Abstract
Lignocellulosic waste (LW) and food waste (FW) have great potential for biomethane production. The recalcitrant nature of LW limits its use in anaerobic processes. Without pretreatment, it is difficult to utilize LW at higher organic loading rates (OLRs) through an anaerobic process. Therefore, pretreatment is a compulsory step for efficient utilization (i.e., at higher OLRs) of LW. There are various techniques of pretreatment of LW, and every method has its own advantages and disadvantages and generally makes the process expansive. Pretreatment of LW can change the biomass structure by removing lignin, increasing the surface area, and decreasing the crystalline nature and length of the polymer chain. On the other hand, FW is readily digestible biomass and can be utilized at a comparatively higher organic loading rate than that of LW. Although mono-anaerobic digestion of FW at higher organic loading rates leads to the accumulation of higher volatile fatty acids, the process becomes unstable. This chapter provides state-of-the-art knowledge on the current status of mono-anaerobic digestion of LW and FW for sustainable biogas production, limitations such as pretreatment, low organic loading rates, higher hydraulic retention time, low buffering capacity, and higher accumulation of volatile fatty acids. Besides, the advantages of co-anaerobic digestion of LW with FW over mono-digestion of LW and FW, the technological advancements being made are also discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
4.1 Process of Sustainable Production of Biogas
4.1.1 Anaerobic Digestion
Biodegradable matter is decomposed by microorganisms in the presence or absence of oxygen. The process of decomposition of any organic substance in the presence of oxygen is known as aerobic digestion, whereas if the decomposing takes place in the absence of oxygen, it is generally known as anaerobic digestion. A general process of anaerobic digestion process is presented in Fig. 4.1. Biogas is one of the types of renewable energy which is generally an outcome of the anaerobic digestion process. Amongst the various advantages, one of the main advantages of anaerobic digestion is that a diversity of biodegradable matters can be utilized via this process (Dolan et al. 2011).
Biogas generally constitutes methane (CH4), carbon dioxide (CO2), hydrogen sulfide (H2S), hydrogen, nitrogen, ammonia, oxygen, and water. However, primarily CH4 and CO2 comprise approximately 90% (Naik et al. 2010; Choudhary et al. 2020a). Apart from CH4, one of the very important outcomes of this process is digestate. Digestate is generally a slurry that produces after the biochemical reaction during the anaerobic digestion process. Dominantly, it is water and a very small quantity of solids. This is generally very rich in nutrients and widely used as a fertilizer during farming (Tampio et al. 2016). Therefore, anaerobic digestion diminishes the demand for fossil fuels which otherwise would be required during the generation of conventional chemical fertilizers. Anaerobic digestion has other several advantages as well, such as it reduces the reliance on the usages of fossil fuels and hence indirectly helps in the curtailment of greenhouse gases into the environment which generally takes place during the burning of the conventional fossil fuel.
Due to all these advantages, currently anaerobic digestion is becoming a very popular waste management technology across the world. It is also being practiced and becoming very popular amongst the industries in developing countries due to its simple design, working, and requirement of low-capital investment (Börjesson and Mattiasson 2008; Forgács 2012).
Under optimum system variables such as organic loading rate (OLR) and temperature, the process may yield high usage of biomass, i.e., the ratio of energy output/input ratio of 28 MJ/kg (Kabir et al. 2015).
The best feature about this technology is that small-scale and pilot-scale anaerobic reactors can be developed at the local level and be fed with materials available at the regional level. Due to this reason, a huge number of small-scale (household) anaerobic digesters can be found in developing countries. Based on some studies available, approximately 30 million, 3.8 million, and approximately 200,000 anaerobic digesters are running in China, India, and Nepal, respectively (Jiang et al. 2011; Rajendran et al. 2012). However, in African nations, this technology has not been established much, and only very few anaerobic digesters are running at the small-scale level (Amigun et al. 2008). On the other hand, the scenario of anaerobic digesters is opposite in European countries and America. In these regions, the anaerobic digesters are larger than small-scale household digesters when compared with developing countries. In Europe, various waste materials such as sludge, energy crops, and different animal dungs are utilized in anaerobic digestion, and approximately 10,000 anaerobic reactors are working here. According to a study, the anaerobic digesters will be increased by five-fold in Europe in future, whereas a study reported that the number of biogas plants will reach 200 million by 2020 (Deublein and Steinhauser 2008).
4.1.2 Biochemistry of Anaerobic Digestion
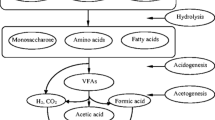
Anaerobic digestion is a process in which various biological and chemical processes occur simultaneously. During this process, the biodegradable matter is degraded by a variety of microorganisms and biogas is a major output of this process. Hydrolysis, acidogenesis, acetogenesis, and methanogenesis are the biochemical process which simultaneously takes place within the reactor (Fig. 4.2). In these processes, a variety of bacteria and archaea are involved which have a syntrophic relationship with each other (Deublein and Steinhauser 2008).
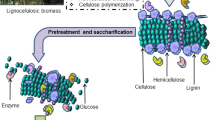
In anaerobic digestion, hydrolysis is the primary step. During hydrolysis, hydrolytic bacteria are involved which hydrolyze the biodegradable matter. Generally, in this process, large molecules are broken down into smaller ones, i.e., polymers are degraded into soluble monomers and oligomers. Biochemistry of anaerobic digestion of FW is illustrated in Fig. 4.3.
Cellulases, hemicellulases, lipases, amylases, and proteases are the enzymes associated in this phase (Taherzadeh and Karimi 2008). Almost, all types of biodegradable matters can be decomposed, and all the abovementioned enzymes are involved in this. However, it has been noticed that during the anaerobic digestion of any lignocellulosic biomass, lignin does not decompose (Fernandes et al. 2009).
Hydrolysis is a time-dependent process, and it decisively depends upon the nature of biodegradable matter, i.e., complex or rapidly digestible. For example, the hydrolysis of food waste is rapid (Choudhary et al. 2020b) when compared with any lignocellulosic biomass. Nevertheless, in the case of complex biodegradable matter, rapid hydrolysis can be accomplished if suitable enzymes are generated by the microorganisms and suitable contact between biodegradable matter and enzyme is achieved (Taherzadeh and Karimi 2008). Even though, the complex biodegradable matter may take a few weeks to decompose (Deublein and Steinhauser 2008). Therefore, the hydrolysis phase in a complex biodegradable matter such as lignocellulosic wastes is measured as the rate-limiting step (Taherzadeh and Karimi 2008).
Acidogenesis is an acid-forming phase in the anaerobic digestion process. The by-products of the hydrolysis phase are utilized in this phase and are further converted into volatile fatty acids (VFAs) with the help of obligate and facultative anaerobes. Propionic acid, valeric acid, formic acid, acetic acid, and butyric acid are amongst the main VFAs produced during this phase. Apart from the VFAs, alcohols and hydrogen are other by-products in this process. Hydrogen has a major role during this process; it regulates the expected by-products in this phase.
A low partial hydrogen pressure results in hydrogen, carbon dioxide, and acetate. On the contrary, if partial pressure exerted by the hydrogen is high, then the formation of alcohols and VFAs takes place. Therefore, during the process, the condition should be optimal, i.e., to avoid high partial pressure, otherwise accumulation of VFA takes place which may result in the inhibition of the whole process. These products are furthermore decomposed under the optimal conditions for the production of biogas (Schink 1997).
