Abstract
Retinopathy of prematurity (ROP) and familial exudative vitreoretinopathy (FEVR) can have very similar clinical presentations. Both diseases have abnormal development of retinal vessels and lead to severe vitreoretinopathy which causes blindness in newborn infants. The single most important difference is prematurity. In ROP, the most important risk factors are gestational age and low birth weight. In FEVR, it is the genetic mutation. Identifying the underlying mutations in the causative gene can predict the prognosis of patients with FEVR. ROP tends to resolve naturally or with treatment, but FEVR is a lifelong disease. Even we know that the clinical characteristics and risk factors between both diseases are different; the clinical similarity makes differential diagnosis difficult, especially in FEVR patients who were born prematurely. In such a scenario, patients could exhibit features of FEVR or ROP or both and found to have a discrepancy between birth history and fundus appearance, thus ROPER/fROP was used to describe these patients under such conditions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Familial exudative vitreoretinopathy (FEVR)
- FZD4
- LRP5
- Retinopathy of prematurity (ROP)
- ROPER
- TSPAN12
- Win pathway
- ZNF408
1 Introduction
In regards to eye development, a particular Wnt pathway, Norrin-FZD4, has been identified as playing a major role in retinal angiogenesis. Signaling through this pathway is necessary for the development and maintenance of retinal vasculature [1,2,3,4]. Mutations affecting genes of this pathway can result in several pediatric vitreoretinopathies, such as Norrie disease (ND), familial exudative vitreoretinopathy (FEVR), and pseudo-glioma and osteoporosis syndrome [5,6,7,8]. Wnt pathway mutations also have been reported in Coats disease and persistent fetal vasculature (PFV) [9,10,11,12].
Retinopathy of prematurity (ROP) seldom reports to have an association of genetic mutation. However, some studies have identified Wnt pathway mutations such as FZD4, LRP5, and TSPAN12 in patients with advanced ROP [13,14,15] Mutations in both the NDP and FZD4 genes have been reported in ROP cases and make Wnt pathway mutations a candidate for developing severe ROP [14]. However, most ROP patients do not show any genetic mutation, in contrast, as high as 50% of FEVR patients are associated with known genetic mutations of the Wnt pathway [16, 17].
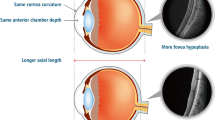
FEVR occurs in an X-linked (NDP), autosomal recessive (LRP5), or autosomal dominant inheritance pattern (FZD4, TSPAN12, ZNF408). Most reported cases of autosomal dominant FEVR have been associated with mutations in the FZD4 gene [18]. ROP and FEVR have overlapping phenotypes, and premature birth history makes it difficult to differentiate between both diseases. In the past, ROP is distinguished from FEVR by premature birth and the lack of a family history of retinal diseases (Table 7.1). But as advances in infertility technology and neonatal care improve, so does the survival rate of newborns. Therefore, more cases of premature infants with features of FEVR are recognized. Testing for FEVR genetic mutations is useful in patients with suspected FEVR and ROP. Studies have shown that the [P33S(;)P168S] FZD4 variant is associated with FEVR and ROP [18, 19].
Genetic testing may be beneficial to screen premature infants when we suspect a FEVR mutation [18]. Clinically, identifying the underlying mutations in the causative gene can help us manage the patient and predict the prognosis of the disease. Patients with positive gene mutation tend to have more severe phenotypes that progress and develop recurring retinal detachment. In ROP, acute progression to retinal detachment should be monitored more strictly and the suspicion of ROPER/fROP should be entertained [19,20,21].
This chapter focuses on FEVR patients who are born prematurely and exhibit features of FEVR, ROP, or both. Discrepancies between birth, family history, and fundus appearance should alert to the possible diagnosis of FEVR (Table 7.1 and Fig. 7.1). In such a scenario, ROPER/fROP was postulated to be used to refer to these patients.
Representative fundus photos of a male infant diagnosed with FEVR but born prematurely at GA 26 weeks, BW 900 g. Photo was taken at PMA 41 weeks, both eyes showing exudation, tractional retinal detachment, vascular anomalies posteriorly, and radial temporal retinal fold in the left eye, which resembled typical features of FEVR
Also staging in both diseases was compared in Table 7.2 [22].
