctiin from the fruits of plant (Boldizsár et al. 2010). Using bioactivity-guided isolation and fractionation, Lappaol A, Lappaol C, Lappaol F, arctiin, and arctigenin E were isolated and later characterized from the ethanolic extract of A. lappa seeds (Ming et al. 2004). High-performance liquid chromatography (HPLC), mass spectrometry (MS), liquid chromatography (LC), and ultra-performance liquid chromatography (UPLC) quantitative analytical tools have been used to isolate and characterize arctigenin and arctiin in the roots, leaves, and seeds (Ferracane et al. 2010). A high-speed counter-current chromatography (HSCCC) was used to find the pure compound arctiin from the fruit extract of the plant. More than 49% of arctiin has been obtained by modern analytical techniques based on NMR and LC-MS (Wang et al. 2005).
10.2.2 Fatty Acids and Esters
Miyazawa and colleagues found 11 compounds in the methanolic extract of A. lappa. Among these, 10 belonged to fatty acid (Arctium et al. 2005). The compounds were identified as stearic acid, methyl stearate, methyl palmitate, palmitic acid, oleic acid, methyl linolenate, methyl oleate, linoleic acid, methyl linoleate, and linolenic acid. Iyazawa and colleagues in 2005 (Iyazawa et al. 2005) reported methyl palmitate, methyl linoleate, sitosterol-β-d-glucopyranoside, and methyl linolenate that showed an inhibitory effect against α-glucosidase. Later, Kuo et al. (2012) isolated and characterized methyl oleate, linolenic acid, and methyl-α-linolenate as the chief constituents from the n-hexane fraction of roots of the plant. The presence of palmitic acid, stearic acid, linoleic acid, and oleic acid has also been reported from the fruits of the plant (Boldizsár et al. 2010).
10.2.3 Acetylenic Compounds
Washino and colleagues in 1986 (Washino et al. 1986), identified and characterized 9 sulfur-containing acetylenic compounds, namely, arctinone-a & b, arctinol-a & b, arctinal, arctic acid-b & c, arctinone-a acetate, and methyl arctate-b from the plant. On spectral and chemical analysis, these compounds were found to be the products of 5′-(1-propynyl)-2′,2-bimethyl-5-yl. Later, presence of few guaianolides linked with sulfur-containing acetylenic compounds, viz. lappaphen-a & b, lactone, dehydrocostus, and dehydrodihydrocostus lactone, were isolated and characterized from the acetone extracts of the plant root (Washino et al. 1986). The plant possesses several bioactive constituents having acetylenic linkages that have demonstrated antibacterial, antifungal, and anti-edematogenic activities (Maria et al. 2016).
10.2.4 Phytosterols
A study carried out by Ahangarpour and colleagues on A. lappa, reported a natural phytosterol, daucosterol from its seeds (Ahangarpour et al. 2017). Other species of the plant, viz. A. tomentosum was found to contain two steroids (β-sitosterol and daucosterol). Ming et al. (2004), using bioactivity guided fractionation technique isolated β-sitosterol and daucosterol from ethanolic seed extracts of A. lappa. Later in 2005, sitosterol-β-d-glucopyranoside was also isolated from the ethanolic extract of the plant (Miyazawa et al. 2005).
10.2.5 Polysaccharides
Ferracane et al. (2010) for the first time reported the presence of pectic polysaccharides in edible roots of A. lappa. After that Watanabe in 1993, and more recently Carlotto and colleagues in 2016, isolated several polysaccharides like pectic substances; rhamnogalacturonan with neutral sugars; hemicellulose (xyloglucan, xylan, galactan, arabinan, and arabinogalactan); cellulose, arabinose, and galactose from cell walls and roots of A. lappa; and leaves and roots of A. minus (Kato and Watanabe 1993). Biologically active inulin type fructofuranans and other fructo-oligosaccharides were isolated from the roots of A. lappa but in small quantity (Kardošová et al. 2003). It has been observed that these water-soluble polysaccharides obtained from the plant significantly increase the dysregulation of pro-inflammatory cytokines TNF-α, IL-6 and IL1β, and anti-inflammatory cytokines IL-10 (Wang et al. 2019).
