Abstract
The critical stages during the growth of crops are the uniform seed germination, early seedling growth, and uniform plant stand. Low crop yield is attributed to uneven seed germination and seedling growth. Therefore, the quality of seed can be improved through priming in addition to the field management techniques for better seed germination. Priming is a physiological technique of seed hydration and drying to enhance the pregerminative metabolic process for rapid germination, seedling growth, and final yield under normal as well as stressed conditions. The primed seeds show faster and uniform seed germination due to different enzyme activation, metabolic activities, biochemical process of cell repair, protein synthesis, and improvement of the antioxidant defense system as compared to unprimed seeds. There are many techniques of seed priming which are broadly divided into conventional methods (hydro-priming, osmo-priming, nutrient priming, chemical priming, bio-priming, and priming with plant growth regulators) and advanced methods (nano-priming and priming with physical agents). However, priming is strongly affected by various factors such as temperature, aeration, light, priming duration, and seed characteristics. This chapter highlights the priming mechanism and the available technologies as a tool for superficial seed germination and crop stand. An experiment with reference to the importance of priming toward vigor seed germination and seedling growth was conducted, and its results have been added in this chapter.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Seed treatment before sowing is the foundation for activation of seed resources that in combination with external ingredients could contribute to the efficient plant growth and high yield. Various physiological and non-physiological techniques are available for enhancing seed performance as well as to combat environmental constraints. The physiological treatments for improving seed germination and stand establishment are composed of seed hydration techniques such as humidification, wetting, and presoaking. The other techniques for promoting germination are comprised of chemical treatments, seed inoculation with beneficial microbes, and seed coating. Seed priming is a physiological method of controlled hydration and drying to enhance sufficient pre-germinative metabolic process for rapid germination (Dawood 2018). This is one of the economic and feasible technologies for uniform seed development in most of the field crops. Other benefits include efficient nutrients uptake, water use efficiency, release photo- and thermo-dormancy, maturity, and crop yield (Hill et al. 2008; Bagheri 2014; Lara et al. 2014; Dutta 2018). However, many factors affect the performance of seed priming such as plant species, priming duration, temperature, priming media, and their concentration and storage conditions. The treated seed with a proper reagent can germinate better, for instance, seed treatment with inorganic salts (KCl and KNO3) enhanced the germination and growth rates. The KCl improved the starch and protein contents, whereas KNO3 increased the fruit size and quality (Singh et al. 2015a, b).
The other priming reagents involved in breaking seed dormancy are gibberellic acid (GA) and cytokinins (Assefa et al. 2010). Priming induces a set of biochemical changes such as enzyme activation, metabolism of germination inhibitors, repair of cell damages, and imbibition to promote germination (Farooq et al. 2010). Priming also modifies biochemical and physiological nature of embryo and affects the release of substances during germination phase II that activates the production of hydrolytic enzymes. These substances make high-energy compounds and essential chemicals for the germinating seedlings available (Renugadevi and Vijayageetha 2006). Therefore, the positive effects of seed priming are highly attributed to various biochemical phenomena such as improvement of the antioxidant defense system and restoration of metabolic activities through the synthesis of proteins and nucleic acids (RNA and DNA) (Di Girolamo and Barbanti 2012).
2 History of Seed Priming
Various seed treatment techniques are introduced and examined for uniform germination under different environmental conditions. Evenari (1984) reported that the efforts for improving seed germination and growth are dated back to ancient Greeks. Theophrastus (371–287 B.C.), during an investigation, observed that cucumber seeds when soaked in water result in faster and uniform germination as compared to unprimed seeds (Theophrastus, Enquiry into Plants, Book VII, I.6). Likewise, the Roman naturalist Gaius Plinius Secundus (23–79 A.D.) in his Encyclopedia reported the positive effects of presoaking of cucumber seeds in honey and water for seed germination (Gaius 1949–1954). Afterward, in 1539–1619, the French botanist Oliver de Serres reported about the seed soaked in manure water for 2 days and then dried before sowing as an effective way of seed treatment for better crop growth. Charles Darwin tested osmo-priming on lettuce and cress seeds in seawater and observed high germination in the treated seeds as compared to nonprimed seed (Darwin 1855). The modern concept of seed priming is presented by Ells (1963), who highlighted the critical parameters related to seed treatment. He observed high germination rates when seeds were treated with a specific nutrient solution. Koehler (1967) reported that treatment with salt solution promotes RNA accumulation that, in turn, enhances other physiological process and results in high seed emergence. May et al. (1962) stated that seed drying for certain time at specific level after priming exerts beneficial effect and leads to fast germination under normal as well as stressful conditions (Berrie and Drennan 1971). Heydecker et al. (1973) used organic chemical polyethylene glycol (PEG) H–(O–CH2–CH2)n–OH, a high molecular weight compound, for seed pretreatment to boost germination and avoid several problems associated with salts treatment like hardening. The priming technology, so far in research and development, has been adopted as a novel technique for getting a uniform crop standby several seed and agricultural companies.
3 Phenomenon of Seed Priming
After sowing, seeds remain in the soil for a certain period to absorb water and some essential nutrients for their growth. Seed priming is a technique to reduce this time and makes the germination quickly and uniformly. In addition to hydration, priming also reduces the sensitivity of seed to external environmental factors (Afzal et al. 2016). Priming promotes seed germination under three stages such as imbibition, germination, and growth (Fig. 1). During the imbibition stage, the water uptake promotes protein synthesis and respiratory activities through messenger ribonucleic acid (mRNA). The second stage is related to the initiation of different physiological activities related to germination such as protein synthesis, mitochondria synthesis, and alteration in soluble sugars (Varier et al. 2010). The critical factor during seed priming is the controlled water uptake during the second stage, before the emergence and growth of radical from the seed coat during the last stage. The second stage (germination) is much sensitive to environmental factors than the third stage (Côme and Thévenot 1982). Therefore, during priming, the seeds that have passed through the second stage could germinate under variant environmental conditions as compared to unprimed seeds (Corbineau and Côme 2006).
4 Methods of Seed Priming
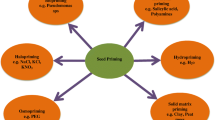
There are several techniques of seed priming that are broadly divided into conventional and advanced methods. The traditional techniques are comprised of hydro-priming, osmo-priming, nutrient priming, chemical priming, bio-priming, seed priming with plant growth regulators, and priming with plant extracts, whereas the advanced techniques of seed priming include seed priming through nanoparticles and priming through physical agents (Table 1). These techniques are described below.
4.1 Conventional Seed Priming Methods
4.1.1 Hydro-priming
Hydro-priming is a simple and economical technique in which seeds are soaked in water for a specific period and dried to a certain moisture level before sowing (Singh et al. 2015a, b). This technology is useful in areas with adverse environmental conditions including high heat and drought stress. Hydro-priming improves the water uptake efficiency and seed hydration under such conditions (McDonald 2000). However, it is critical to maintaining optimum humidity and temperature to avoid radical projection, as hydro-priming may result in noncontrolled water uptake (Taylor et al. 1998). The key technique of hydro-priming is “drum priming” invented by Rowse (1991). Drum priming is comprised of a drum containing seed lot connected to a boiler producing vapors. The vapors upon entering the drum condense into water droplets. During treatment, the increase in relative seed mass is measured along with strict control of the volume of the water and time required for seed hydration (Warren and Bennett 1997). Various research studies have explained that during drought stress conditions, hydro-priming increases the germination and seedling growth by 3–4 times as compared to nonpriming (Kaur et al. 2002). Likewise, Sung and Chiu (1995) reported that watermelon seeds when subjected to hydro-priming result in fast germination and seedling growth. In addition to several food crops such as Allium porrum, coriander, pyrethrum, and wheat, hydro-priming resulted in a uniform early germination in many desert plants like desert cacti (Dubrovsky 1996).
4.1.2 Osmo-priming
The method of osmo-priming is patented by Heydecker et al. (1973). It is a widely used commercial technique in which seeds are hydrated to a controlled level to allow pregermination metabolic activities (Halmer 2004). During the process, seeds are exposed to a controlled level of imbibition because of excess water entry to seed resulting in reactive oxygen species (ROS) accumulation as well as oxidative damage of cellular components such as proteins, lipid membranes, and nucleic acids. Osmo-priming through a delayed water entry to seed reduces the ROS accumulation and thus protects the cell from oxidative injury. Osmotica such as PEG [H-(O-CH2-CH2)n-OH], sugar, mannitol (C6H14O6), and sorbitol (C6H14O6) are added to the solution for lowering water uptake. In addition, different salts such as NaNO3, MgCl2, NaCl, and KNO3 are used for osmo-priming. Singh et al. (2014) experimented on osmo-priming using cowpea. They used KNO3 as priming solution with three levels of time durations (6, 8, and 10 h). Their results showed that in comparison to unprimed treatments, osmo-priming was proved superior in terms of all germination and growth parameters. Furthermore, osmo-priming with KNO3 showed greater results than hydro-priming for all tested parameters. Osmo-priming is technically and financially more feasible as compared to hydro-priming, because osmo-primed seed results in quicker germination with low cost and better water conservation, thus providing a promising alternative to the farmers (Moradi and Younesi 2009). Jett et al. (1996) stated that the controlled seed hydration in osmo-priming preserves plasma membrane and causes quicker germination. However, during solution selection for osmo-priming, the morphology of seed should be considered as the semipermeable outer layers in some seeds are highly sensitive that affect the efficacy of priming (Pill 1995). The semipermeability of this outer layer is due to the presence of amorphous tissue between seed coat and pericarp that inhibits solute exchange, thus controlling priming agent and water to enter to the seed (Zhou et al. 2013). The internal osmotic equilibrium and nutritional balance of the seed will be disturbed if the solution is not properly selected according to the permeability of the seed due to the penetration of ions released from priming solution (salts) (Bradford 1995).