From characteristic features, FEVR can be distinguished from ROP by:
-
Familial pattern
-
Progression after birth in childhood or adulthood
-
Absence of severe prematurity (mostly older than 28 weeks)
-
Absence of supplementary oxygenation at birth
-
Presence of more exudation and trend to reactivate or recur after treatment
-
Asymmetric disease
With ROP:
-
Mostly occurs as an acute disease after birth; stable after the acute episode.
-
Premature with low birth weight/young gestational age.
-
The disease follows a predictable chronological time course.
-
Usually does not progress or recur in childhood.
-
The cumulative exposure to supplemental oxygen.
-
Usually symmetrical disease.
In premature infants who are not at risk for ROP, it is easier to identify features associated to FEVR rather than ROP. If the premature infant is at risk of ROP, many of the clinical features associated with FEVR may overlap with those commonly found in ROP, making a definite diagnosis of FEVR challenging (Figs. 7.2 and 7.3).
Representative photos of a male infant diagnosed with fROP/ROPER born prematurely at GA 29 weeks, BW 1330 g. Color fundus was taken at PMA 47 weeks. The right eye demonstrated stage 3 ROP. The color fundus showed typical ridge tissue, retinal hemorrhage, and active neovascularization. Fluorescein angiogram demonstrated classic homogenous vascularization pattern at ridge tissue and focal leakage of neovascularization, which are more characteristic of ROP
The same patient as Fig. 7.2: The left eye showed anomalous retinal vasculature posteriorly and the anomalous circumferential peripheral vessels, as well as the asymmetry between both eyes, making it more like FEVR. So, this case looks like a hybrid for both ROP and FEVR
The management and follow-up of the two disorders are different, and both may lead to severe vitreoretinopathy with tractional detachments and potential blindness. Positive genetic testing results may support the diagnosis of FEVR, but it is important to remember that a negative result or negative family history does not rule out the diagnosis [23, 24] (Table 7.3). In FEVR, 50% of the cases have no known mutation and a negative family history can simply signify a new mutation. Also, the follow up of these patients is quite different. Patients with ROP require very close monitoring, especially at a certain gestational age. FEVR is a lifelong disease that requires lifelong management. ROP is a disease that resolves naturally or with treatment. The management for fROP/ROPER is retinal photocoagulation to the avascular area as is in ROP. Anti-VEGF can be considered as an adjuvant to laser especially in cases when there is generalized leakage in the retinal vessels. Monitoring these cases with wide-field fluorescein angiography (WFA) is essential to diagnose early recurrences or progression of the disease to avoid tractional retinal detachments. If detachment occurs, scleral buckles alone or in combination with vitrectomies may be needed.
In one of the author’s centers, Queen Sirikit National Institute of Child Health (QSNICH), Bangkok, 125 cases of FEVR were reported during 2013–2017. Twenty-four patients (20%) were born prematurely, and angiographic testing showed characteristics of FEVR-like changes rather than ROP. In premature infants with FEVR, 9/24 cases (37.5%) were at risk of ROP (GA </= 28 weeks or BW </=1500 g) whose mean GA was 27 weeks and mean birth weight 1460 g; 15/24 cases (62.5%) were not at risk of ROP whose mean GA was 37 weeks and mean birth weight 2300 g. Berrocal et al. had reported 9 FEVR cases who were born prematurely with a mean GA of 30 weeks and mean birth weight 1226 g and showed these cases to be FEVR in the presence of prematurity. These studies aimed to show a better classification of these cases and the proposed name of ROPER (ROP vs. FEVR) for premature infants who behaved like FEVR cases [25, 26]. Drenser et al. termed this disease “fROP.” This subset of fROP/ROPER will have the risks of continual disease activity and will alter the treatment, follow-up, and family counseling [12].
2 Widefield Fluorescein Angiography in ROP, FEVR, and fROP/ROPER
Imaging is important for the management of ROP and FEVR. The diagnosis of both FEVR and ROP are based on ophthalmoscopy or color photography findings, and WFA helps in identifying the early or flat neovascularization and more accurately identify the borders between vascular and avascular retina. WFA also allows for early identification of vascular changes that are not seen by ophthalmoscopy or color photography [27,28,29]. WFA is the best diagnostic tool particularly in eyes with atypical fundus findings in premature infants suspected of having ROPER. It provides additional information by identifying angiographic characteristics of ROP versus FEVR-like syndrome or may contain features of both [29]. WFA is also useful in monitoring disease progression, regression, and treatment planning in pediatric vitreoretinopathies such as ROP and FEVR.