The by-products of the acidogenesis phase are subjected to two different pathways. In the first pathway, hydrogen, acetate, and carbon dioxide can straightforwardly be used by methanogens for the production of methane. Hence, alcohols carrying greater than one carbon atoms and VFAs carrying greater than two carbon atoms are further degraded to hydrogen and acetate in this phase by obligatory hydrogen-producing bacteria (Bryant 1979; Schink 1997).
During the methanogenesis phase, anaerobic archaea convert acetate or hydrogen to methane and carbon dioxide.
4.1.3 Variable of AD
Various factors such as pH, retention time, temperature, organic loading rate (OLR), mixing, and macronutrient and micronutrient availability may influence the optimum anaerobic digestion decisively. Thus, there is always a need to monitor and maintain these parameters for the optimum performance of the microorganisms (Ward et al. 2008). The characteristics and nature of the substrate is also a very important factor. The decision of the OLR has a great dependency on the nature of the substrate. A decision of an optimum OLR may help in overall optimum performance of the process; therefore, there is a huge requirement to monitor the OLR regularly. Generally, the OLR has been defined as the feed (Kg VS) added per meter cube of the working volume of the digester with respect to time, i.e., Kg VS/m3/d. The organic loading rate of any process can be computed using Eq. (4.1).
Generally, the reactor is started up with lower OLR and later increased gradually up to the optimum OLR. If the system is fed with higher OLR than the optimum, then generally accumulation of higher VFA has been experienced by many researchers and which further led to lower CH4 content, higher CO2, higher pH, and higher H2S concentration, i.e., instability of the reactor and overall anaerobic digestion process (Mata-Alvarez et al. 2000; Bouallagui et al. 2004; Choudhary et al. 2020b). Nonetheless, reactors running over extremely low OLR have no techno-economic feasibilities because the real capacity of the reactor has not been utilized.
Apart from that, another crucial parameter is retention time and generally reported as hydraulic retention time (HRT). The HRT is the time for which liquid sludge exist in the digester. It is also generally called solid retention time (SRT), which indicates the duration spent by a solid particle within the reactor or with microorganisms (Appels et al. 2008). The HRT can be calculated using Eq. (4.2).
HRT is calculated based on the following formula, where V is the working volume of the digester in m3, Q is the flow rate of the sludge (m3/day).
Generally, if the feed is complex to digest, then HRT is more significant; on the other hand, if the feed is easily digestible, then SRT is more important (Speece 2008). To increase the efficiency of the process, a short retention time is generally favorable. A shorter retention time may reduce the overall capital investment of the project(Chandra et al. 2012). There is always necessity of managing OLR and HRT in such a manner that optimum anaerobic digestion can occur. This means that while running the digester at higher OLR, the HRT should be appropriately higher to ensure an adequate interaction between the substrate and the microorganisms(Demirer and Chen 2005). For continuously and semi-continuous anaerobic reactors, HRT and SRT are equal. Nonetheless, these reactors are not subjected to re-circulation; in the case of re-circulation, the HRT and SRT will increase.
Temperature is amongst the most important parameters which can affect the whole anaerobic digestion process decisively. Generally, temperature fluctuations during the anaerobic digestion process may be favorable for a certain group of microorganisms, but may not be favorable for the other groups. The process of methanogenesis is most affected by any fluctuation in the temperature during the anaerobic digestion process. Anaerobic digestion is performed at three temperature ranges, i.e., thermophilic, mesophilic, and psychrophilic. The growth optimal is around 10, 37, and 50 °C for thermophilic, mesophilic, and psychrophilic, respectively (Kashyap et al. 2003; Wiegel et al. 2007; Coelho et al. 2011).
pH is also an important parameter during the anaerobic digestion process. During the process of hydrolysis, acidogenesis, acetogenesis, and methanogenesis, there is a requirement of a wide range of pH (Mittal 1996). A significant number of microorganisms prefer the pH in the range of 7.0–8.5 (Kanokwan 2006). There are, however, microorganisms that can survive in an acidic and basic range of pH. The acidogenic microorganisms can survive in the acid range, i.e., near pH 5.0. Nevertheless, in accordance with the microorganisms involved in all the processes, the pH of the anaerobic process needs to be maintained in the range of 6.6–7.3 (Babel et al. 2004; Sitorus et al. 2013; Zhou et al. 2016). The pH beyond this range may affect the overall process, more specifically during the methanogenesis process (Kim et al. 2004; Taconi et al. 2008). An optimum OLR is highly recommended for the optimum pH during the whole process, which is attributed to the fact that if the anaerobic digester is subjected to the higher OLR, it may affect the intermediate by-products such as VFA which may further affect the pH adversely. It is recommended that the concentration of VFA should always be less than 2000 mg/L for an optimum anaerobic digestion process (Jain and Mattiasson 1998).
However, if the concentration of the VFA is higher in the system, it can be lowered by adding or maintaining the alkalinity in the system. The alkalinity can be defined as the buffering capacity of the system and generally measured in terms of mg/L as CaCO3. If there is adequate alkalinity present in the system, then it can neutralize the high VFA concentration, and hence, this will lead the system towards the optimum pH, i.e., near to 7.0. Few substrates have the alkalinity by default such as animal dungs. On the other hand, few substrates have low-buffering capacities such as food wastes and lignocellulosic wastes (Banks and Humphreys 1998). Therefore, monitoring of alkalinity and VFA in such systems becomes mandatory. A great indicator of the stability of the anaerobic digestion process is the ratio of VFA to alkalinity. It is suggested that for optimum anaerobic digestion, this ratio should be nearly about 0.3–0.4. However, it should never exceed 0.8. The VFA/alkalinity ratio exceeding 0.8 shows system instability, and hence, process inhibition, or less efficiency of the process (Wang et al. 2012).
Nutrients are significantly important for any anaerobic digestion process. There must always be an abundance of nutrients in the system, and even a small shortage of them can cause process inhibition. The synthesis and growth of enzymes are associated with biochemical and metabolic pathways of the process’s microorganisms. Generally, nutrients can be categorized into two types, i.e., micronutrients and macronutrients (Mara and Horan 2003).
The much known fundamental macronutrients are nitrogen (N), phosphorus (P), carbon (C), and sulfur (S). These are very important for the multiplication and growth of microorganisms. During the anaerobic digestion process, carbon and nitrogen levels play even more critical role. Nitrogen is very important for the overall development and growth of the microorganisms, whereas carbon acts as food for the microorganisms. Deficiency of nitrogen in any system may lead to unsatisfactory consumption of the carbon, or in other words, it will prohibit the growth of the microorganisms (Resch et al. 2011). As a result of that, the overall biogas production will be reduced. Therefore, the ratio of C to N is always a decisive parameter during the anaerobic digestion process (Hobsen et al. 1981; Chandra et al. 2012), and it can be adjusted/optimized by adjusting the ratios of substrates during the design of OLR.