10.2.6 Derivatives of Caffeoylquinic Acid (Carboxylic Acids)
They are the main bioactive phenolic constituents of Arctium species and the high antioxidant potential is thought to be due to these compounds. The roots of A. lappa have been found to contain derivatives of caffeoylquinic acid, viz. 1-0-,5-O-dicaffeoylquinic acid (Yang et al. 2012). Both chlorogenic acid and caffeic acid are present in the skin of roots of the plant; however, the quantity of former is more (Chen et al. 2004). HPTLC technique has been used as qualitative chemical profiling tool to estimate chlorogenic acid in roots. It has been reported that caffeoylquinic acid and its derivatives exhibit diverse biological activities like reduction in diet-induced obesity through modulation of peroxisome proliferator-activated receptor alpha (PPARα) and liver X receptors alpha (LXRα) transcription (Huang et al. 2015) and anti-ulcerogenic activity (Lee et al. 2010).
10.2.7 Flavonoids
Flavanols and flavones are the two main flavonoids reported from A. lappa. Quercetin-3-O-rhamnoside has been reported from the leaves of the plant. Later in the year 1971 Saleh and colleagues reported more phenolic compounds such as luteolin, quercetin, quercetin and rutin from the roots, leaves, fruits, and seeds of A. lappa in their work (Saleh and Bohm 1971).
10.3 Volatile Compounds
Until most recently, almost 100 volatile compounds have been reported from A. lappa. Details about these compounds (name, species, part, and the analytical techniques employed for isolation and identification) are described in Table 10.2. Some of the chemical structures of volatile compounds have also been given Fig. 10.4.
10.3.1 Hydrocarbons
Washino and colleagues in their studies on the plant isolated 14 hydrocarbon compounds from the seeds, leaves, and roots of the plant. These include tetradecane, tetracosane, pentadecane, pentacosane, 1-pentadecene, 2-nepthalenemethanol, nonadecane, hexacosane, heptacosane, 1-heptadecene, eicosane, dihydroaplotaxene, cloven, and aplotaxene (Washino et al. 1986).
10.3.2 Aldehydes
Work performed by Wang as well as Washino and colleagues in the year 1986 and 2004, respectively, reported 19 aldehydes namely 4-methoxybenzaldehyde, tridecanal, propanal, pentanal, phenylacetaldehyde, (E)-2-octanal, nonanal, 3-methylpropanal, (E)-2-hexenal, (Z)-3-hexenal, hexanal, heptanal, dodecanal, decanal, butanal, benzaldehyde, octanal, and undecanal from the roots of the plant (Washino et al. 1986; Wang et al. 2005). In literature, there is only one reported aldehyde, alkyl aldehyde nonanal that has been found in all parts of plant, viz. leaves, roots, and seeds (Tables 10.3 and 10.4).
10.4 Pharmcological Profile of A. lappa
In traditional system of medicine A. lappa has been extensively used as an ethnomedicinal plant mostly in Europe, Asia, and North America and has been commonly used to treat numerous illnesses like rheumatoid arthritis, gout, Type 1 and 2 diabetes, and dermatological complications (Azizov et al. 2012). The plant has been used for the treatment of various diseases ranging from acute and chronic inflammation, arthritis, and various skin-related problems, namely, rough skin conditions such as eczema and psoriasis to cancer treatments as well (Kolacz et al. 2014b). Its roots have been employed as an antidote to mercury poisoning (Maghsoumi-Norouzabad et al. 2016). A. lappa has also been used to treat alopecia (loss of hair) among adults (Kolacz et al. 2014a, b). It has shown wide range of pharmacological activities like, anticancer, antidiabetic, antioxidative, anti-inflammatory, antimicrobial, hepatoprotective, gastroprotective, antifertility, antiallergic, and anti ulcerative colitis, etc. Table 10.5 shows the individual compounds possessing biological activity with possible mechanism of actions.
10.4.1 Anticancer Potential
Cancer therapy is very difficult because it is a complex and curatively challenging disease owing to its intra- and inter-tumor heterogeneity, which makes it difficult to target. Since anticancer therapy resistance is increasing day by day, research is being carried out to overcome this resistance. An important approach in this regard is the interdisciplinary approach, wherein research is being carried out to isolate and characterize new bioactive molecules from natural products having significant medical outcome and minimum off-target effects. Bioactive molecules that have been reported from the plant have significant anticancer activities in different cancer cell lines and cancer models.