Characteristics of WFA in ROP [27, 28]:
-
Vascular tuft formation
-
Focal capillary dilatation
-
Neovascularization and leakage
-
Classic homogenous vascularization pattern at the ridge
-
Arteriovenous shunting
Characteristics of WFA in FEVR [22, 23]:
-
More vascular branching
-
Venous–venous shunting
-
Anomalous circumferential peripheral vessels
-
Peripheral avascular abnormalities
-
Supernumerous vascular abnormalities at the junction
-
Central or peripheral telangiectasias
-
Late phase disc leakage
Characteristic of WFA in fROP/ROPER [25, 26]:
-
Contain features of both ROP and FEVR
-
Irregular sprouts vascularization at the junction
-
Distinct pruning of vessels
-
Pinpoints areas of hyperfluorescence
-
Segmental areas of vascular leakage
3 Conclusion
Both ROP and FEVR can exhibit dragged disc, ectopic macular, and pseudostrabismus due to fibrovascular tractional lesions from active avascular areas. If patients were born prematurely less than 37 weeks and present with vascular reactivity and demonstrate fresh exudation, leaky vascular areas, or asymmetric retinal findings, then FEVR is the more likely diagnosis. ROP is a disease of prematurity and resolves with time. The fundus features of adults with ROP complications are secondary to persistent avascular area, lattice degeneration, rhegmatogenous components at the edge of treated versus untreated retina, or tractional retinal detachment from vitreous contraction. Patients with fROP/ROPER will continue to progress throughout their lifetime with exudation, neovascularization, vitreous hemorrhage, Coats-like response, tractional retinal detachment, and even vasoproliferative tumor.
When not certain about the diagnosis of fROP/ROPER in premature infants who demonstrate more characteristics of FEVR than ROP in acute diseases, we recommend performing fluorescein angiography and genetic testing. This allows for a better understanding of the clinical presentation of the patient. A definite diagnosis will lead to better management of the patient and the prevention of visual compromise. fROP/ROPER tends to pose a continuing risk of progression and reactivation which is unlike classic ROP that is mostly stable after the resolution of active disease. The best advice is that if it is not behaving like ROP, think of FEVR.
References
Wang Y, Rattner A, Zhou Y, et al. Norrin/frizzled 4 signaling in retinal vascular development and blood brain barrier plasticity. Cell. 2012;151:1332–44.
Braunger BM, Tamm ER. The different functions of norrin. Adv Exp Med Biol. 2012;723:679–83.
Ohlmann A, Tamm ER. Norrin: molecular and functional properties of an angiogenic and neuroprotective growth factor. Prog Retin Eye Res. 2012;31:243–57.
Ye X, Wang Y, Nathans J. The norrin/Frizzled4 signaling pathway in retinal vascular development and disease. Trends Mol Med. 2010;16:417–25.
Sims KB. NDP-related retinopathies. In: Pagon RA, Adam MP, Ardinger HH, et al., editors. Gene reviews(R). Seattle, WA: University of Washington; 1993.
Chamney S, McLoone E, Willoughby CE. A mutation in the Norrie disease gene (NDP) associated with familial exudative vitreoretinopathy. Eye (Lond). 2011;25:1658.
Chen ZY, Battinelli EM, Fielder A, et al. A mutation in the norrie disease gene (NDP) associated with X-linked familial exudative vitreoretinopathy. Nat Genet. 1993;5:180–3.
Dickinson JL, Sale MM, Passmore A, et al. Mutations in the NDP gene: contribution to norrie disease, familial exudative vitreoretinopathy and retinopathy of prematurity. Clin Experiment Ophthalmol. 2006;34:682–8.
Robitaille JM, Wallace K, Zheng B, et al. Phenotypic overlap of familial exudative vitreoretinopathy (FEVR) with persistent fetal vasculature (PFV) caused by FZD4 mutations in two distinct pedigrees. Ophthalmic Genet. 2009;30:23.
Black GC, Perveen R, Bonshek R, et al. Coats’ disease of the retina (unilateral retinal telangiectasis) caused by somatic mutation in the NDP gene: a role for norrin in retinal angio- genesis. Hum Mol Genet. 1999;8:2031–5.