4.2 Types of Lignocellulosic and Food Wastes
4.2.1 Lignocellulosic Material as a Substrate
As we have discussed in the previous section of this chapter, for the production of biogas, various substrates such as animal dungs, organic fraction of municipal solid waste, wastewater, sewage sludge, and agricultural residues can be used (Koniuszewska et al. 2020; Ferdeș et al. 2020; Atelge et al. 2020; Choudhary et al. 2020a, b, c). Amongst these substrates, several are lignocellulosic. There is ample availability of lignocellulosic substrates across the world. The carbohydrate content present in the lignocellulosic wastes makes it more attractive for the production of biogas via anaerobic digestion. Generally, lignocellulosic substrates can be divided into two categories, i.e., lignocellulosic residuals and cultivated feedstocks, known as energy crops. The major drawback with the lignocellulosic residuals is that they have a high percentage of lignin and therefore are less suitable for the utilization in anaerobic digestion. Due to this reason, only lignocellulosic residues as substrate (without pretreatment and co-digestion) have relatively low methane yield (Kainthola et al. 2019a). On the other hand, energy crops have a smaller fraction of lignin when compared to lignocellulosic residuals. Energy crops primarily consist of cellulose and hemicellulose (Kabir et al. 2015; Chen et al. 2018). Moreover, along with cellulose and hemicellulose, the energy crops’ residues consist of various non-structural carbohydrates such as fructose, fructans, pectins, glucose, sucrose, and extractives (Kabir et al. 2014). The utilization of lignocellulosic wastes such as giant reed stems, wheat straw (Dell’Omo and Spena 2020), rice straw (Liu et al. 2019), corn stover (You et al. 2019), and Napier grass (Phuttaro et al. 2019) is common across the world (Kainthola et al. 2019a).
4.2.2 Food Waste as a Substrate
Food waste (FW) is a great substrate for anaerobic digestion, and it has a huge potential for producing biomethane (Pramanik et al. 2019; Choudhary et al. 2020b). FW generally consists of complex and organic material. There are various types of FW, such as vegetable and fruit waste, brewery waste, kitchen waste, and dairy waste (Xu et al. 2018). The composition and characteristics of FW vary with the geographical area (Meng et al. 2015; Xu et al. 2018). FW consists of carbohydrates, fats, protein, and sugar. FW is generally acidic and has less alkalinity. Fisgativa et al. (2016) studied various types of food waste and reported that the average pH of FW was 5.1; the C/N was reported at 18.5%. Also, they have reported carbohydrates, protein, and fat fraction in the FW as 57.2%, 62.2%, and 15%, respectively (Fisgativa et al. 2016). Generally, carbohydrates and protein have a rapid hydrolysis rate when compared to lipids.
Vegetable and fruit waste have low lipid and comparatively higher cellulose content. Due to the presence of animal fat and vegetable oil, the kitchen waste carries high lipid content (Bong et al. 2018). The lipid content may vary in the range of 11.8–33.22% in the case of fruit and vegetable and kitchen waste, respectively (Wang et al. 2014; Yong et al. 2015). FW with higher lipid content can produce significantly higher biomethane when compared to protein and carbohydrates (Li et al. 2017). Nevertheless, very high lipid content may inhibit the process as well because of the formation of a high concentration of long-chain fatty acids (Leung and Wang 2016; Li et al. 2017). FW carrying significantly higher carbohydrate may decisively affect the C/N ratio. This is attributed to the fact that high carbohydrate content may increase carbon content, and hence, quick acidification may occur due to heavy loading of carbon into the system (Li et al. 2017).
The total solid may fall in the range of 10.7–41% in any type of food waste which indicates significantly higher moisture content, i.e., about 60–90%. Due to the presence of higher moisture content, FW is also considered as a rapidly digestible substrate for the anaerobic digestion (Zhang et al. 2014).
The C/N of the FW may vary in the range of 12.7–28.84. The pH generally falls in the acidic range, i.e., 4.1–6.5. The biomethane potential of every variety of FW may vary in the range of 346–551.4 mL/gVS, which is comparatively higher than animal dungs and various other wastes (Lehtomäki et al. 2007).
4.3 Mono-digestion and its Limitations
When only one substrate is fed into the digester, such process is referred to as mono-digestion. Mono-digestion of lignocellulosic waste and FW has several limitations that will be discussed further.
During mono-digestion of FW and lignocellulosic substrates when the anaerobic digester runs at comparatively higher OLRs, the accumulation of VFA is a major limitation of the anaerobic mono-digestion process. Due to this reason, the process faces several challenges such as instability, ammonia inhibition, insufficient alkalinity, production of H2S, and less ultimate biomethane potential.
On the other hand, if the digester runs at lower OLRs, then the process becomes economically unfeasible. Secondly, to enhance the biomethane yield of the process, often various pretreatments are suggested, which again makes the process less environment friendly and economically less attractive (Mata-Alvarez et al. 2011; Nghiem et al. 2017).
In the case of lignocellulosic substrates, the C/N ratio is significantly high, which creates nitrogen deficiency during the anaerobic digestion process. Therefore, the risk of production of inhibitors such as furfural and hydroxymethylfurfural becomes very high. Moreover, due to the presence of lignin, hydrolysis occurs at a relatively slower pace and consequently the HRT of the process increases significantly (Kabir et al. 2013; Yong et al. 2015; Achinas et al. 2017). Apart from this, the low C/N ratio is also a major limitation of the mono-digestion of FW (David et al. 2018).
4.4 Pretreatment Technologies
4.4.1 Pretreatment of FW
FW generally consists of a rapidly digestible fraction and complex organic fraction. The rapidly digestible fraction in FW is often carbohydrates, and the complex organic fractions are lipids and proteins. Hence, complete biomethane potential is not achieved without pretreatment. With the help of pretreatment, biodegradability of recalcitrant organic fraction of FW can be increased significantly. It is a well-understood fact that in the case of complex substrates such as lignocellulosic substrate, hydrolysis is a rate-limiting phase, whereas for rapidly digestible substrates such as FW, methanogenesis is the rate-limiting step (Li et al. 2018). The efficiency of hydrolysis can be decisively affected by the operating temperature and nature of the organic matter (Srisowmeya et al. 2020). Various methods have been used to speed up the hydrolysis rate.
Often during the physical pretreatments, the size of substrates is reduced and the morphological structure of the substrates is also changed and therefore increases its solubilization (Ma et al. 2018). An increment of 28% in methane has been noted while reducing the particle size (by mechanical grinding) of the FW by 53%. Nevertheless, excessive reduction of the particle size has resulted in the accumulation of VFA and later high methane content. Hence, during the mechanical pretreatment, the primary objective should be to optimize the particle size of the substrate.
The solubility and accessibility of the FW can also be increased by ultrasonication. Ultrasonication reduces the complexity of the substrate by reducing its particle size mechanically. By ultrasonication, methane yield can be increased by 1.21–1.58 times (Nasr et al. 2012). For rapidly digestible substrates such as kitchen waste, microwave pretreatments along with electromagnetic energy have neither been found much effective for hydrolysis nor these are found economically feasible (Shahriari et al. 2013).
During the thermal pretreatment, the surface area of the organic matter is increased and therefore increasing the contact between microorganisms and the organic matter, thereby leading to better methane yield. Longer retention time (>4 h) and higher temperature (>120 °C) during the pretreatment have shown adverse effects on proteins and carbohydrates and resulted in bioproducts such as melanoidins and amodori which are difficult to degrade under anaerobic conditions (Vavilin et al. 2008). Additionally, during longer retention time and thermal pretreatment, loss of volatile solids and sugar occurs (Eskicioglu et al. 2006). Therefore, thermal pretreatment for longer retention time and a higher temperature is not recommended (Ariunbaatar et al. 2014b). The FW with the higher complex fraction ozonation pretreatment is more appropriate (Ariunbaatar et al. 2014b).