Arctigenin, a natural lignan, that has been isolated from the seeds of A. lappa possesses antitumor activity. Its effect is shown by modulating the tumor cells that are susceptible to the effects of the nutrient-poor environment (Awale et al. 2006). In lung adenocarcinoma, arctigenin is found to increases the proportion of cells in the cell cycle (G0/G1) phase in A549 cell line (Susanti et al. 2013). It also decreases levels of proteins that are involved in GI/S phase checkpoint signaling, including cyclin-dependent kinases CDK2, CDK7, cyclin E & H, nuclear protein mapped to the AT locus (NPAT), and protein cyclin-dependent kinases (p-CDK) (Susanti et al. 2013). In Canadian population, A. lappa has been shown to improve health-related quality of life (HRQOL) and check cancer development, and is one the active ingredients present in herbal products, viz. “Flor-Essence” and “Essiac” recommended for prolonging of survival and enhancement of health-related quality of life (HRQOL) among cancer patients (Tamayo et al. 2000).
10.4.2 Inhibition of JAK-STAT Signaling
In a study conducted on mice-bearing gain- or loss-of-function gene mutations which encode Janus kinases signal transducer and activator of transcription proteins (JAK-STAT) signaling pathway, it was observed that this pathway emerged as a central means of communication node for the immune system. Work performed by Yao et al. (2011) revealed that arctigenin from A. lappa inhibited IL-6 and exerted inhibitory effects on STAT3 tyrosine phosphorylation through suppression of JAK1 & 2 and Schmidt-Ruppin A-2 (Src), a proto-oncogene tyrosine-protein kinase.
10.4.3 Antidiabetic Activity
In traditional system of medicine, roots of A. lappa have been used as first choice treatment for diabetes. A study conducted by Ahangarpour and colleagues reported that ethanolic extract of burdock roots administered orally to streptozocin-induced diabetic rats lowered levels of glucose and increased levels of insulin in blood significantly (Ahangarpour et al. 2017). A. lappa markedly decreased very low-density lipoproteins (VLDL), serum total cholesterol (TC), and triglycerides (TG) in diabetic mice (Ahangarpour et al. 2017).
10.4.4 Antimicrobial Activity
Roots and leaves of A. lappa are eaten in salad in folk medicine. In vitro studies have shown potential prebiotic effect (Moro et al. 2018). Lyophilized leaf extract of the plant exhibited antimicrobial activity, especially against bacteria that are related to endodontic pathogens such as pseudomonas aeruginosa, lactobacillus acidophilus, candida albicans, and bacillus subtilis (Pereira et al. 2005). Chlorogenic acid obtained from its root extract has shown antibacterial activity against Klebsiella pneumoniae and has also been found to possess anti-β-lactamase activity (Rajasekharan et al. 2017). Besides it also inhibits the formation of biofilm by Escherichia coli and candida (Chan et al. 2011).
10.4.5 Ulcerative Colitis
T cells (T helper 1 & 17 cells) and other related cytokines are said to be involved in the pathogenesis of ulcerative colitis. A. lappa has been shown to give relief against ulcerative colitis. Arctigenin from A. lappa inhibited proliferation of T cells in a dose-dependent manner that was induced by concanavalin A. It actually downregulates RORγt (Wu et al. 2015). There is enough evidence that A. lappa, more particularly arctigenin, significantly reduces subarachnoid hemorrhage–induced vasospasm in animal models (Tabassum et al. 2018).
10.4.6 Dermatological Effects of A. lappa
People from North America, Asia, and Europe have been using leaves of A. lappa and related species for various kinds of dermatological conditions, viz. psoriasis, abscesses, acne, ichthyosis, eczema, boils, and rashes. These actions might be due to the occurrence of phenolic compounds in the plant. Chan and colleagues reported that the antioxidant and anti-inflammatory potential of these compounds assist in detoxifying and mediate healing action of the plant (Chan et al. 2011). Burdock is used as an ingredient in various commercial cosmetic products because of the presence of various hydroxycinnamic acid derivatives which contribute in antimicrobial, anti-inflammatory, anti-collagenase, and anti-tyrosinase activities as well protection against ultraviolet radiations (Ahangarpour et al. 2017).