4.1.3 Nutrient Priming
The saturation of seeds with a certain concentration of nutrients for a specific period before sowing is known as nutrient priming (Shivay et al. 2016). Priming of crop seeds with either micro- or macronutrients enhances the nutrient substances and augments the germination, sprout (seedling) development, and water uptake efficiency. Micronutrient seed priming is a well-known technique to increase the osmosis for water regulation in seeds during the germination period (Singh 2007). For instance, seed primed with sodium molybdate dihydrate (0.02% and 0.04%) for 5 h improved the yield of mung bean (Umair et al. 2011). Likewise, macronutrient seed priming is the most effective technique (Rakshit et al. 2013). For example, potassium (K) is a mineral nutrient, and soaking of crop seeds with K increases the tolerance possibility of plant life against the different environmental stress conditions (Cakmak 2005). Grain yield of wheat and chickpea can be improved by priming with Zn solutions (Arif et al. 2007). Likewise, in moderately Zn-deficient soils such as alkaline soil, Zn priming is helpful to mimic Zn deficiency in plants (Harris et al. 2008). It also improves crop appearance, growth, yield, and nutrient absorption (Shivay et al. 2016). Chickpea seeds primed in 0.05% solution of ZnSO4.7H2O (zinc sulfate heptahydrate) give a 19% high seeds production and have 29% more seed’s Zn as compared to that of nonprimed chickpea seeds (Harris et al. 2008). Besides, mineral nutrient priming increases plants’ tolerance to various environmental stress conditions (Marschner 1995). Ascorbic acid is also an essential vitamin nutrient and has been used for seed priming because it is an antioxidant in nature. Seeds primed with elevated concentrations of internal plant ascorbate retain the antioxidant capability of plants and protect them from oxidative stress damage (Zhou et al. 2009). Tavili et al. (2009) reported that seed treatment with ascorbic acid increases the germination rate of Agropyron elongatum grown in salt stress conditions.
4.1.4 Chemical Priming
Numerous chemicals are in use to soak a variety of crops seeds before germination. Natural and synthetic chemicals like choline, chitosan, putrescine, ethanol, paclobutrazol, ZnSO4, KH2PO4, CuSO4, and Se are used in seed priming to enhance growth and tolerance in crop plants (Jisha et al. 2013). Pretreatment of seeds with these chemicals increases crop plants growth, and plants attain resistance to various abiotic stresses. Priming of Salvia L. and Capsicum annuum L. seeds and other crop species with butenolide compound promotes seedling vitality and emergence (Demir et al. 2012). The improved seedling appearance, because of butenolide pretreatment, lessens the chance of plant pathogens’ attack. Similarly, presoaking rice seeds with (1% or 5%) ethanol solution results in more rapidly and consistent germination rate and high leaf numbers (Farooq et al. 2006). Putrescine is another chemical compound that can be used for seed treatment. Soaking seeds of tobacco in this compound solution develop the cold stress tolerance at the stage of germination and seedling growth through the regulation of antioxidant system (Xu et al. 2011). Also presoaking with a paclobutrazol compound developed salt stress tolerance for Catharanthus roseus because of antioxidant system regulation (Jaleel et al. 2007). Shahrokhi et al. (2011) described that seed priming of turf grass with this compound in drought stress affects the plant physiology, though it is associated with the concentration of the paclobutrazol solution and the temperament of the cultivar. In addition, chitosan is a large-sized cationic polysaccharide molecule generally obtained during waste materials of seafood processing. Priming with chitosan increased disease resistance, the rate, and percentage of germination and the lipolytic activity of lipase, GA3, and indole-3-acetic acid (IAA) quantity and as well enhanced the quality of seeds for crop plants (Shao et al. 2005). Wheat seeds pretreatment in chitosan solution stimulated resistance to several diseases of crops and improved seed quality (Reddy et al. 1999). Seeds layered with chitosan increased seed germination rate and tolerance in seedlings of hybrid rice during stress condition (Ruan and Xue 2002). Maize seeds when soaked in different acidic solutions of chitosan increased the vitality of seedlings (Shao et al. 2005). Furthermore, in cold stress conditions, it enhanced the maize seeds germination velocity and hence benefited the seedlings growth (Guan et al. 2009).
4.1.5 Bio-priming
It is a seed-presoaking technique along with the inoculation of beneficial microorganisms. It combines both the biological agent (microorganisms) and physiological soaking (seed hydration) phase. Callan and Coworkers first depicted the bio-priming in 1990 for the biological management of Pythium pre-emergence of sh2 sweet corn. Incorporated imbibitions with a biocontrol mediator at certain temperature enhance fortification. Additionally, seed priming along with the beneficial microorganisms possibly will promote the maturity of the crop plants, mainly if the inoculated microorganisms colonize the rhizosphere of the plant and maintain plant physiology and plant growth for a longer period (Bennett and Whipps 2008). Since it is a biological approach to use both bacteria and fungi, competitors will also counter to both soil and seeds endured pathogens (Afzal et al. 2016). Callan et al. (1990) reported that bio-priming involves the varnishing of seeds by bacterial biocontrol negotiator like Pseudomonas aureofaciens Kluyver AB254 strain and hydrating at 23 °C for 20 h in damped vermiculite or on damped germination blotters in self-sealing plastic bags. The leakage of seeds release during the period of bio-priming could provide nutrients and strength for inoculated biocontrol agents (Wright et al. 2003). These flattering environments contribute to the migration and propagation of inoculated biocontrol mediators above the surface of the seed and assist water and nutrients uptake throughout the bio-priming period. Priming by a diverse group of beneficial microbes could not only augment the seed’s nature but also boost seedling strength and capability to combat both biotic and abiotic stresses (Rakshit et al. 2015). The microbes mostly designed for bio-priming of seeds belong to Pseudomonas spp., Enterobacter spp., Trichoderma spp., and Bacillus spp. (Raj et al. 2004). For vegetable seeds, adequate bio-priming remedies were accomplished with Trichoderma harzianum strain, followed by Trichoderma pseudokoningii, Bacillus spp., Gliocladium spp., and Pseudomonas fluorescens (Ilyas 2006). Recently, bio-priming used as a substitute to control several soil- and seed-borne pathogens. For instance, mutual response of both Trichoderma harzianum and Pseudomonas fluorescens, when applied on pepper seeds as bio-priming agent, results in a significant growth of seedlings (Kumar et al. 2010; Reddy 2012). Various rhizobacterial inoculants are used as priming agents to control pathogenic fungal strains and to enhance crop yield. Most of the rhizospheric bacterial strains boost plant growth and physiology and thus are called plant growth-promoting rhizobacteria (PGPR) (Tonelli et al. 2011). Within the roots of tomato and rice plants, mycorrhizal fungi activate the aggregation of several transcripts and proteins that also predicts function in the plant defense mechanism (Pozo and Azcon-Aguilar 2007).
4.1.6 Priming with Plant Growth Regulators (PGR)
Seed treatment with plant growth regulators (PGR) is known to mitigate the harmful effects of several environmental stresses (Bahrani and Pourreza 2012; Jisha et al. 2013). Mendoza et al. (2002) reported that priming pepper seeds in salicylic acid protected the seedlings from adverse effects of a chill. Bell pepper primed with GA3 (200 ppm) showed a higher rate of several physiological parameters including germination, shoot root length, and seedling vigor indices as compared to the control (Yogananda et al. 2004). In this context, indole-3-acetic acid (IAA), one of the prime auxins in plants, regulated cell division, enhanced photosynthetic activities, and activated the translocation of carbohydrates that enhance root initiation, flowering, and fruit setting and ripening (MacDonald 1997; Awan et al. 1999; Naeem et al. 2004). Similarly, gibberellins having antagonistic effects with abscisic acid (ABA) regulate seed germination and plant growth. Abu-Muriefah (2017) stated that the improved seed germination due to GA3 priming might be due to its effect on stored food within seeds. He further described that GA3 is known to stimulate the synthesis and production of the hydrolases resulting in the germination of seeds. Certain hydrolase enzymes are involved in seed germination process that makes the endosperm accessible to the embryo. In addition, cytokinins, ethylene, abscisic acid, and salicylic acid are some of the other plant growth promoter hormones that promote growth and regulate plant responses under various stress conditions. Similarly, certain new compounds such as KNO3 (3%), KH2PO4 (2%), and PEG solutions (10%) are now being used as priming agents showing enhanced germination as compared to nonprimed seeds (Korkmaz and Korkmaz 2009; Ozbay and Susluoglu 2016).