Pretreatment with the help of hydrolytic enzymes is also a highly efficient technique (Ma et al. 2018). It can increase the hydrolytic efficiency by substrate-specific action of enzymes owing to high energy recovery. Although pretreatment with the help of hydrolytic enzymes ensures enhanced methane yield, it has economical limitations on a commercial scale (Ma et al. 2018).
Due to the wide diversity of FW, it is very difficult to choose the most suitable pretreatment method though the application of pretreatments is important to ensure the utmost efficiency and methane yield (Ariunbaatar et al. 2014a).
4.4.2 Pretreatment of Lignocellulosic Biomass
In case of lignocellulosic waste, the biodegradable fraction becomes unavailable for the microorganisms involved in the process, and hence, it results in lower methane yield. Sometimes, even this may become the reason for the accumulation of inhibitory compounds within the digester. Therefore, various process enhancement techniques are recommended which increases the hydrolysis rate and overall degradability of the substrate. It is recommended that pretreatment must fulfill certain criteria such as (a) during the process, there should not be any formation of inhibitory substances; (b) there should not be much loss of carbohydrate; and (c) it should be cost-effective. The fundamental of pretreatment of any lignocellulosic biomass includes two processes, i.e., separation of lignin from the overall structure and exposing the rest matrix to degrading enzymes and disruption of the lignocellulosic matrix into cellulose, hemicellulose, and lignin (Sun and Cheng 2002; Vivekanand et al. 2012). Alike FW, hydrolysis is a rate-limiting step in the case of lignocellulosic biomasses, especially in cases of recalcitrant agriculture residues.
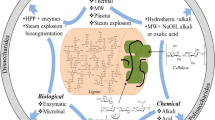
Figure 4.4 represents the various pretreatment techniques for lignocellulosic biomass. Often physical, chemical, thermophysical, thermochemical, and biological pretreatments are done to treat the lignocellulosic biomasses. A very basic and preliminary technique to break the lignin structure is grinding (size reduction). Grinding helps in increasing the surface area, porosity, altering the polymerization degree, and diminishing the crystallinity of the biomass (Hajji and Rhachi 2013; Zhang and Banks 2013; Lindmark et al. 2014).
Various chemical agents are also used as a catalyst for disrupting and delignification of the bond of the lignocellulosic matrix in various biomass substrates (Boonterm et al. 2016). Different acids used during the pretreatment process are HNO3, H2O2, H2SO4, HCl, etc.
Lime, ammonia, NaOH, Na2CO3, etc., are used in alkaline pretreatments. Pretreatment by alkaline agents can increase the surface area, porosity, altering the polymerization degree, and disrupting the lignin of the biomass. Aqueous ethanol and acetone–butanol–ethanol are used for pretreatments as an organic solvent.
Although the usage of chemical agents is simple and effective, sometimes it is observed that these pretreatments produce inhibitory compounds that may further need treatment or they may inhibit the anaerobic digestion process.
Temperature is also used as a tool for pretreatment of different lignocellulosic substrates, and such methods are referred to as thermal pretreatment. Thermal pretreatment can increase the porosity of the surface and enhance the destruction of the lignin layer. Any liquid at higher temperatures hydrolyses the lignocellulosic fraction of the biomass. This is because at high temperature and pressure, water molecules break down into OH− and H3O+ that further assists in catalytic conversion of lignocellulosic biomass. Delignification and improved porosity can be achieved at a higher temperature and short reaction time with dilute H2SO4, ammonia recycle percolation (APR), or steam explosion. Usually, after thermal pretreatments, an increased cellulosic content is achieved. Moreover, during this process, removal of some hemicellulosic content also takes place, which further helps in increase of the surface for enzymatic attack.
Under optimum microwave intensity and irradiance time, the hemicellulose and cellulose removal efficiency can be improved up to 30.6–43.3% (Ma et al. 2009). In fact, with the help of microwave pretreatment, a delignification of 6% can be achieved and as a result of that, hydrolysis is improved (Zhu et al. 2005). Although furan derivatives, phenolic substances, etc., are some inhibitory compounds that are produced during pretreatment with microwave irradiation, which later disturbs the anaerobic digestion. Therefore, such inhibitors are also a decisive step during bioconversion of lignocellulosic substrate to methane (Palmqvist and Hahn-Hägerdal 2000; Putro et al. 2016). The production of inhibitory compounds and higher operation cost and energy demand are the biggest drawbacks of physical, thermal, and chemical pretreatments.
On the other hand, biological pretreatments with microbes and enzymes provide the significantly environment friendly solution for the bioconversion of lignocellulosic substrates. Though, one of the main problems with pretreatments with microbes and enzymes is lesser surface area accessibility of microbes and microbial products on lignocellulose for efficient conversion to hydrolytic products (Kainthola et al. 2019b). The microbial route was found to be one of the economic and effective ways for the delignification and cellulolytic hydrolysis (Ghosh and Bhattacharyya 1999).
Microorganisms such as white, brown, and soft-rot fungi are involved in lignin and hemicellulose degradation. Temperature and pH during the anaerobic digestion process can hinder the biological pretreatment process. In such circumstances, extremophiles are also found to be a good alternative because they can sustain even in the harsh environment. During the last decade, various microorganisms have been developed that can sustain in an extreme environment and work efficiently. Clostridium thermocellum, Caldicellulosiruptor saccharolyticus, and Caldicellulosiruptor bescii DSM 6725 are some thermophilic bacteria that have gained huge attention in the past 10 years (Li et al. 2014).
4.5 Co-Digestion and its Advantages
When two substrates are digested simultaneously in an anaerobic digester for the production of biogas, the process is generally referred to as anaerobic co-digestion. In the last few decades, anaerobic co-digestion has gained ample popularity amongst the researchers and industries because anaerobic co-digestion has improved the process in various aspects when compared to mono-digestion. As in most of the cases, co-digestion provides missing nutrients, buffer, and sometimes moisture content required in the digester which has a positive synergic effect on the overall process (Mata-Alvarez et al. 2000).
We have already discussed the importance of the C/N ratio in the anaerobic digestion process. Mixing any substrate such as any lignocellulosic substrate whose C/N is comparatively high with a substrate whose C/N is low such as FW can optimize the overall C/N of the process.
In case of lignocellulosic substrates, high C/N ratio, lignin percentage, and contamination with pesticides can be resolved with the help of anaerobic co-digestion (Kainthola et al. 2019a). Some of the advantages of anaerobic co-digestion are (a) enhancement of the overall process stabilization, (b) weakening the inhibitory effects, (c) establishment of adequate moisture content within the digester, (d) higher OLR when compared to mono-digestion, (e) positive synergism during the digestion, (f) micronutrient and macronutrient balance, (f) enhance the economic feasibility of the process, (g) enhanced biomethane potential, and (h) improved digestibility of the individual substrate (cellulose and hemicellulose) and buffering capacity (Griffin et al. 1998; Zheng et al. 2014; Mata-Alvarez et al. 2014).