10.4.7 Hepatoprotective and Gastroprotective Activity
In vivo and in vitro antioxidant potential of the plant has been reported by Duh, Lin, and their colleagues in their work, which also showed that the plant possesses excellent hepatoprotective activity (Duh 1998; Lin et al. 2000). In 2018, Fierascu et al. (2018) reported antioxidant potential of A. lappa using phosphomolybdate and 2,2-diphenyl-1-picrylhydrazyl (DPPH) assays and reported that entire extract of burdock exhibits very high antioxidant potential because of the presence of very large quantity of polyphenols. A. lappa is also found to possess gastroprotective activity due to presence of 1,3-dicaffeoylquinic acid that has been isolated and characterized from the ethanolic fraction of the plant (Carlotto et al. 2015).
10.4.8 Clinical Trials
In a cohort study conducted in Japan in 2013 on safety and toxicity effects of “GBS-01,” an orally administered drug containing arctigenin as one of the ingredient on gemcitabine-resistant pancreatic cancer, patients were given GBS-1. Blood toxicity, dose-limited toxicities, and non-blood of grade 3–4 toxicities were taken as main endpoints after first 4 weeks of the trial. Increase in gamma-glutamyl transferase (GGT), total serum levels of glucose, and bilirubin were some of the adverse effects noted on the oral administration of GBS-01 (Tabassum et al. 2018).
10.5 Conclusion
A. lappa (burdock) seeds, leaves, roots, and fruits contain many phytoconstituents including volatile and nonvolatile compounds that have therapeutic potential against various kinds of diseases. Though roots are more frequently used, other parts of the plant have also shown a good amount of phytoconstituents, and hence promise. A survey into the literature shows that burdock and its isolated compounds possess a wide range of therapeutic uses, viz. anti-inflammatory, anticancer, antidiabetic, anti-obesity, hepatoprotective, and gastroprotective. The promising medicinal uses of the plant, however, necessitate to have an understanding about its adverse effects, while using it for various ailments. Therefore, further studies are important for better understanding of the role of the plant in preventing and treating any disease as well as any associated off-target effects of the plant.
References
Aboutabl EA, El-Tantawy ME, Shams MM (2013) Chemical composition and antimicrobial activity of volatile constituents from the roots, leaves, and seeds of Arctium lappa L.(Asteraceae) grown in Egypt. Egypt Pharm J 12(2):173
Ahangarpour A, Heidari H, Oroojan AA, Mirzavandi F, Khalil Nasr Esfehani ZDM (2017) Antidiabetic, hypolipidemic and hepatoprotective effects of Arctium lappa root’s hydro-alcoholic extract on nicotinamide-streptozotocin induced type 2 model of diabetes in male mice. Avicenna J Phytomed 7(2):169–179
An XY, Iong JX, In QX (2003) Simultaneous determination of chlorogenic acid, forsythin and arctiin in chinese traditional medicines preparation by reversed phase-HPLC. Chem Pharm Bull (Tokyo) 51(April):421–424
Arctium L, Iyazawa MM, Agi NY, Aguchi KT (2005) Inhibitory compounds of α-glucosidase activity from Arctium lappa L. Biochem Biotechnol 54(11):589–594
Awale S, Lu J, Kalauni SK, Kurashima Y, Tezuka Y, Kadota S, Esumi H (2006) Identification of arctigenin as an antitumor agent having the ability to eliminate the tolerance of cancer cells to nutrient starvation. Cancer Res 66(3):1751–1757. https://doi.org/10.1158/0008-5472.CAN-05-3143
Azizov UM, Khadzhieva UA, Rakhimov DA, Mezhlumyan LG, Salikhov SA (2012) Chemical composition of dry extract of Arctium lappa roots. Chem Nat Compd 47(6):1038–1039. https://doi.org/10.1007/s10600-012-0142-3