4.1.7 Priming with Plant Extract
Allelochemicals such as phenolic compounds, terpenoids, flavonoids, saponins, alkaloids, and steroids may inhibit or stimulate plant growth (Narwal 1994). Saponins can enhance nutrient absorption as they are readily soluble in water. Alkaloids, saponins, and phenolic compounds present in the leaves of various plants are involved in the production of antioxidant activities and protect the plants against pathogens (Satish et al. 2007). Embryo and other associated structures are generally assumed to be activated by certain physiologically active substances that result in more water absorption and eventually in higher vigor index due to the development of an efficient roots system (Rangaswamy et al. 1993). Some plants are rich in saponin and alkaloids such as Chlorophytum leaves, while others are rich in terpenoids, steroids, flavonoids, and antiquinone such as neem leaves (Raphael 2012; Chakraborthy et al. 2014). Dawood et al. (2012) reported that amalgamation of fenugreek seeds (10%) or guava leaves or lantana leaves (20%) into the soil significantly enhanced carbohydrates and photosynthetic pigments of leaf tissues in sunflower. Similarly, the reduction in mortality and high seedling vigor in tomato was reported by priming tomato seeds with Azadirachta, Chlorophytum, and Vinca (Prabha et al. 2016).
4.2 Advanced Methods of Seed Priming
4.2.1 Seed Priming Through Nanoparticles
Nanotechnology utilizes nanoparticles less than 100 nm in size, and it has a promising role in transforming food production and agriculture (Fraceto et al. 2016). The excessive use of chemical fertilizers can be reduced by utilizing nanomaterials in agriculture (Upadhyaya et al. 2017). In this context, priming seeds with nanoparticles has been reported to enhance seed germination and vigor in many crops. Ghafari and Razmjoo (2013) reported that seed priming with calcium-phosphate, SiO2, ZnO, and Ag nanoparticles enhanced germination and seedling development. The mechanism behind high seed germination in nano-priming is the greater penetration via seed coat that improves nutrient and water uptake efficiency of the seed (Dutta 2018).
4.2.2 Seed Priming Through Physical Agents
The magnetic field, UV radiation, gamma radiation, X-rays, and microwaves are some of the physical agents that are used for seed priming (Bilalis et al. 2012). Priming with magnetic field has been reported to improve germination rate, vigor, and seedling biomass as well as tolerance to various environmental stresses. The tolerances to different stresses and improved germination rate have been attributed to a reduction in reactive oxygen species (ROS) with increasing activities of antioxidant enzymes (Bhardwaj et al. 2012; Araujo et al. 2016). However, the effects of ionizing radiation such as gamma (γ) rays are dose and intensity dependent. These rays interact with cellular components directly and are reported to improve the germination at lower doses (less than 10 Gy). Certain changes in hormonal network of plant cells take place that in turn trigger the antioxidative capacity and lead to early dormancy breaking and improved germination (Qi et al. 2015). The application of mechanical waves (ultrasound) is another physical method of priming having a frequency in the range of 20–100 kHz. In ultrasound priming, mechanical pressure is imposed on seed coat that increases the seed’s porosity known as acoustic cavitation and activation of enzymatic and other biological reactions due to greater water uptake in the seed. Thus, in ultrasonic priming, mass transfer of the absorbed water is enhanced that allows it to react freely with the cell embryo.
5 Factors Affecting Seed Priming
Seed priming is highly affected by various biotic and abiotic factors such as aeration, temperature, time, and seed quality. Among these, aeration is the most important and effective factor affecting seed respiration, seed viability, and seed emergence/germination (Bujalski and Nienow 1991; Heydecker et al. 1973). Heydecker and Coolbear (1977) and Bujalski et al. (1989) reported the impact of aeration in seed priming by observing enhanced germination percentage in aerated PEG solution treatment as compared to nonaerated. Similarly, the temperature is another important factor influencing the germination of seeds. Basra et al. (2005) reported that optimum temperature ranges from 15 to 30 °C for most of the seed germination. On the other hand, McDonald (2000) reported slow germination at a lower priming temperature. Wahid et al. (2008) documented a range of 15–20 °C for seed priming, and the duration of priming may extend from almost 8 h to 14 days based on plant species, osmotic solution, osmotic potential, and temperature (Finch-Savage et al. 1991). Seed quality is another important factor in seed germination, and a viable and vigorous seed is the first most necessary for seed priming (Cantliffe 1987). Other seed characteristics also play a role in seed priming and germination process. For instance, Patanè et al. (2008) reported osmo-priming with PEG solution unsuitable for sorghum seeds priming. Sorghum is rich in tannin that could be removed with the solution during treatment and hence leads to lower seed germination. In this regard, Passam et al. (1989) stated that the salt solution is effective as compared to mannitol and PEG solutions. Similarly, O’Sullivan and Bouw (1984) reported KNO3 and K3PO4 as an effective priming solution in pepper seeds as compared to PEG.
6 Seed Priming: Physiological Basis and Plant Response
6.1 Occurrence of Seed Germination and Seedling Growth
In vegetable crops, high yield and growth are primarily associated with seedling health and early emergence which induces a potential to cope with various biotic and abiotic stresses. These all result in high yield and quality crops (Cantliffe 2003). Seed priming is one of the primitive techniques used to enhance early seed emergence and initiates several processes involved in seed germination (Asgedom and Becker 2001). Therefore, seed priming boosts the imbibition and metabolic processes resulting in enhanced seed germination, germination uniformity, and seedling growth and development in both normal and stress conditions (Ansari et al. 2012; Dey et al. 2014; Nayban et al. 2017). Several studies have reported that seed priming can enhance early seed emergence and growth in stress conditions as compared to untreated seeds (Bradford 1986; Chen et al. 2012). Yogananda et al. (2004) observed the increased physiological response of bell pepper treated with GA3 and KNO3 solution. Similarly, Yadav et al. (2011) also reported cold and salt stress tolerance in primed seeds of pepper with 100% survival. In another study, Ahmadvand et al. (2012) evaluated the effect of KNO3-primed seeds of soybean in various cultivars and reported a significant increase in germination and seed emergence followed by increased physiological parameters like fresh weight, dry weight, root length, and shoot length.
6.2 Crop Nutrition and Yield
Crop nutrient deficiency known as hidden hunger is a global dilemma resulting in low yields and poor-quality products (Dey et al. 2014). Seed priming is an approach to provide nutrients to the seed emergence and activate various biochemical processes necessary for seed germination. This not only provides the nutrients to establish seedling growth and emergence but also helps in enrichment of grain nutrient status (Singh et al. 2015a, b) and has been proved an economical method of nutrients application as compared to soil application and has been found economical as compared to soil application (Slaton et al. 2001). Ajouri et al. (2004) reported Zn as an effective priming agent in barley (Hordeum vulgare L.). Another study was done to assess the Zn content of early grown radicles and coleoptiles and was found much higher up to 200 mg kg−1. The study concluded that Zn might be involved in various processes during early seed development (Cakmak 2005). Additionally, higher contents of Zn may enhance seed resistance to soil-borne diseases and ensure crop growth and development (Marschner 1995). Several studies by Khan et al. (2008), Bhowmick et al. (2010), Umair et al. (2013), and Bhowmick (2013) have reported seed priming as a suitable strategy for increased crop yields. Harris (2006) reported that increased crop yield might be attributed to crop density and individual crop performance. Srivastava and Bose (2012) studied the effect of (Mg(NO3)2 and KNO3) salts as a priming agent for rice crop and reported increased physiological response to primed seeds as compared to untreated. Similarly, Arif et al. (2008) also studied the effect of priming seeds in soybean and reported priming increased seed emergence and establishment and attributed all this to the early metabolic activities as it activates radical protrusion. However, they reported that extended priming duration might decrease crop yield and 6 h was found as a suitable duration for soybean seed priming.