4.6 Recent Developments in Co-Digestion of Lignocellulosic Biomass and Food Wastes
In the last few years, researchers have performed extensive research on the co-digestion of lignocellulosic biomass and food wastes. In this regard, Kainthola et al. (2020) co-digested rice straw with food waste for the determination of methane yield for various C/N (i.e., 25, 30, and 35) ratios using a 1 L anaerobic digester. They have found almost similar methane yield for all the C/N ratios, i.e., 294.17 ± 3.78 L/KgVS. Besides, they have reported 71.09% more methane yield when compared to mono-digestion. However, in the same study, during the optimization of the process (i.e., pH = 7.32, C/N = 30, and F/M = 1.87), co-digestion resulted in 94.41% more methane yield when compared to mono-digestion (Kainthola et al. 2020).
Mu et al. (2020) used urban-derived food waste and co-digested it with yard waste. In this investigation, they found co-digestion a more promising alternative when compared to mono-digestion. They have found that due to co-digestion various parameters such as C/N ratio and buffering capacity have improved. The mono-digestion of yard waste resulted in a yield of 49.0 ± 5.0 mL methane/g VS, while co-digestion of yard waste and food waste resulted in 360.0 ± 30.2 mL methane/g VS (Mu et al. 2020).
David et al. (2018) co-digested three types of lignocellulosic substrates (corn stover (CS), Prairie cordgrass (PCG), and unbleached paper (UBP)) with food waste at thermophilic temperature. During this investigation, they reported that co-digestion can overcome the limitation of mono-digestion of individual substrates specifically, low buffering capacity, accumulation of VFA, and low C/N in case of FW. All lignocellulosic wastes co-digested with food wastes have shown synergetic enhancement in methane yield. However, the highest methane yield was reported for the combination of FW-PCG-CS followed by FW-PCG. A better volatile solid reduction was found in those two mixtures when compared to mono-digestion. David et al. (2018) also stated that pretreatment of lignocellulosic substrates increased the readily available sugar for the anaerobic digestion; however, it increased the cost of the overall process. David et al. (2018) also conducted their investigation without any pretreatment of the lignocellulosic substrate and emphasized the fact that during the co-digestion, consortia can play a vital role if pretreatment is not performed. They also reported that although maintaining thermophilic temperature during the digestion process will increase the costs of the overall process, it provides extra advantages of digesting the substrate at higher loading rate and at lesser retention time (David et al. 2018).
Helenas Perin et al. (2020) studied the influence of garden waste on the anaerobic digestion of food waste. In this study, they noted 86 L/d biogas production, at OLR of 0.47 L/g VS in specific methane yield when compared to mono-digestion of food waste (17 L/d biogas production at OLR 0.006 L/g VS in specific methane yield), thus indicating the possibility of optimization of the overall process (Helenas Perin et al. 2020).
Panigrahi et al. (2020) studied the co-digestion of food waste & yard waste and stated that it is an efficient technique for sustainable bioenergy generation. They reported for maximum methane production, high C/N ratio, and recalcitrant nature of yard waste are a huge bottleneck. Therefore, they thermally pretreated the yard in this study, and further, it was co-digested with the food waste to enhance nutrient balance for the overall methane production. Besides, the optimization of F/M (food/microorganism) was also performed. They reported the highest methane potential of 431 mL/gVS when F/M ratio was 1.5 (Panigrahi et al. 2020).
Shi et al. (2018) investigated the co-digestion of wheat straw and FW using five different ratios at mesophilic and thermophilic temperatures. They reported that the synergic effects improved the overall stability and performance of the process at the same (OLR = 3.0 g VS/L/d). Both the reactors of mono-digestion showed system instability. However, reactors running at thermophilic temperature have shown 4.9–14.8% higher methane yield when compared to mesophilic reactors (Shi et al. 2018).
Tayyab et al. (2019) investigated the biomethane potential of pretreated Parthenium weed and also studied its co-digestion with catering food. They set up various lab-scale digesters with different mixing ratios (0:100, 20:80, 60:40, 40:60, 80:20, and 100:0 on total solid basis) for the determination of methane yield and to study the effect of co-digestion. They observed that the reactor with 60% catering food and 40% pretreated Parthenium weed yielded maximum accumulative biogas (5532 mL/L). On the basis of their experimental study, Tayyab et al. (2019) concluded that pretreated Parthenium weed as a potential substrate if co-digested with catering food waste.
Zou et al. (2020) aimed to accelerate the hydrolysis of corn cob during anaerobic digestion with the help of FW. The authors used FW as an acidic agent for the pretreatment of the corn cob. This is attributed to the fact that during the anaerobic digestion of FW, acidification occurs which can accelerate the hydrolysis of lignocellulose. In the beginning, the optimum mixing ratio of FW, corn cob, was reported as 1:3. The hydrolysis rate was increased by 28% when compared to mono-digestion of corn cob. A reduction of 6.7% in cellulose crystallinity and 13.2% in cellulose was also achieved at this mixing ratio. However, during the stage of methane generation, the mixing ratio of food waste and corn cob reported as 1:6 has shown maximum methane production as 401.6 mL/g·VS. During the kinetic study of cellulose/hemicellulose degradation, it was found that pretreatment of corn cob with food waste improved the degradation of cellulose (Zou et al. 2020).
4.7 Conclusion
This chapter focuses on the different types of types of lignocellulosic (LW) and food wastes (FW) that can be utilized as substrates for the production of biogas through anaerobic digestion under various temperature ranges. The process of mono-digestion of both the substrates (LW and FW) has several disadvantages. Therefore, the pretreatment techniques have been recognized as an important step before the digestion process of both substrates. There is clear scientific evidence present in which pretreatment has been found to be a recognized technique in the context of improved specific biomethane potential. Nevertheless, pretreatment has not proved to be cost-effective and environmental friendly for anaerobic digestion. On the other hand, the co-anaerobic digestion of LW with FW proved to be a more promising alternative when compared to mono-anaerobic digestion of an individual for synergistic enhancements in the context to trace elements, buffering capacity, high easily biodegradable components, and C/N ratio.