Donald G Barceloux (2008) Medical toxicology of natural substances: foods, fungi, medicinal herbs, plants, and venomous animals. Wiley Online Library. https://doi.org/10.1002/9780470330319
Boldizsár I, Füzfai Z, Tóth F, Sedlák É, Borsodi L, Molnár-perl I (2010) Mass fragmentation study of the trimethylsilyl derivatives of arctiin, matairesinoside, arctigenin, phylligenin, matairesinol, pinoresinol and methylarctigenin: their gas and liquid chromatographic analysis in plant extracts. J Chromatogr A 1217(10):1674–1682. https://doi.org/10.1016/j.chroma.2010.01.019
Burgmans MHDJL, Burton LC, Smallfield BM (1992) The production of Burdock (Arctium lappa L.) root in New Zealand—a preliminary study of a new vegetable. Agron Soc N Z 22:67–70
Carlotto J, da Silva LM, Dartora N, Maria-Ferreira D, Sabry DDA, Filho APS et al (2015) Identification of a dicaffeoylquinic acid isomer from Arctium lappa with a potent anti-ulcer activity. Talanta 135:50–57. https://doi.org/10.1016/j.talanta.2014.11.068
Carlotto J et al (2016) Polysaccharides from Arctium lappa L.: chemical structure and biological activity. Int J Biol Macromol 91:954–960. ISSN 0141-8130
Chan Y-S, Cheng L-N, Wu J-H, Chan E, Kwan Y-W, Lee SM-Y et al (2011) A review of the pharmacological effects of Arctium lappa (burdock). Inflammopharmacology 19(5):245–254. https://doi.org/10.1007/s10787-010-0062-4
Chan Y, Cheng L, Wu J, Chan E, Kwan Y, Lee SM et al (2010) A review of the pharmacological effects of Arctium lappa (burdock). Inflammopharmacology 19(5):245–254
Chen F-A, Wu A-B, Chen C-Y (2004) The influence of different treatments on the free radical scavenging activity of burdock and variations of its active components. Food Chem 86(4):479–484. https://doi.org/10.1016/j.foodchem.2003.09.020
Duh P-D (1998) Antioxidant activity of burdock (Arctium lappa Linné): its scavenging effect on free-radical and active oxygen. J Am Oil Chem Soc 75(4):455–461. https://doi.org/10.1007/s11746-998-0248-8
Ferracane R, Graziani G, Gallo M, Fogliano V, Ritieni A (2010) Metabolic profile of the bioactive compounds of burdock (Arctium lappa) seeds, roots and leaves. J Pharm Biomed Anal 51:399–404. https://doi.org/10.1016/j.jpba.2009.03.018
Fierascu RC, Georgiev MI, Fierascu I, Ungureanu C, Avramescu SM, Ortan A et al (2018) Mitodepressive, antioxidant, antifungal and anti-inflammatory effects of wild-growing Romanian native Arctium lappa L. (Asteraceae) and Veronica persica Poiret (Plantaginaceae). Food Chem Toxicol 111:44–52. https://doi.org/10.1016/j.fct.2017.11.008
Huang K, Liang X, Zhong Y, He W, Wang Z (2015) 5-Caffeoylquinic acid decreases diet-induced obesity in rats by modulating PPARα and LXRα transcription. J Sci Food Agric 95(9):1903–1910. https://doi.org/10.1002/jsfa.6896
Ichihara A, Oda K, Numata Y, Sakamura S (1976) Lappaol A and B, novel lignans from Arctium lappa L. Tetrahedron Lett 17:3961–3964
Jeelani S, Khuroo MA (2012) Triterpenoids from Arctium lappa. Nat Prod Res 26(7):654–658. https://doi.org/10.1080/14786419.2010.541886
Kardošová A, Ebringerová A, Alföldi J, Nosál’ová G, Fraňová S, Hřı́balová, V. (2003) A biologically active fructan from the roots of Arctium lappa L., var. Herkules. Int J Biol Macromol 33(1–3):135–140. https://doi.org/10.1016/S0141-8130(03)00079-5
Kato Y, Watanabe T (1993) Isolation and characterization of a xyloglucan from gobo (Arctium lappa L.). Biosci Biotechnol Biochem 57(9):1591–1592. https://doi.org/10.1271/bbb.57.1591
Kolacz NM, Jaroch MT, Bear ML, Hess RF (2014a) The effect of burns & wounds (B&W)/burdock leaf therapy on burn-injured Amish patients: a pilot study measuring pain levels, infection rates, and healing times. J Holist Nurs 32(4):327–340. https://doi.org/10.1177/0898010114525683