6.3 Seed Priming for Stress Management
Field crops are subject to various types of biotic and abiotic stress like herbivores, pathogens attack as well as cold, heat, heavy metal, nutrient deficiency, salinity, and drought (Fedoroff et al. 2010). Among these stresses, salinity, drought, and temperature cause low growth and development (Jaleel et al. 2007; Thakur et al. 2010). These types of stress induce ROS production in a plant cell that results in cell injury and ultimately plants failure. In order to combat these ROS, plants have a self-defense mechanism that eliminates ROS species and protects plant cells from damage (Baxter et al. 2013). The intensity of stress injury depends upon various stress-related factors like stress tolerance, timing, and intensity (Niinemets 2009). Seed priming is considered one of the most promising techniques to enhance seed resistance to all these biotic and abiotic stresses (Van Hulten et al. 2006). Uchida et al. (2002) reported the importance of compounds used in seed priming technique as they alleviate the deteriorating effects of both biotic and abiotic stresses concerning plant growth. Kibinza et al. (2011) explained that germination percentages were improved by priming that was due to the considerable drop in H2O2 accumulation. Catalase activity was restored by priming that protects the stressed seeds from reactive oxygen species. Furthermore, Kester et al. (1997) reported that protein content in various plant tissues might be increased by seed priming via improved performance of protein synthesis system and increased production of L-isoaspartyl methyltransferase enzyme that plays an important role in repairing plant tissue proteins. Priming also enhances seed germination by enhancing the activities of protease and amylase that hydrolyze protein and starch into simple forms to make them available for the embryo (Miransari and Smith 2014). Sajedi et al. (2011) stated that ROS produced under PbCl2 stress may be reduced by hormonal priming. Similarly, in another study, Pereira et al. (2009) showed improved seedling emergence by osmo-priming on carrot seed germination under extreme temperature conditions and water stress.
7 Assessment of Priming Effects on Plant Growth and Development
7.1 Seed Priming Using Compost Extract for Improving Germination Parameters
Compost tea, a liquid extract, is obtained when compost is steeped in water for a period with the aim of transferring soluble organic matters (OM), beneficial microbes, and nutrients into the solution (Mohd Din et al. 2017). The application of this microbial- and nutrient-rich tea is known for stimulating the plant’s growth and soil fertility (Ahmad et al. 2017). Compost tea is prepared aerobically (aerated tea) or anaerobically (nonaerated tea); however, little evidence exists as regards which method either aerated or nonaerated is more beneficial for agricultural purposes (Amos 2017). There have been numerous studies for evaluating the potential of compost tea in suppressing the plant diseases (Mengesha et al. 2017). However, there are limited studies on the effect of compost tea on crop growth and development (Kim et al. 2015). Therefore, an assessment that was carried out aims to examine the stimulatory and inhibitory effects of compost teas on seed germination and seedling growth through a novel method of seed priming in compost tea, since no previous study has been conducted.
7.2 Preparation and Characteristics of Compost Tea
The compost used for compost teas production was prepared from food waste using an in-vessel compost bioreactor (Waqas et al. 2017). Two different types of compost teas, i.e., aerated and nonaerated, were prepared by mixing compost and water at 1:10 ratio (i.e., 1 kg dry compost in 10 L distilled water). The mixture was steeped at 25 °C for 72 h. A standard brewing method was used for steeping and extraction period during which the compost was in contact with water (Scheuerell and Mahaffee 2006). The aeration, in aerated compost tea, was maintained on a continuous basis by stirring the solution through a mechanical agitator with 80 revolutions per minute (RPM) throughout the extraction period of 72 h. The schematic diagram of the bioreactor used for making the aerated compost tea is shown in Fig. 2. Nonaerated compost tea was prepared by using a standard method of bucket fermentation (Diver 2002). This approach is referred to as the European-style or European method for compost tea preparation and dates back to hundreds of years (Brinton et al. 2004). During this process, the mixture was initially stirred and then left undisturbed at 25 °C for 3 days (Weltzien 1991). After the designated steeping time, the compost extracts were filtered through a muslin cloth. Different physiochemical characteristics of the compost tea were determined by following the standard methods of analysis, and the results are presented in Table 2. For experimentation, the compost tea was diluted using distilled water. The dilution concentrations were 0% (100% extract), 25% (75% extract, 25% water), 50% (50% extract, 50% water), 75% (25% extract, 75% water), and 100% (100% water). The 100% compost tea diluted solution was used as a control treatment for comparing the concentration effects of both compost teas.
7.3 Seed Priming and Experimental Setup
Seed priming in the compost tea was introduced by soaking the mung bean seeds in compost tea for a definite period of time. Seeds’ surfaces were disinfected to avoid any bacterial or fungal contamination by imbibing them in a mixture of ethanol and distilled water (70:30 v/v) for 5 min and subsequently rinsing them with distilled water. Ten seeds of mung bean were then primed/soaked over dampened filter paper to assess the compost tea phytotoxicity. 20 ml of a diluted solution of each compost tea was used as a priming solution in 15 cm petri dish (Fisher brand, Fisher Scientific, Waltham MA). The petri dishes were incubated for 24 h at 25 °C in the dark and were then covered with a plastic wrapping to avoid any water loss during the priming (Mavi 2014). After priming, the seeds were washed with distilled (for consistency reasons) water and dried back to about 14% grain moisture at 36 °C in drying oven (Pame et al. 2015). A factorial experiment in a totally randomized design was used to investigate the effects of compost tea aeration methods (aerated vs. nonaerated) and priming treatments (primed vs. unprimed) on a lot of ten mung bean seeds at five different dilution concentrations of 0%, 25%, 50%, 75%, and 100%. The tea dilution at 100% solution was referred to control treatment as it contained 0% compost tea and 100% distilled water. Three replicates were used for each treatment combination. Ten seeds of mung bean were homogenously distributed on two layers of sterile Whatman™ filter paper in each petri dish. For unprimed seeds, 5 ml of each diluted solution of compost tea was applied to moisten the filter paper, whereas the petri dishes containing primed seed in compost tea were applied with only distilled water when required. The petri dishes were monitored daily, and an equal amount of tea solutions/distilled water was supplied when necessary to keep the moisture of seeds or seedlings at adequate levels. Petri dishes were incubated in a growth chamber at 27 °C for 14 days. During experimentation, different germination and growth parameters were determined through standard formulas, and the obtained data were subjected to analysis of variance (ANOVA) and least significant difference (LSD) test at 5% probability level (Steel et al. 1997).
7.4 Effects on Germination Parameters
7.4.1 Germination Rate and Germination Index (GI)
The ANOVA results indicated that compost teas’ dilution and seed priming with compost tea significantly affected (state the level of significance, e.g., α = 0.05) the seed germination and germination index. No significant differences on seed germination rate and germination index were observed for the aeration system, i.e., aerated and nonaerated teas exert the same effect on germination. The germination rate of the seeds exposed to aerated compost tea was equal to 86.3%, like nonaerated tea (86%). Among priming effects, the highest stimulation in germination rate was recorded for primed seeds, resulting in 91.6% germination, whereas that of unprimed seeds was recorded at 80.6% (Table 3). Similarly, among the tea dilution concentrations, the highest germination rate (94.2%) was observed in dilution of tea to 50%. However, a drastic reduction in germination rate was observed with increasing the tea concentration, and it was noticed that at 0% dilution (100% tea concentration), the lowest germination rate of 72.5% was observed (Table 3). Similarly, the two-way interaction between the aeration system with priming and dilution and the three-way combined interaction of aeration, dilution, and priming showed a nonsignificant effect on germination rate.
Similarly, the results showed that the maximum GI of 4.5 was estimated for primed seed, whereas the lowest GI of 2.5 was recorded for unprimed seed (Table 3). It was observed that priming had an advantageous effect in terms of seed germination parameters. Similarly, the mean data for tea dilution showed that the maximum GI of 3.8 in relation to control treatment was recorded at a dilution of tea to 50%, which was similar to 75% diluted solution with GI of 3.8 (Table 3). Moreover, like germination rate, reduction in values of GI was also observed in the low diluted solutions of compost tea. The results showed that the minimum GI values of 2.8 and 3.3 were recorded at 0 and 25% diluted solutions, respectively (Table 3). In addition, the ANOVA results showed significant effects of the interaction of aeration with dilution ratios. The interaction data revealed that the maximum GI value of 4 was recorded in aerated tea at 50% dilution followed by dilution of tea to 75% with GI of 3.9 in the same tea (Table 4). Similarly, for nonaerated compost tea, the values for same tea diluted concentration (50 and 75%) were observed to be lower than aerated tea. For nonaerated compost tea, the results showed that the maximum GI value of 3.9 had been recorded for control treatment followed by 75% and 50% dilution with the GI of 3.8 and 3.6, respectively. However, for both aerated and nonaerated teas, the lowest value for GI was observed for dilution of teas to 25% and 0% (Table 4).
7.4.2 Mean Germination Time (MGT) and Seed Vigor Index (SVI)
MGT showed the rapidity of germination; hence, the lower the value of MGT, the earlier is the germination. The ANOVA results showed highly significant variation for MGT in dilution and priming of compost tea. The mean data in Table 3 revealed that the rapid germination was recorded for primed seeds with least MGT value of 4.8, whereas the higher MGT value of 7.1 for unprimed depicts the delayed seed germination. For dilution of tea, the lowest MGT value of 5.3 was noticed at 75% tea dilution. However, this value for MGT was statistically near to dilution of tea to 50% (5.6). Similarly, the maximum MGT value of 6.5 was computed for control treatment that showed low rapidity of seed germination. For aeration, the value for MGT in aerated tea was 6.1, which was statistically similar to the MGT value of 5.9 for nonaerated tea (Table 3). This nonsignificant difference showed that the aeration system did not affect MGT of the tested seeds. The overall results revealed that priming reduced the MGT by 31.2% in comparison with unpriming. Similarly, compared to control, the solution containing 25% and 50% compost tea reduced the MGT by 18.4 and 12.9%, respectively.