References
Achinas S, Achinas V, Euverink GJW (2017) A technological overview of biogas production from biowaste. Engineering 3:299–307. https://doi.org/10.1016/J.ENG.2017.03.002
Amigun B, Sigamoney R, von Blottnitz H (2008) Commercialisation of biofuel industry in Africa: a review. Renew Sust Energ Rev 12:690–711. https://doi.org/10.1016/j.rser.2006.10.019
Appels L, Baeyens J, Degrève J, Dewil R (2008) Principles and potential of the anaerobic digestion of waste-activated sludge. Prog Energy Combust Sci 34:755–781. https://doi.org/10.1016/j.pecs.2008.06.002
Ariunbaatar J, Panico A, Esposito G et al (2014a) Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl Energy 123:143–156. https://doi.org/10.1016/j.apenergy.2014.02.035
Ariunbaatar J, Panico A, Frunzo L et al (2014b) Enhanced anaerobic digestion of food waste by thermal and ozonation pretreatment methods. J Environ Manag 146:142–149. https://doi.org/10.1016/j.jenvman.2014.07.042
Atelge MR, Krisa D, Kumar G et al (2020) Biogas production from organic waste: recent Progress and perspectives. Waste Biomass Valorization 11:1019–1040. https://doi.org/10.1007/s12649-018-00546-0
Babel S, Fukushi K, Sitanrassamee B (2004) Effect of acid speciation on solid waste liquefaction in an anaerobic acid digester. Water Res 38:2417–2423. https://doi.org/10.1016/j.watres.2004.02.005
Banks CJ, Humphreys PN (1998) The anaerobic treatment of a ligno-cellulosic substrate offering little natural pH buffering capacity. Water Sci Technol 38:29–35. https://doi.org/10.1016/S0273-1223(98)00494-6
Bong CPC, Lim LY, Lee CT et al (2018) The characterisation and treatment of food waste for improvement of biogas production during anaerobic digestion – a review. J Clean Prod 172:1545–1558. https://doi.org/10.1016/j.jclepro.2017.10.199
Boonterm M, Sunyadeth S, Dedpakdee S et al (2016) Characterization and comparison of cellulose fiber extraction from rice straw by chemical treatment and thermal steam explosion. J Clean Prod 134:592–599. https://doi.org/10.1016/j.jclepro.2015.09.084
Börjesson P, Mattiasson B (2008) Biogas as a resource-efficient vehicle fuel. Trends Biotechnol 26:7–13. https://doi.org/10.1016/j.tibtech.2007.09.007
Bouallagui H, Torrijos M, Godon JJ et al (2004) Two-phases anaerobic digestion of fruit and vegetable wastes: bioreactors performance. Biochem Eng J 21:193–197. https://doi.org/10.1016/j.bej.2004.05.001
Bryant MP (1979) Microbial methane production—theoretical aspects. J Anim Sci 48:193–201. https://doi.org/10.2527/jas1979.481193x
Chandra R, Takeuchi H, Hasegawa T (2012) Methane production from lignocellulosic agricultural crop wastes: a review in context to second generation of biofuel production. Renew Sust Energ Rev 16:1462–1476. https://doi.org/10.1016/j.rser.2011.11.035
Chen D, Gao A, Cen K et al (2018) Investigation of biomass torrefaction based on three major components: hemicellulose, cellulose, and lignin. Energy Convers Manag 169:228–237. https://doi.org/10.1016/j.enconman.2018.05.063
Choudhary A, Kumar A, Govil T et al (2020a) Sustainable production of biogas in large bioreactor under psychrophilic and mesophilic conditions. J Environ Eng 146:04019117. https://doi.org/10.1061/(ASCE)EE.1943-7870.0001645
Choudhary A, Kumar A, Kumar S (2020b) Techno-economic analysis, kinetics, global warming potential comparison and optimization of a pilot-scale unheated semi-continuous anaerobic reactor in a hilly area: for north Indian hilly states. Renew Energy 155:1181–1190. https://doi.org/10.1016/j.renene.2020.04.034
Choudhary A, Kumar A, Kumar S (2020c) National municipal solid waste energy and global warming potential inventory: India. J Hazard Toxic Radioact Waste 24:06020002. https://doi.org/10.1061/(ASCE)HZ.2153-5515.0000521
Coelho NMG, Droste RL, Kennedy KJ (2011) Evaluation of continuous mesophilic, thermophilic and temperature phased anaerobic digestion of microwaved activated sludge. Water Res 45:2822–2834. https://doi.org/10.1016/j.watres.2011.02.032
David A, Govil T, Tripathi AK et al (2018) Thermophilic anaerobic digestion: enhanced and sustainable methane production from co-digestion of food and lignocellulosic wastes. Energies 11:2058. https://doi.org/10.3390/en11082058
Dell’Omo PP, Spena VA (2020) Mechanical pretreatment of lignocellulosic biomass to improve biogas production: comparison of results for giant reed and wheat straw. Energy 203:117798. https://doi.org/10.1016/j.energy.2020.117798
Demirer GN, Chen S (2005) Two-phase anaerobic digestion of unscreened dairy manure. Process Biochem 40:3542–3549. https://doi.org/10.1016/j.procbio.2005.03.062
Deublein D, Steinhauser A (2008) History and status to date in other countries. In: Deublein D, Steinhauser A (eds) Biogas from waste and renewable resources. Wiley-VCH Verlag GmbH & Co, Mörlenbach, pp 35–42
Dolan T, Cook MB, Angus AJ (2011) Financial appraisal of wet mesophilic AD technology as a renewable energy and waste management technology. Sci Total Environ 409:2460–2466. https://doi.org/10.1016/j.scitotenv.2011.03.011
Eskicioglu C, Kennedy KJ, Droste RL (2006) Characterization of soluble organic matter of waste activated sludge before and after thermal pretreatment. Water Res 40:3725–3736. https://doi.org/10.1016/j.watres.2006.08.017
Ferdeș M, Dincă MN, Moiceanu G et al (2020) Microorganisms and enzymes used in the biological pretreatment of the substrate to enhance biogas production: a review. Sustainability 12:7205. https://doi.org/10.3390/su12177205
Fernandes TV, Klaasse Bos GJ, Zeeman G et al (2009) Effects of thermo-chemical pre-treatment on anaerobic biodegradability and hydrolysis of lignocellulosic biomass. Bioresour Technol 100:2575–2579. https://doi.org/10.1016/j.biortech.2008.12.012
Fisgativa H, Tremier A, Dabert P (2016) Characterizing the variability of food waste quality: a need for efficient valorisation through anaerobic digestion. Waste Manag 50:264–274. https://doi.org/10.1016/j.wasman.2016.01.041
Forgács G (2012) Biogas production from Citrus wastes and chicken feather: pretreatment and co-digestion
Ghosh A, Bhattacharyya BC (1999) Biomethanation of white rotted and brown rotted rice straw. Bioprocess Eng 20:297–302. https://doi.org/10.1007/s004490050594
Griffin ME, McMahon KD, Mackie RI, Raskin L (1998) Methanogenic population dynamics during start-up of anaerobic digesters treating municipal solid waste and biosolids. Biotechnol Bioeng 57:342–355. https://doi.org/10.1002/(SICI)1097-0290(19980205)57:3<342::AID-BIT11>3.0.CO;2-I
Hajji A, Rhachi M (2013) The influence of particle size on the performance of anaerobic digestion of municipal solid waste. Energy Procedia 36:515–520. https://doi.org/10.1016/j.egypro.2013.07.059
Helenas Perin JK, Biesdorf Borth PL, Torrecilhas AR et al (2020) Optimization of methane production parameters during anaerobic co-digestion of food waste and garden waste. J Clean Prod 272:123130. https://doi.org/10.1016/j.jclepro.2020.123130