Kolacz NM, Jaroch MT, Bear ML, Hess RF (2014b) The effect of burns & wounds (B&W)/Burdock leaf therapy on burn-injured Amish patients. J Holist Nurs 32(4):327–340. https://doi.org/10.1177/0898010114525683
Kuo D, Hung M, Hung C, Liu L, Chen F, Shieh P et al (2012) Body weight management effect of burdock (Arctium lappa L.) root is associated with the activation of AMP-activated protein kinase in human HepG2 cells. Food Chem 134(3):1320–1326. https://doi.org/10.1016/j.foodchem.2012.03.023
Lee B-I, Nugroho A, Bachri MS, Choi J, Lee KR, Choi JS et al (2010) Anti-ulcerogenic effect and HPLC analysis of the caffeoylquinic acid-rich extract from Ligularia stenocephala. Biol Pharm Bull 33(3):493–497. https://doi.org/10.1248/bpb.33.493
Li J et al (2013) Preparation of inulin-type fructooligosaccharides using fast protein liquid chromatography coupled with refractive index detection. J Chromatogr A 1308:52–57. https://doi.org/10.1016/j.chroma.2013.08.012
Lin S, Chung T, Lin C, Ueng T-H, Lin Y, Lin S, Wang L (2000) Hepatoprotective effects of Arctium Lappa on carbon tetrachloride- and acetaminophen-induced liver damage. Am J Chin Med 28(02):163–173. https://doi.org/10.1142/S0192415X00000210
Liu D, Qu W, Zhao L, Yu J (2012) A novel dimeric diarylheptanoid from the rhizomes of Alpinia officinarum. Chin Chem Lett 23(2):189–192. https://doi.org/10.1016/j.cclet.2011.11.013
Lou Z, Wang H, Zhu S, Zhang M, Gao C, Ma C, Wang ZL (2010) Improved extraction and identification by ultra-performance liquid chromatography tandem mass spectrometry of phenolic compounds in burdock leaves. J Chromatogr A 1217:2441–2446
Machado FB, Yamamoto RE, Zanoli K, Nocchi SR, Novello CR, Schuquel IT, Sakuragui CM, Luftmann H, Ueda-Nakamura T, Nakamura CV, de Mello JC (2012) Evaluation of the antiproliferative activity of the leaves from Arctium lappa by a bioassay-guided fractionation. Molecules (Basel, Switzerland) 17(2):1852–1859. https://doi.org/10.3390/molecules17021852
Maghsoumi-Norouzabad L, Alipoor B, Abed R, Eftekhar Sadat B, Mesgari-Abbasi M, Asghari Jafarabadi M (2016) Effects of Arctium lappa L. (Burdock) root tea on inflammatory status and oxidative stress in patients with knee osteoarthritis. Int J Rheum Dis 19(3):255–261. https://doi.org/10.1111/1756-185X.12477
Maria B, Werner FDP, Sassaki GL, Iacomini M, Cipriani TR (2016) Polysaccharides from Arctium lappa L.: chemical structure and biological activity. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2016.06.033
Ming DS, Guns ES, Eberding A, Towers GHN (2004) Isolation and characterization of compounds with anti-prostate cancer activity from Arctium lappa L. using bioactivity-guided fractionation. Pharm Biol 42(1):44–48. https://doi.org/10.1080/13880200490505474
Miyazawa M, Yagi N, Taguchi K (2005) Inhibitory compounds of α-glucosidase activity from Arctium lappa L. J Oleo Sci 54(11):589–559
Moro TMA, Celegatti CM, Pereira APA, Lopes AS, Barbin DF, Pastore GM, Clerici MTPS (2018) Use of burdock root flour as a prebiotic ingredient in cookies. LWT 90:540–546. https://doi.org/10.1016/j.lwt.2017.12.059
Park S, Hong S, Han X et al (2007) Lignans from Arctium lappa and their inhibition of LPS-induced nitric oxide production. Chem Pharm Bull (Tokyo) 55(1):150–152
Pereira JV, Bergamo DCB, Pereira JO, França S d C, Pietro RCLR, Silva-Sousa YTC (2005) Antimicrobial activity of Arctium lappa constituents against microorganisms commonly found in endodontic infections. Braz Dent J 16(3):192–196. https://doi.org/10.1590/S0103-64402005000300004
Rajasekharan SK, Ramesh S, Bakkiyaraj D, Elangomathavan R, Kamalanathan C (2015) Burdock root extracts limit quorum-sensing-controlled phenotypes and biofilm architecture in major urinary tract pathogens. Urolithiasis 43(1):29–40. https://doi.org/10.1007/s00240-014-0720-x