Robitaille JM, Zheng B, Wallace K, et al. The role of frizzled- 4 mutations in familial exudative. Br J Ophthalmol. 95(4):574–9.
Drenser KA, Dailey W, Vinekar A, et al. Clinical presentation and genetic correlation of patients with mutations affecting the FZD4 gene. Arch Ophthalmol. 2009;127:1649–54.
Kondo H, Kusaka S, Yoshinaga A, et al. Genetic variants of FZD4 and LRP5 genes in patients with advanced retinopathy of prematurity. Mol Vis. 2013;19:476–85.
Wu WC, Drenser K, Trese M, et al. Retinal phenotype- genotype correlation of pediatric patients expressing muta- tions in the Norrie disease gene. Arch Ophthalmol. 2007;125:225–30.
Hiraoka M, Takahashi H, Orimo H, et al. Genetic screening of Wnt signaling factors in advanced retinopathy of prematurity. Mol Vis. 2010;16:2572–7.
Hiroyuki K. Complex genetics of familial exudative vitreoretinopathy and related paediatric retinal detachments. Taiwan J of Ophthalmol. 2015;5:56–62.
Mukai S, et al. The Wnt signaling pathway in familial exudative Vitreoretinopathy and Norrie diseases. Sem in Ophthalmology. 2007;22:211–7.
Dailey WA, Gryc W, Garg PG, Drenser KA. Frizzled-4 variations associated with retinopathy and intrauterine growth retardation: a potential marker for prematurity and retinopathy. Ophthalmology. 2015;122:1917–23.
MacDonald ML, Goldberg YP, Macfarlane J, Samuels ME, Trese MT, Shastry BS. Genetic variants of frizzled-4 gene in familial exudative vitreoretinopathy and advanced retinopathy of prematurity. Clin GenetClin Genet. 2005;67:363e366.
Ells A, Guernsey DL, Wallace K, et al. Severe retinopathy of prematurity associated with FZD4 mutations. Ophthalmic Genet. 2010;31:37e43.
Drenser KA, Dailey W, Vinekar A, Dalal K, Capone A Jr, Trese MT. Clinical presentation and genetic correlation of patients with mutations affecting the FZD4 gene. Arch Ophthalmol. 2009;127:1649e1654.
Kashani AH, Brown KT, Chang E, et al. Diversity of retinal vascular anomalies in patients with familial exudative vitreoretinopathy. Ophthalmology. 2014;121:2220–7.
Ranchod TM, Ho LY, Drenser KA, Capone A Jr, Trese MT. Clinical presentation of familial exudative vitreoretinopathy. Ophthalmology. 2011;118(10):2070–5.
Liche F, Majji AB. Familial exudative vitreoretinopathy. Ophthalmology. May 2012;119(5):1093.
Gologorsky D, Chang JS, Hess DJ, Berrocal AM. Familial exudative vitreoretinopathy in a premature child. Ophthalmic Surg Lasers Imaging Retina. 2013;44:603–5.
John VJ, Mc Clintic JI, Hess DJ, Berrocal AM. Retinopathy of prematurity versus familial exudative vitreoretinopathy: report on clinical and angiographic findings. Ophthalmic Surg Lasers Imaging Retina. 2016;47:14–9.
Zepeda-Romero LC, Oregon-Miranda AA, Lizarraga-Barrón DS, et al. Early retinopathy of prematurity findings identified with fluorescein angiography. Graefes Arch Clin Exp Ophthalmol. 2013;251:2093–7.
Azad R, Chandra P, Khan MA, Darswal A. Role of intravenous fluorescein angiography in early detection and regression of retinopathy of prematurity. J Pediatr Ophthalmol Strabismus. 2008;45:36–9.
Ng EYJ, Lanigan B, O’Keefe M. Fundus fluorescein angiography in the screening for and management of retinopathy of prematurity. J Pediatr Ophthalmol Strabismus. 2006;43:85–90.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Amphornphruet, A., Berrocal, A.M. (2021). Familial Exudative Vitreoretinopathy or Retinopathy of Prematurity. In: Wu, WC., Lam, WC. (eds) A Quick Guide to Pediatric Retina. Springer, Singapore. https://doi.org/10.1007/978-981-15-6552-6_7
Download citation
DOI: https://doi.org/10.1007/978-981-15-6552-6_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-6551-9
Online ISBN: 978-981-15-6552-6
eBook Packages: MedicineMedicine (R0)