SVI expressed the level of seed performance and activity during the germination and seedling emergence. It was found that SVI was significantly affected by aeration, tea dilution, and priming. Moreover, the results indicated that the two-way interaction between aeration with dilution and priming also had significant effects toward SVI. The results showed that among the aeration systems, the utmost SVI of 221.7 was recorded for aerated tea, leaving behind nonaerated tea with the value of 135.3 (Table 3). Similarly, among the priming effects, the highest values were recorded for primed seed that was 234.1 whereas the least values (122.9) were estimated for unprimed seeds. In addition, compost tea dilution showed high variation toward SVI. The highest SVI (243.4) was observed for dilution of tea to 75% that was statistically like 50% diluted tea (241), whereas the least SVI (73.2) was recorded for dilution of tea to 0% (Table 3). Furthermore, the mean data for the two-way interaction of aeration with dilution showed that the maximum SVI (336) was observed for aerated tea diluted at 50% (Table 4). This value was found statistically similar to 75% tea dilution with the SVI value of 320.7. Similarly, the lowest SVI of 66.3 was recorded at 0% tea dilution in nonaerated (Table 4). Furthermore, the mean data for the interaction of priming with dilution showed that the utmost values for SVI (353.5) were observed for primed seeds with a dilution of tea to 50% followed by 25 and 75% diluted tea solutions with SVI values of 272 and 270.3, respectively (Table 5), whereas the minimum SVI (28.2 and 45.8) was noticed for dilution of tea to 0 and 25% in unprimed seeds, respectively (Table 5). The combined interaction of aeration, dilution, and priming showed that highest SVI (526.7) was noticed in primed seeds with the dilution of aerated compost tea to 50%. Similarly, the second highest peak value of 370.7 for SVI also observed for primed seed with aerated tea at tea dilution to 75%. Conversely, the lowest SVI (25.7 and 30.7) was noticed for unprimed seeds at 0% tea dilution in both the aerated and nonaerated teas, respectively (Fig. 3).
7.4.3 Effects on Root and Shoot Length
The tea dilution and priming and their combined interaction affected significantly (a = 0.05) mung bean root and shoot length. However, the effect of aeration on the shoot length was observed nonsignificant. The results showed that the maximum mean root length (2.4 cm) was recorded for aerated tea. Similarly, the mean data for priming depict that the utmost root (2.5 cm) and shoot length (7.1 cm) were observed for primed seed, whereas for unprimed seed, the observed root and shoot length were 1.5 and 3.8 cm, respectively (Table 3). In addition, the mean data for compost tea dilution showed that the maximum root length (2.7 and 2.5 cm) was observed for dilution of teas to 75 and 50%, whereas the highest shoot length (7.2 cm) was noticed for dilution of tea to 75%. Correspondingly, the minimum root (0.9 cm) and shoot length (3.3 cm) was estimated for dilution of tea to 0% (Table 3). The interaction of aeration with dilution showed that among the aerated and nonaerated compost teas, the highest root length (3.6 cm) was recorded for tea dilution to 75% that was statistically similar to dilution of tea to 50% (3.4 cm) (Table 4). Similarly, the highest shoot length (7.7 cm) was recorded in aerated compost tea at 75% dilution, whereas the least values for root and shoot length (0.9 and 3.1 cm) were observed in nonaerated compost tea at 0% dilution followed by 25% dilution of the same tea (Table 4). As the ANOVA results also showed significant differences among the interaction between priming and tea dilution, the utmost root length (3.6 cm) was observed for primed seed in compost tea at 50% dilution, whereas for shoot length, the maximum value (8.6 cm) was recorded at 25% dilution. The bare minimum root and shoot length (0.4 and 0.6 cm) were calculated for unprimed seeds treated with compost tea at 0% dilution followed with 25% dilution in the same tea (Table 5). The combined interaction of aeration, tea dilution, and priming revealed that the maximum seedling root length (5.3 cm) was recorded for primed seed at 50% diluted solution of the aerated tea (Fig. 4). Similarly, the highest shoot length of 10 and 9.2 cm was recorded for seeds primed with dilution of aerated tea to 25 and 50%, respectively (Fig. 5), whereas the maximum inhibition in root and shoot length was resulted by 100% tea concentration (0% dilution). The recorded root and shoot length at the respective 0% dilution were 0.4 and 0.73 cm in aerated and 0.5 and 0.6 cm in nonaerated compost tea (Figs. 4 and 5).
8 Limitations and Perspective in Seed Priming Technology
Seed priming has been developed as a promising technology for superficial crop stand in a variety of environmental conditions. However, many protocols such as seed desiccation (redrying) after priming may affect different physiochemical process which reduces seed longevity and viability (Heydecker and Gibbins 1977; Halmer 2004). Other conditions for posttreatment such as storage temperature, air composition, and moisture also negatively affect seed viability (Schwember and Bradford 2005). Similarly, the prolonged seed treatment during priming may also cause loss of seed tolerance to desiccation (Sliwinska and Jendrzejczak 2002). Priming itself in certain circumstances may also cause different problems. For instance, all priming protocols may not lead to significant germination and growth where inappropriate priming conditions may cause degradation of the protective proteins (Capron et al. 2000). Hence, it is critical to select specific priming protocol for different plants about germination and growth in different environmental conditions. Thus, for filling the gap and successful application of priming technology, detailed studies focusing on treatment technologies, gene expressions, and molecular mechanisms need to be fully explored (Araujo et al. 2016). Correspondingly, the advanced methods of seed priming such as priming with nanoparticles may also have deleterious effects on environment, plant, and human health. In this regard, solid studies need to be performed for resolving the impact of nanomaterials when enter the food chain by using them in agriculture. Extensive researches are still required for each priming technology in terms of optimal dose, exposure time, and dose rate that could affect plant growth and development.
9 Conclusions
Seed priming is the physiological process of controlled seed hydration to enhance sufficient pregerminative metabolic process, efficient nutrient uptake and water use efficiency, breaking dormancy, timely maturity, and crop yield. During imbibition, the water uptake promotes protein synthesis and respiratory activities by using extant messenger ribonucleic acid (mRNA) with the initiation of different physiological activities related to germination. This technology has been found to be the most feasible and economical for uniform seed emergence in most of the field crops. There are many well-developed seed-priming techniques such as hydro-priming, osmo-priming, nutrient priming, chemical priming, bio-priming, priming with plant growth regulators, priming with plant extracts, seed priming through nanoparticles, and priming through physical agents. However, priming technology still has several limitations. The prolonged seed treatment during priming may cause loss of seed tolerance to desiccation that reduces seed viability. Similarly, all priming protocols may not lead to significant germination and growth where inappropriate priming conditions may cause degradation in the protective proteins. Hence, extensive researches are required in selecting specific priming protocol for different plants regarding germination and growth under various environmental conditions.