Rajasekharan SK, Ramesh S, Satish AS, Lee J (2017) Antibiofilm and anti-β-lactamase activities of burdock root extract and chlorogenic acid against Klebsiella pneumoniae. J Microbiol Biotechnol 27(3):542–551. https://doi.org/10.4014/jmb.1609.09043
Saleh NAM, Bohm BA (1971) Flavonoids of Arctium minus (Compositae). Experientia 27(12):1494–1494. https://doi.org/10.1007/BF02154314
Savina AA, Sheichenko VI, Stikhin YV et al (2006) Sesquiterpene lactones in juice of great burdock leaves. Pharm Chem J 40:624–626. https://doi.org/10.1007/s11094-006-0207-3
Susanti S, Iwasaki H, Inafuku M, Taira N, Oku H (2013) Mechanism of arctigenin-mediated specific cytotoxicity against human lung adenocarcinoma cell lines. Phytomedicine 21(1):39–46. https://doi.org/10.1016/j.phymed.2013.08.003
Swamy MK (2019) Arctium species secondary metabolites chemodiversity and bioactivities. Front Plant Sci 10:834. https://doi.org/10.3389/fpls.2019.00834
Tabassum S, Perk AA, Qureshi MZ, Sabitaliyevich UY, Zhenisovna TG, Farooqi AA (2018) Arctium lappa. Nonvitamin and nonmineral nutritional supplements, vol 2. Elsevier Inc. https://doi.org/10.1016/B978-0-12-812491-8.00039-4
Tamayo C, Richardson MA, Diamond S, Skoda I (2000) The chemistry and biological activity of herbs used in flor-essence? Herbal tonic and Essiac. Phytother Res 14(1):1–14. https://doi.org/10.1002/(SICI)1099-1573(200002)14:1<1::AID-PTR580>3.0.CO;2-O
The Scientific Foundation for Herbal Medicinal Products (2003) Scientific Cooperative on Phytotherapy
Wang D, Bădărau AS, Swamy MK, Shaw S, Maggi F, da Silva LE et al (2019) Arctium species secondary metabolites chemodiversity and bioactivities. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00834
Wang X, Li F, Sun Q, Yuan J, Jiang T, Zheng C (2005) Application of preparative high-speed counter-current chromatography for separation and purification of arctiin from Fructus Arctii. J Chromatogr A 1063:247–251. https://doi.org/10.1016/j.chroma.2004.11.077
Wang Y, Zhang N, Kan J, Zhang X, Wu X, Sun R et al (2019) Structural characterization of water-soluble polysaccharide from Arctium lappa and its effects on colitis mice. Carbohydr Polym 213:89–99. https://doi.org/10.1016/j.carbpol.2019.02.090
Washino T, Yoshikura M, Obata S (1986) New sulfur—containing acetylenic compounds from Arctium lappa. Agric Biol Chem 50(2):263–269
Wu X, Dou Y, Yang Y, Bian D, Luo J, Tong B et al (2015) Arctigenin exerts anti-colitis efficacy through inhibiting the differentiation of Th1 and Th17 cells via an mTORC1-dependent pathway. Biochem Pharmacol 96(4):323–336. https://doi.org/10.1016/j.bcp.2015.06.008
Yang Y-N, Zhang F, Feng Z-M, Jiang J-S, Zhang P-C (2012) Two new neolignan glucosides from Arctii fructus. J Asian Nat Prod Res 14(10):981–985. https://doi.org/10.1080/10286020.2012.729050
Yao X, Zhu F, Zhao Z, Liu C, Luo L, Yin Z (2011) Arctigenin enhances chemosensitivity of cancer cells to cisplatin through inhibition of the STAT3 signaling pathway. J Cell Biochem 112(10):2837–2849. https://doi.org/10.1002/jcb.23198
Yoo JM, Yang JH, Yang HJ, Cho WK, Ma JY (2016) Inhibitory effect of fermented Arctium lappa fruit extract on the IgE-mediated allergic response in RBL2H3 cells. Int J Mol Med 37(2):501–508
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Mir, S.A. et al. (2022). Arctium lappa: A Review on Its Phytochemistry and Pharmacology. In: Masoodi, M.H., Rehman, M.U. (eds) Edible Plants in Health and Diseases . Springer, Singapore. https://doi.org/10.1007/978-981-16-4959-2_10
Download citation
DOI: https://doi.org/10.1007/978-981-16-4959-2_10
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4958-5
Online ISBN: 978-981-16-4959-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)