Hobsen PN, Bousfield S, Summers R (1981) Methane production from agricultural and domestic wastes. Springer
Jain SR, Mattiasson B (1998) Acclimatization of methanogenic consortia for low pH biomethanation process. Biotechnol Lett 20:771–775. https://doi.org/10.1023/B:BILE.0000015920.45724.29
Jiang X, Sommer SG, Christensen KV (2011) A review of the biogas industry in China. Energy Policy 39:6073–6081. https://doi.org/10.1016/j.enpol.2011.07.007
Kabir MM, del Pilar Castillo M, Taherzadeh MJ, Horváth IS (2013) Effect of the N-Methylmorpholine-N-oxide (NMMO) pretreatment on anaerobic digestion of Forest residues. Bioresources 8:5409–5423
Kabir MM, Niklasson C, Taherzadeh MJ, Horváth IS (2014) Biogas production from lignocelluloses by N-methylmorpholine-N-oxide (NMMO) pretreatment: effects of recovery and reuse of NMMO. Bioresour Technol 161:446–450. https://doi.org/10.1016/j.biortech.2014.03.107
Kabir MM, Forgács G, Sárvári Horváth I (2015) Biogas from lignocellulosic materials. In: Karimi K (ed) Lignocellulose-based bioproducts. Springer International Publishing, Cham, pp 207–251
Kainthola J, Kalamdhad AS, Goud VV (2019a) A review on enhanced biogas production from anaerobic digestion of lignocellulosic biomass by different enhancement techniques. Process Biochem 84:81–90. https://doi.org/10.1016/j.procbio.2019.05.023
Kainthola J, Kalamdhad AS, Goud VV, Goel R (2019b) Fungal pretreatment and associated kinetics of rice straw hydrolysis to accelerate methane yield from anaerobic digestion. Bioresour Technol 286:121368. https://doi.org/10.1016/j.biortech.2019.121368
Kainthola J, Kalamdhad AS, Goud VV (2020) Optimization of process parameters for accelerated methane yield from anaerobic co-digestion of rice straw and food waste. Renew Energy 149:1352–1359. https://doi.org/10.1016/j.renene.2019.10.124
Kanokwan B (2006) Online monitoring and control of the biogas process. Technical University of Denmark
Kashyap DR, Dadhich KS, Sharma SK (2003) Biomethanation under psychrophilic conditions: a review. Bioresour Technol 87:147–153. https://doi.org/10.1016/S0960-8524(02)00205-5
Kim IS, Hwang MH, Jang NJ et al (2004) Effect of low pH on the activity of hydrogen utilizing methanogen in bio-hydrogen process. Int J Hydrog Energy 29:1133–1140. https://doi.org/10.1016/j.ijhydene.2003.08.017
Koniuszewska I, Korzeniewska E, Harnisz M, Czatzkowska M (2020) Intensification of biogas production using various technologies: a review. Int J Energy Res 44:6240–6258. https://doi.org/10.1002/er.5338
Lehtomäki A, Huttunen S, Rintala JA (2007) Laboratory investigations on co-digestion of energy crops and crop residues with cow manure for methane production: effect of crop to manure ratio. Resour Conserv Recycl 51:591–609. https://doi.org/10.1016/j.resconrec.2006.11.004
Leung DYC, Wang J (2016) An overview on biogas generation from anaerobic digestion of food waste. Int J Green Energy 13:119–131. https://doi.org/10.1080/15435075.2014.909355
Li W, Zheng P, Guo J et al (2014) Characteristics of self-alkalization in high-rate denitrifying automatic circulation (DAC) reactor fed with methanol and sodium acetate. Bioresour Technol 154:44–50. https://doi.org/10.1016/j.biortech.2013.11.097
Li Y, Jin Y, Borrion A et al (2017) Effects of organic composition on mesophilic anaerobic digestion of food waste. Bioresour Technol 244:213–224. https://doi.org/10.1016/j.biortech.2017.07.006
Li W, Loh K-C, Zhang J et al (2018) Two-stage anaerobic digestion of food waste and horticultural waste in high-solid system. Appl Energy 209:400–408. https://doi.org/10.1016/j.apenergy.2017.05.042
Lindmark J, Lagerkvist A, Nilsson E et al (2014) Evaluating the effects of electroporation pre-treatment on the biogas yield from ley crop silage. Appl Biochem Biotechnol 174:2616–2625. https://doi.org/10.1007/s12010-014-1213-7
Liu Y, Fang J, Tong X et al (2019) Change to biogas production in solid-state anaerobic digestion using rice straw as substrates at different temperatures. Bioresour Technol 293:122066. https://doi.org/10.1016/j.biortech.2019.122066
Ma H, Liu W-W, Chen X et al (2009) Enhanced enzymatic saccharification of rice straw by microwave pretreatment. Bioresour Technol 100:1279–1284. https://doi.org/10.1016/j.biortech.2008.08.045
Ma C, Liu J, Ye M et al (2018) Towards utmost bioenergy conversion efficiency of food waste: pretreatment, co-digestion, and reactor type. Renew Sust Energ Rev 90:700–709. https://doi.org/10.1016/j.rser.2018.03.110
Mara D, Horan NJ (2003) Handbook of water and wastewater microbiology. Elsevier
Mata-Alvarez J, Macé S, Llabrés P (2000) Anaerobic digestion of organic solid wastes. An overview of research achievements and perspectives. Bioresour Technol 74:3–16. https://doi.org/10.1016/S0960-8524(00)00023-7
Mata-Alvarez J, Dosta J, Macé S, Astals S (2011) Codigestion of solid wastes: a review of its uses and perspectives including modeling. Crit Rev Biotechnol 31:99–111. https://doi.org/10.3109/07388551.2010.525496
Mata-Alvarez J, Dosta J, Romero-Güiza MS et al (2014) A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew Sust Energ Rev 36:412–427. https://doi.org/10.1016/j.rser.2014.04.039
Meng Y, Li S, Yuan H et al (2015) Evaluating biomethane production from anaerobic mono- and co-digestion of food waste and floatable oil (FO) skimmed from food waste. Bioresour Technol 185:7–13. https://doi.org/10.1016/j.biortech.2015.02.036
Mittal K (1996) Biogas systems: principles and applications. New Age International Limited Publishers, New Delhi
Mu L, Zhang L, Zhu K et al (2020) Anaerobic co-digestion of sewage sludge, food waste and yard waste: synergistic enhancement on process stability and biogas production. Sci Total Environ 704:135429. https://doi.org/10.1016/j.scitotenv.2019.135429
Naik SN, Goud VV, Rout PK, Dalai AK (2010) Production of first and second generation biofuels: a comprehensive review. Renew Sust Energ Rev 14:578–597. https://doi.org/10.1016/j.rser.2009.10.003
Nasr N, Elbeshbishy E, Hafez H et al (2012) Comparative assessment of single-stage and two-stage anaerobic digestion for the treatment of thin stillage. Bioresour Technol 111:122–126. https://doi.org/10.1016/j.biortech.2012.02.019
Nghiem LD, Koch K, Bolzonella D, Drewes JE (2017) Full scale co-digestion of wastewater sludge and food waste: bottlenecks and possibilities. Renew Sust Energ Rev 72:354–362. https://doi.org/10.1016/j.rser.2017.01.062
Palmqvist E, Hahn-Hägerdal B (2000) Fermentation of lignocellulosic hydrolysates. II: inhibitors and mechanisms of inhibition. Bioresour Technol 74:25–33. https://doi.org/10.1016/S0960-8524(99)00161-3
Panigrahi S, Sharma HB, Dubey BK (2020) Anaerobic co-digestion of food waste with pretreated yard waste: a comparative study of methane production, kinetic modeling and energy balance. J Clean Prod 243:118480. https://doi.org/10.1016/j.jclepro.2019.118480