References
Abu-Muriefah SS (2017) Phytohormonal priming improves germination and antioxidant enzymes of soybean (Glycine max) seeds under lead (Pb) stress. Biosci Res 14(1):42–56
Adhikari T, Kundu S, Rao AS (2013) Impact of SiO2 and Mo nano particles on seed germination of rice (Oryza sativa L.). Int J Agric Food Sci Technol 4(8):809–816
Afzal I, Rehman HU, Naveed M, Basra SMA (2016) Recent advances in seed enhancements. In New challenges in seed biology-basic and translational research driving seed technology. InTech, pp 47–74
Ahmad I, Basra SMA, Akram M, Wasaya A, Ansar M, Hussain S, Iqbal A, Hussain SA (2017) Improvement of antioxidant activities and yield of spring maize through seed priming and foliar application of plant growth regulators under heat stress conditions. Semina: Ciências Agrárias 38:47–56
Ahmadvand G, Soleimani F, Saadatian B, Pouya M (2012) Effect of seed priming with potassium nitrate on germination and emergence traits of two soybean cultivars under salinity stress conditions. Am Eurasian J Agric Environ Sci 12:769–774
Ajouri A, Asgedom H, Becker M (2004) Seed priming enhances germination and seedling growth of barley under conditions of P and Zn deficiency. J Plant Nutr Soil Sci 167(5):630–636
Amos D (2017) Aerated compost tea (ACT) to improve soil biology and to act as a biofertiliser/biofungicide, pp 1–2. http://orgprints.org/31042/. Accessed 2 June 2017
Ansari O, Chogazardi H, Sharifzadeh F, Nazarli H (2012) Seed reserve utilization and seedling growth of treated seeds of mountain rye (Secale montanum) as affected by drought stress. Cercetari Agronomice in Moldova 45(2):43–48
Araujo SDS, Paparella S, Dondi D, Bentivoglio A, Carbonera D, Balestrazzi A (2016) Physical methods for seed invigoration: advantages and challenges in seed technology. Front Plant Sci 7:646
Arif M, Waqas M, Nawab K, Shahid M (2007) Effect of seed priming in Zn solutions on chickpea and wheat. Afr. Crop Sci Conf Proc 8:237–240
Arif M, Jan MT, Marwat KB, Khan MA (2008) Seed priming improves emergence and yield of soybean. Pak J BotPak J Bot 40(3):1169–1177
Asgedom H, Becker M (2001) Effects of seed priming with nutrient solutions on germination, seedling growth and weed competitiveness of cereals in Eritrea. In: Proceedings of Deutscher Tropentag. University of Bonn & ATSAF, Margraf Pub. Press, Weickersheim, p 282
Assefa MK, Hunje R, Koti RV (2010) Enhancement of seed quality in soybean following priming treatment. Karnataka J Agric Sci 23:787–789
Awan IU, Baloch MS, Sadozai NS, Sulemani MZ (1999) Stimulatory effect of GA3 and IAA on ripening process, kernel development and quality of rice. Pakistan J Biol Sci(Pakistan) 2:410–412
Bagheri MZ (2014) The effect of maize priming on germination characteristics, catalase and peroxidase enzyme activity and total protein content under salt stress. Int J Biosci 4(2):104–112
Bahrani A, Pourreza J (2012) Gibberellic acid and salicylic acid effects on seed germination and seedlings growth of wheat (Triticum aestivum L.) under salt stress condition. World Appl Sci J 18(5):633–641
Basra SMA, Farooq M, Tabassam R, Ahmad N (2005) Physiological and biochemical aspects of pre-sowing seed treatments in fine rice (Oryza sativa L.). Seed Sci Technol 33(3):623–628
Baxter A, Mittler R, Suzuki N (2013) ROS as key players in plant stress signalling. J Exp Bot 65(5):1229–1240
Benavides-Mendoza A, Ramirez-Rodriguez H, Robledo-Torres V, Hernandez-Davila J, Ramirez-Mezquitic JG, Bacopulos-Te-llez E, Bustamante-Garcia MA (2002) Seed treatment with salicylates modifies stomatal distribution, stomatal density and the tolerance to cold stress in pepper seedlings. In: Proceedings of the 16th international pepper conference (Tampico, Tamaulipas, Mexico (Vol. 2), November
Bennett AJ, Whipps JM (2008) Dual application of beneficial microorganisms to seed during drum priming. Appl Soil Ecol 38(1):83–89
Berrie AMM, Drennan DSH (1971) The effect of hydration-dehydration on seed germination. New Phytol 70(1):135–142
Bhardwaj J, Anand A, Nagarajan S (2012) Biochemical and biophysical changes associated with magnetopriming in germinating cucumber seeds. Plant Physiol Biochem 57:67–73
Bhowmick MK (2013) Seed priming and foliar nutrition towards enhancing Lathyrus productivity under rice-utera system. In: Proceedings of the Centenary Session, 100th Indian Science Congress, Section of Agriculture and Forestry Sciences, 3–7 Jan 2013, Kolkata, West Bengal, India, p 219
Bhowmick M K, Biswas PK, Sen P, Bhattacharyya P (2010) Studies on seed priming, row spacing and foliar nutrition in chickpea under rainfed conditions in West Bengal, India. In: Proceedings of the international seminar on climate change and environmental challenges of 21st century, December, pp 7–9
Bilalis DJ, Katsenios N, Efthimiadou A, Karkanis A, Efthimiadis P (2012) Investigation of pulsed electromagnetic field as a novel organic pre-sowing method on germination and initial growth stages of cotton. Electromagn Biol Med 31(2):143–150
Bradford KJ (1986) Manipulation of seed water relations via osmotic priming to improve germination under stress conditions. HortScience (USA)
Bradford KJ (1995) Water relations in seed germination. Seed Dev Germination 1(13):351–396
Brinton W, Storms P, Evans E, Hill J (2004) Compost teas: microbial hygiene and quality in relation to method of preparation. Biodynamics. 249:36–45
Bujalski W, Nienow AW (1991) Large-scale osmotic priming of onion seeds: a comparison of different strategies for oxygenation. Sci Hortic 46(1–2):13–24
Bujalski W, Nienow AW, Gray D (1989) Establishing the large scale osmotic priming of onion seeds by using enriched air. Ann Appl Biol 115(1):171–176
Cakmak I (2005) The role of potassium in alleviating detrimental effects of abiotic stresses in plants. J Plant Nutr Soil Sci 168(4):521–530
Callan NW, Mathre D, Miller JB (1990) Bio-priming seed treatment for biological control of Pythium ultimum pre-emergence damping-off in sh-2 sweet corn. Plant Dis 74:368–372
Cantliffe DJ (1987) Priming of lettuce for early and uniform emergence under conditions of environmental stress. Acta Hortic 122:29–38
Cantliffe DJ (2003) Seed enhancements. Acta Hortic 607:53–59
Capron I, Corbineau F, Dacher F, Job C, Côme D, Job D (2000) Sugar beet seed priming: effects of priming conditions on germination, solubilization of 11-S globulin and accumulation of LEA proteins. Seed Sci Res 10(3):243–254
Chakraborthy GS, Aeri V, Verma P, Singh S (2014) Phytochemical and antimicrobial studies of Chlorophytum borivilianum. Pharmacophore 5:258–261
Chen K, Fessehaie A, Arora R (2012) Dehydrin metabolism is altered during seed osmopriming and subsequent germination under chilling and desiccation in Spinaciaoleracea L. cv. Bloomsdale: possible role in stress tolerance. Plant Sci 183:27–36
Côme D, Thévenot C (1982) Environmental control of embryo dormancy and germination. In: The physiology and biochemistry of seed development, dormancy and germination, pp 271–298
Corbineau F, Côme D (2006) Priming: a technique for improving seed quality. Seed testing international. ISTA News Bulletin No 132:38–40
Darwin C (1855) Effect of salt-water on the germination of seeds. Gardeners Chron Agric Gaz 47:773
Dawood MG (2018) Stimulating plant tolerance against abiotic stress through seed priming. In: Advances in seed priming. Springer, Singapore, pp 147–183
Dawood MG, El-Awadi ME, El-Rokiek KG (2012) Physiological impact of fenugreek, guava and lantana on the growth and some chemical parameters of sunflower plants and associated weeds. J Ame Sci 8(6):166–174
Demir I, Ozuaydın I, Yasar F, Van Staden J (2012) Effect of smoke-derived butenolide priming treatment on pepper and salvia seeds in relation to transplant quality and catalase activity. S Afr J Bot 78:83–87
Dey S, Pramanik K, Mukherjee S, Poddar S, Barik K (2014) Effect of seed priming and straw mulching on growth and productivity of chickpea (Cicer arietinum L.) under rainfed condition. SATSA Mukhaptra Annu Tech Issue 18:126–131
Di Girolamo G, Barbanti L (2012) Treatment conditions and biochemical processes influencing seed priming effectiveness. Ital J Agron 7(2):25
Diver S (2002) Notes on compost teas: a supplement to the ATTRA publication: compost teas for plant disease control. Appropriate Technology Transfer for Rural Areas (ATTRA), National Sustainable Agriculture Information Service, AR. (National Center for Appropriate Technology, Montana). Retrieved March, 5, 2012
Dubrovsky JG (1996) Seed hydration memory in Sonoran Desert cacti and its ecological implication. Am J Bot 83(5):624–632
Dutta P (2018) Seed priming: new vistas and contemporary perspectives. In: Advances in seed priming. Springer, Singapore, pp 3–22
Ells JE (1963) The influence of treating tomato seed with nutrient solutions on emergence rate and seedling growth. In Proc Amer Soc Hort Sci 83:684–687
Evenari M (1984) Seed physiology: its history from antiquity to the beginning of the 20th century. Bot Rev 50(2):119–142
Farahbakhsh H (2012) Germination and seedling growth in un-primed and primed seeds of Fenel as affected by reduced water potential induced by NaCl. Int Res J Appl Basic Sci 3(4):737–744
Farooq M, Basra SM, Rehman H, Mehmood T (2006) Germination and early seedling growth as affected by pre-sowing ethanol seed treatments in fine rice. Int J Agric BiolInt J Agric Biol 8:19–22
Farooq M, Basra SMA, Rehman H, Saleem BA (2008) Seed priming enhances the performance of late sown wheat (Triticum aestivum L.) by improving chilling tolerance. J Agron Crop Sci 194(1):55–60
Farooq M, Wahid A, Lee DJ (2009) Exogenously applied polyamines increase drought tolerance of rice by improving leaf water status, photosynthesis and membrane properties. Acta Physiol Plant 31(5):937–945
Farooq M, Basra SM, Wahid A, Ahmad N (2010) Changes in nutrient-homeostasis and reserves metabolism during rice seed priming: consequences for seedling emergence and growth. Agric Sci China 9(2):191–198
Fedoroff NV, Battisti DS, Beachy RN, Cooper PJ, Fischhoff DA, Hodges CN et al (2010) Radically rethinking agriculture for the 21st century. Science 327(5967):833–834
Finch-Savage WE, Gray D, Dickson GM (1991) The combined effects of osmotic priming with plant growth regulator and fungicide soaks on the seed quality of five bedding plant species. Seed Sci Technol 19(2):495–503
Fraceto LF, Grillo R, de Medeiros GA, Scognamiglio V, Rea G, Bartolucci C (2016) Nanotechnology in agriculture: which innovation potential does it have? Front Environ Sci 4:20
Gaius PS (1949) Naturalishistoria, vol. IV–VII, Books 12–27 (trans: Rackham H, Jones WHS, Eichholz DE). Harvard University Press, Massachussets and William Heinemann, London
Ghafari H, Razmjoo J (2013) Effect of foliar application of nano-iron oxidase, iron chelate and iron sulphate rates on yield and quality of wheat. Int JAgron Plant Prod 4(11):2997–3003
Guan YJ, Hu J, Wang XJ, Shao CX (2009) Seed priming with chitosan improves maize germination and seedling growth in relation to physiological changes under low temperature stress. J Zhejiang Univ Sci B 10(6):427–433
Halmer P (2004) Methods to improve seed performance in the field. In: Handbook of seed physiology, pp 125–65
Harris D (2006) Development and testing of “on-farm” seed priming. Adv Agron 90:129–178
Harris D, Rashid A, Miraj G, Arif M, Yunas M (2008) ‘On-farm’seed priming with zinc in chickpea and wheat in Pakistan. Plant Soil 306(1–2):3–10
Heydecker W, Coolbear P (1977) Seed treatments for improved performance-survey and attempted prognosis. Seed Sci Technol 5:353–425
Heydecker W, Gibbins BM (1977) The‘priming’of seeds. In: Symposium on seed problems in horticulture, vol 83, pp 213–224
Heydecker W, Higgins J, Gulliver RL (1973) Accelerated germination by osmotic seed treatment. Nature 246(5427):42
Hill H, Bradford KJ, Cunningham J, Taylor AG (2008) Primed lettuce seeds exhibit increased sensitivity to moisture during aging. Acta Hortic 782:135–141
Ilyas S (2006) Seed treatments using matriconditioning to improve vegetable seed quality. J Agronomi Indonesia 34(2)
Jaleel CA, Gopi R, Manivannan P, Panneerselvam R (2007) Responses of antioxidant defense system of Catharanthus roseus (L.) G. Don. Topaclobutrazol treatment under salinity. Acta Physiol Plant 29(3):205–209
Jett LW, Welbaum GE, Morse RD (1996) Effects of matric and osmotic priming treatments on broccoli seed germination. J Am Soc Hortic Sci 121(3):423–429
Jisha KC, Vijayakumari K, Puthur JT (2013) Seed priming for abiotic stress tolerance: an overview. Acta Physiol Plant 35(5):1381–1396
Kaur S, Gupta AK, Kaur N (2002) Effect of osmo-and hydropriming of chickpea seeds on seedling growth and carbohydrate metabolism under water deficit stress. Plant Growth Regul 37(1):17–22
Kester ST, Geneve RL, Houtz RL (1997) Priming and accelerated ageing affect L-isoaspartyl methyltransferase activity in tomato (Lycopersicon esculentum Mill.) seed. J Exp Bot 48(4):943–949
Khaliq A, Aslam F, Matloob A, Hussain S, Geng M, Wahid A, ur Rehman H (2015) Seed priming with selenium: consequences for emergence, seedling growth, and biochemical attributes of rice. Biol Trace Elem Res 166(2):236–244
Khan A, Khalil SK, Khan AZ, Marwat KB, Afzal A (2008) The role of seed priming in semi-arid area for mung bean phenology and yield. Pak J Bot 40(6):2471–2480
Kibinza S, Bazin J, Bailly C, Farrant JM, Corbineau F, El-Maarouf-Bouteau H (2011) Catalase is a key enzyme in seed recovery from ageing during priming. Plant Sci 181(3):309–315
Kim MJ, Shim CK, Kim YK, Hong SJ, Park JH, Han EJ, Kim JH, Kim SC (2015) Effect of aerated compost tea on the growth promotion of lettuce, soybean, and sweet corn in organic cultivation. Plant Pathol J 31(3):259–268
Koehler DE (1967) Thesis. Purdue University, USA
Korkmaz A, Korkmaz Y (2009) Promotion by 5-aminolevulenic acid of pepper seed germination and seedling emergence under low-temperature stress. Sci Hortic 119(2):98–102
Kumar S, Arya MC, Sinfh R (2010) Management of sweet pepper diseases and growth promotion by Pseudomonas fluorescens and Trichoderma harzianum in mid hills of Central Himalayas, India. Indian Phytopathol 63(2):181–186
Lara TS, Lira JMS, Rodrigues AC, Rakocevic M, Alvarenga AA (2014) Potassium nitrate priming affects the activity of nitrate reductase and antioxidant enzymes in tomato germination. J Agric Sci 6(2):72
MacDonald H (1997) Auxin perception and signal transduction. Physiol Plant 100:423–430
Mahajan G, Sarlach RS, Japinder S, Gill MS (2011) Seed priming effects on germination, growth and yield of dry direct-seeded rice. J Crop Improv 25(4):409–417
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic, San Diego. Mineral nutrition of high plant. Academic Press, pp: 330–355
Mavi K (2014) Use of extract from dry marigold (Tagatesspp.) flowers to prime eggplant (Solanum melongenaL.) seeds. Acta Sci Pol-Hortorumcultus 13:3–12
Mavi K (2016) The effect of organic priming with Marigold herbal tea on seeds quality in Aji pepper (Capsicum baccatum var. pendulum Willd.). Mustafa Kemal Üniversitesi Ziraat Fakültesi Dergisi 21(1):31–39
May LH, Milthorpe EJ, Milthorpe FL (1962) Pre-sowing hardening of plants to drought. In: Field crop abstracts, vol 15, pp 93–98
McDonald MB (2000) Seed priming. In: Black M, Bewley JD (eds) Seed technology and its biological basis. Sheffield Academic Press, Sheffield, pp 287–325
Mengesha WK, Powell SM, Evans KJ, Barry KM (2017) Diverse microbial communities in non-aerated compost teas suppress bacterial wilt. World J Microbiol Biotechnol 33(3):49–55
Miransari M, Smith DL (2014) Plant hormones and seed germination. Environ Exp Bot 99:110–121
Mirshekari B, Baser S, Allahyari S, Hamedanlu N (2012) On-farm seed priming with Zn+ Mn is an effective way to improve germination and yield of marigold. Afr J Microbiol Res 6(28):5796–5800
Mohd Din ARJ, Cheng KK, Sarmidi MR (2017) Assessment of compost extract on yield and phytochemical contents of Pak Choi (Brassica Rapa cv. Chinensis) grown under different fertilizer strategies. Commun Soil Sci Plant Anal 48(3):274–284
Moradi A, Younesi O (2009) Effects of osmo-and hydro-priming on seed parameters of grain sorghum (Sorghum bicolor L.). Aust J Basic Appl Sci 3(3):1696–1700
Naeem M, Bhatti IRAM, Ahmad RH, Ashraf MY (2004) Effect of some growth hormones (GA3, IAA and kinetin) on the morphology and early or delayed initiation of bud of lentil (Lens culinaris Medik). Pak J Bot 36(4):801–809
Narwal SS (1994) Allelopathy in crop production. Scientific Publishers, Jodhpur, p 288
Nayban G, Mandal AK, De BK (2017) Seed priming: a low-cost climate-resilient tool for improving germination, growth and productivity of mungbean. SATSA Mukhaptra Annu Tech Issue 21:162–172
Niinemets Ü (2009) Mild versus severe stress and BVOCs: thresholds, priming and consequences. Trends Plant Sci 15(3):145–153
O’Sullivan JOHN, Bouw WJ (1984) Pepper seed treatment for low-temperature germination. Can J Plant Sci 64(2):387–393
Ozbay N, Susluoglu Z (2016) Assessment of growth regulator prohexadione calcium as priming agent for germination enhancement of pepper at low temperature. JAPS: J Anim Plant Sci 26(6):1652–1658