Phuttaro C, Sawatdeenarunat C, Surendra KC et al (2019) Anaerobic digestion of hydrothermally-pretreated lignocellulosic biomass: influence of pretreatment temperatures, inhibitors and soluble organics on methane yield. Bioresour Technol 284:128–138. https://doi.org/10.1016/j.biortech.2019.03.114
Pramanik SK, Suja FB, Zain SM, Pramanik BK (2019) The anaerobic digestion process of biogas production from food waste: prospects and constraints. Bioresour Technol Rep 8:100310. https://doi.org/10.1016/j.biteb.2019.100310
Putro JN, Soetaredjo FE, Lin S-Y et al (2016) Pretreatment and conversion of lignocellulose biomass into valuable chemicals. RSC Adv 6:46834–46852. https://doi.org/10.1039/C6RA09851G
Rajendran K, Aslanzadeh S, Taherzadeh MJ (2012) Household biogas digesters—a review. Energies 5:2911–2942. https://doi.org/10.3390/en5082911
Resch C, Wörl A, Waltenberger R et al (2011) Enhancement options for the utilisation of nitrogen rich animal by-products in anaerobic digestion. Bioresour Technol 102:2503–2510. https://doi.org/10.1016/j.biortech.2010.11.044
Schink B (1997) Energetics of syntrophic cooperation in methanogenic degradation. Microbiol Mol Biol Rev 61:262–280
Shahriari H, Warith M, Hamoda M, Kennedy K (2013) Evaluation of single vs. staged mesophilic anaerobic digestion of kitchen waste with and without microwave pretreatment. J Environ Manag 125:74–84. https://doi.org/10.1016/j.jenvman.2013.03.042
Shi X, Guo X, Zuo J et al (2018) A comparative study of thermophilic and mesophilic anaerobic co-digestion of food waste and wheat straw: process stability and microbial community structure shifts. Waste Manag 75:261–269. https://doi.org/10.1016/j.wasman.2018.02.004
Sitorus B, Sukandar, Panjaitan SD (2013) Biogas recovery from anaerobic digestion process of mixed fruit -vegetable wastes. Energy Procedia 32:176–182. https://doi.org/10.1016/j.egypro.2013.05.023
Speece RE (2008) Anaerobic biotechnology for industrial wastewater treatment. https://pubs.acs.org/doi/pdf/10.1021/es00115a001. Accessed 23 Sept 2020
Srisowmeya G, Chakravarthy M, Nandhini Devi G (2020) Critical considerations in two-stage anaerobic digestion of food waste—a review. Renew Sust Energ Rev 119:109587. https://doi.org/10.1016/j.rser.2019.109587
Sun Y, Cheng J (2002) Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour Technol 83:1–11. https://doi.org/10.1016/S0960-8524(01)00212-7
Taconi KA, Zappi ME, Todd French W, Brown LR (2008) Methanogenesis under acidic pH conditions in a semi-continuous reactor system. Bioresour Technol 99:8075–8081. https://doi.org/10.1016/j.biortech.2008.03.068
Taherzadeh MJ, Karimi K (2008) Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: a review. Int J Mol Sci 9:1621–1651. https://doi.org/10.3390/ijms9091621
Tampio E, Marttinen S, Rintala J (2016) Liquid fertilizer products from anaerobic digestion of food waste: mass, nutrient and energy balance of four digestate liquid treatment systems. J Clean Prod 125:22–32. https://doi.org/10.1016/j.jclepro.2016.03.127
Tayyab A, Ahmad Z, Mahmood T et al (2019) Anaerobic co-digestion of catering food waste utilizing Parthenium hysterophorus as co-substrate for biogas production. Biomass Bioenergy 124:74–82. https://doi.org/10.1016/j.biombioe.2019.03.013
Vavilin VA, Fernandez B, Palatsi J, Flotats X (2008) Hydrolysis kinetics in anaerobic degradation of particulate organic material: an overview. Waste Manag 28:939–951. https://doi.org/10.1016/j.wasman.2007.03.028
Vivekanand V, Ryden P, Horn SJ et al (2012) Impact of steam explosion on biogas production from rape straw in relation to changes in chemical composition. Bioresour Technol 123:608–615. https://doi.org/10.1016/j.biortech.2012.06.088
Wang L-H, Wang Q, Cai W, Sun X (2012) Influence of mixing proportion on the solid-state anaerobic co-digestion of distiller’s grains and food waste. Biosyst Eng 112:130–137. https://doi.org/10.1016/j.biosystemseng.2012.03.006
Wang L, Shen F, Yuan H et al (2014) Anaerobic co-digestion of kitchen waste and fruit/vegetable waste: lab-scale and pilot-scale studies. Waste Manag 34:2627–2633. https://doi.org/10.1016/j.wasman.2014.08.005
Ward AJ, Hobbs PJ, Holliman PJ, Jones DL (2008) Optimisation of the anaerobic digestion of agricultural resources. Bioresour Technol 99:7928–7940. https://doi.org/10.1016/j.biortech.2008.02.044
Wiegel J, Mesbah NM, Abou-El-Ela SH (2007) Life at extreme limits: 2005:275–275. https://doi.org/10.11490/isea.2005.0.275.0
Xu F, Li Y, Ge X et al (2018) Anaerobic digestion of food waste – challenges and opportunities. Bioresour Technol 247:1047–1058. https://doi.org/10.1016/j.biortech.2017.09.020
Yong Z, Dong Y, Zhang X, Tan T (2015) Anaerobic co-digestion of food waste and straw for biogas production. Renew Energy 78:527–530. https://doi.org/10.1016/j.renene.2015.01.033
You Z, Pan S-Y, Sun N et al (2019) Enhanced corn-stover fermentation for biogasproduction by NaOH pretreatment with CaO additive and ultrasound. J Clean Prod 238:117813. https://doi.org/10.1016/j.jclepro.2019.117813
Zhang Y, Banks CJ (2013) Impact of different particle size distributions on anaerobic digestion of the organic fraction of municipal solid waste. Waste Manag 33:297–307. https://doi.org/10.1016/j.wasman.2012.09.024
Zhang C, Su H, Baeyens J, Tan T (2014) Reviewing the anaerobic digestion of food waste for biogas production. Renew Sust Energ Rev 38:383–392. https://doi.org/10.1016/j.rser.2014.05.038
Zheng Y, Zhao J, Xu F, Li Y (2014) Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog Energy Combust Sci 42:35–53. https://doi.org/10.1016/j.pecs.2014.01.001
Zhou J, Zhang R, Liu F et al (2016) Biogas production and microbial community shift through neutral pH control during the anaerobic digestion of pig manure. Bioresour Technol 217:44–49. https://doi.org/10.1016/j.biortech.2016.02.077
Zhu S, Wu Y, Yu Z et al (2005) Pretreatment by microwave/alkali of rice straw and its enzymic hydrolysis. Process Biochem 40:3082–3086. https://doi.org/10.1016/j.procbio.2005.03.016
Zou H, Jiang Q, Zhu R et al (2020) Enhanced hydrolysis of lignocellulose in corn cob by using food waste pretreatment to improve anaerobic digestion performance. J Environ Manag 254:109830. https://doi.org/10.1016/j.jenvman.2019.109830
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Choudhary, A., Kumar, A., Kumar, S. (2022). Co-Digestion of Lignocellulosic Wastes with Food Waste for Sustainable Biogas Production. In: Saini, J.K., Sani, R.K. (eds) Microbial Biotechnology for Renewable and Sustainable Energy. Clean Energy Production Technologies. Springer, Singapore. https://doi.org/10.1007/978-981-16-3852-7_4
Download citation
DOI: https://doi.org/10.1007/978-981-16-3852-7_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-3851-0
Online ISBN: 978-981-16-3852-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)