Pame AR, Kreye C, Johnson D, Heuer S, Becker M (2015) Effects of genotype, seed P concentration and seed priming on seedling vigor of rice. Exp Agric 51(03):370–381
Passam HC, Karavites PI, Papandreou AA, Thanos CA, Georghiou K (1989) Osmo-conditioning of seeds in relation to growth and fruit yield of aubergine, pepper, cucumber and melon in unheated greenhouse cultivation. Sci Hortic 38(3–4):207–216
Patanè C, Cavallaro V, D’Agosta G, Cosentino SL (2008) Plant emergence of PEG-osmoprimed seeds under suboptimal temperatures in two cultivars of sweet sorghum differing in seed tannin content. J Agron Crop Sci 194(4):304–309
Pereira MD, Dias DCFDS, Dias LADS, Araújo EF (2009) Primed carrot seeds performance under water and temperature stress. Sci Agric 66(2):174–179
Pill WG (1995) Low water potential and presowing germination treatments to improve seed quality. In: Seed quality, pp 319–359
Pozo MJ, Azcón-Aguilar C (2007) Unraveling mycorrhiza-induced resistance. Curr Opin Plant Biol 10(4):393–398
Prabha D, Negi S, Kumari P, Negi YK, Chauhan JS (2016) Effect of seed priming with some plant leaf extract on seedling growth characteristics and root rot disease in tomato. Int J Agric Syst 4(1):46–51
Prasad TNVKV, Sudhakar P, Sreenivasulu Y, Latha P, Munaswamy V, Reddy KR et al (2012) Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J Plant Nutr 35(6):905–927
Qi W, Zhang L, Wang L, Xu H, Jin Q, Jiao Z (2015) Pretreatment with low-dose gamma irradiation enhances tolerance to the stress of cadmium and lead in Arabidopsis thaliana seedlings. Ecotoxicol Environ Saf 115:243–249
Raj SN, Shetty NP, Shetty HS (2004) Seed bio-priming with Pseudomonas fluorescens isolates enhances growth of pearl millet plants and induces resistance against downy mildew. Int J Pest Manag 50(1):41–48
Rakshit A, Pal S, Rai S, Rai A, Bhowmick MK, Singh HB (2013) Micronutrient seed priming: a potential tool in integrated nutrient management. SATSA Mukhaptra Annu Tech Issue 17:77–89
Rakshit A, Sunita K, Pal S, Singh A, Singh HB (2015) Bio-priming mediated nutrient use efficiency of crop species. In: Nutrient use efficiency: from basics to advances. Springer, New Delhi, pp 181–191
Rangaswamy A, Purushothaman S, Devasenapathy P (1993) Seed hardening in relation to seedling quality characters of crops. Madras Agric J 80(9):535–537
Raphael E (2012) Phytochemical constituents of some leaves extract of Aloe vera and Azadirachta indica plant species. Glob Adv Res J Environ Sci Toxicol 1(2):014–017
Reddy PP (2012) Bio-priming of seeds. In: Recent advances in crop protection. Springer, New Delhi, pp 83–90
Reddy MVB, Arul J, Angers P, Couture L (1999) Chitosan treatment of wheat seeds induces resistance to Fusarium graminearum and improves seeds quality. J Agric Food Chem 47:67–72
Rehman H, Nawaz Q, Basra SMA, Afzal I, Yasmeen A (2014) Seed priming influence on early crop growth, phenological development and yield performance of linola (Linum usitatissimum L.). J Integr Agric 13(5):990–996
Renugadevi J, Vijayageetha V (2006) Organic seed fortification in cluster bean (Cyamopsistetra gonoloba L.) TAUB. In: International conference on indigenous vegetables and legumes. Prospectus for fighting poverty, hunger and malnutrition, vol 752, December, pp 335–337)
Rowse HR (1991) Methods of priming seeds. UK Patent, 2(192), 781
Ruan SL, Xue QZ (2002) Effects of chitosan coating on seed germination and salt-tolerance of seedlings in hybrid rice (Oryza sativa L.). Acta Agron Sin 28:803–808
Sajedi NA, Ardakani MR, Madani H, Naderi A, Miransari M (2011) The effects of selenium and other micronutrients on the antioxidant activities and yield of corn (Zea mays L.) under drought stress. Physiol Mol Biol Plants 17(3):215–222
Salehi M, Tamaskani F (2008) Pretreatment effect of nanosilver on germination and seedling growth of wheat under salt stress. In: Proceeding of 11th Iranian congress in seed sciences and echnology. Gorgan, Iran
Satish S, Mohana DC, Ranhavendra MP, Raveesha KA (2007) Antifungal activity of some plant extracts against important seed borne pathogens of Aspergillus sp. Int J Agric Technol 3(1):109–119
Scheuerell SJ, Mahaffee WF (2006) Variability associated with suppression of graymold (Botrytis cinerea) on geranium by foliar applications of nonaerated and aerated compost teas. Plant Dis 90:1201–1208
Schwember AR, Bradford KJ (2005) Drying rates following priming affect temperature sensitivity of germination and longevity of lettuce seeds. Hort Sci 40(3):778–781
Shahrokhi M, Tehranifar A, Hadizadeh H, Selahvarzi Y (2011) Effect of drought stress and paclobutrazol-treated seeds on physiological response of Festuca arundinacea L. Master and Lolium perenne L. Barrage. J Biol Environ Sci 5(14):77–85
Shao CX, Hu J, Song WJ, Hu WM (2005) Effects of seed priming with chitosan solutions of different acidity on seed germination and physiological characteristics of maize seedling. J Zhejiang Univ (Agric Life Sci) 31(6):705–708
Shivay YS, Singh U, Prasad R, Kaur R (2016) Agronomic interventions for micronutrient biofortification of pulses. Indian J Agron 61(4th IAC Special Issue):161–172
Singh MV (2007) Efficiency of seed treatment for ameliorating zinc deficiency in crops. Zinc Crops 24–26
Singh A, Dahiru R, Musa M, Sani Haliru B (2014) Effect of Osmopriming duration on germination, emergence, and early growth of Cowpea (Vigna unguiculata (L.) Walp.) in the Sudan Savanna of Nigeria. Int J Agron 2014:1–4
Singh H, Jassal RK, Kang JS, Sandhu SS, Kang H, Grewal K (2015a) Seed priming techniques in field crops-a review. Agric Rev 36(4):251–264
Singh U, Praharaj CS, Shivay YS, Kumar L, Singh SS (2015b) Ferti-fortification: an agronomic approach for micronutrient enrichment of pulses. In Pulses: challenges and opportunities under changing climatic scenario, In: Proceedings of the national conference on “Pulses: challenges and opportunities under changing climatic scenario, vol. 29, pp 208–222
Slaton NA, Wilson CE, Ntamatungiro S, Norman RJ, Boothe DL (2001) Evaluation of zinc seed treatments for rice. Agron J 93(1):152–157
Sliwinska E, Jendrzejczak E (2002) Sugar-beet seed quality and DNA synthesis in the embryo in relation to hydration-dehydration cycles. Seed Sci Technol 30(3):597–608
Srivastava AK, Bose B (2012) Effect of nitrate seed priming on phenology, growth rate and yield attributes in rice (Oryza sativa L.). Vegetos Int J Plant Res 25(2):174–181
Steel RGD, Torrie JH, Dickey D (1997) Principles and procedures of statistics: a biometrical approach, 3rd edn. McGraw Hill Book Company, New York, pp 172–177
Sung JM, Chiu KY (1995) Hydration effect on seedling emergence strength of watermelon seeds differing in ploidy. Plant Sci 110(1):21–26
Tavili A, Zare S, Enayati A (2009) Hydropriming, ascorbic and salicylic acid influence on germination of Agropyro nelongatum host. Seeds under salt stress. Res J Seed Sci 2(1):16–22
Taylor AG, Allen PS, Bennett MA, Bradford KJ, Burris JS, Misra MK (1998) Seed enhancements. Seed Sci Res 8(2):245–256
Thakur P, Kumar S, Malik JA, Berger JD, Nayyar H (2010) Cold stress effects on reproductive development in grain crops: an overview. Environ Exp Bot 67(3):429–443
Tonelli ML, Furlan A, Taurian T, Castro S, Fabra A (2011) Peanut priming induced by biocontrol agents. Physiol Mol Plant Pathol 75(3):100–105
Uchida A, Jagendorf AT, Hibino T, Takabe T, Takabe T (2002) Effects of hydrogen peroxide and nitric oxide on both salt and heat stress tolerance in rice. Plant Sci 163(3):515–523
Umair A, Ali S, Hayat R, Ansar M, Tareen MJ (2011) Evaluation of seed priming in mung bean (Vigna radiata) for yield, nodulation and biological nitrogen fixation under rainfed conditions. Afr J Biotechnol 10(79):18122–18129
Umair A, Ali S, Sarwar M, Bashir K, Tareen MJ, Malik MA (2013) Assessment of some priming techniques in mungbean (Vigna radiata): a green house study. Pak J Agric Res 26:4
Upadhyaya H, Begum L, Dey B, Nath PK, Panda SK (2017) Impact of calcium phosphate nanoparticles on rice plant. J Plant Sci Phytopathol 1:1–10
Van Hulten M, Pelser M, Van Loon LC, Pieterse CM, Ton J (2006) Costs and benefits of priming for defense in Arabidopsis. Proc Natl Acad Sci 103(14):5602–5607
Varier A, Vari AK, Dadlani M (2010) The subcellular basis of seed priming. Curr Sci 99:450–456
Wahid A, Noreen A, Basra SM, Gelani S, Farooq M (2008) Priming-induced metabolic changes in sunflower (Helianthus annuus) achenes improve germination and seedling growth. Bot Stud 49(4):343–350
Waqas M, Nizami AS, Aburiazaiza AS, Barakat MA, Ismail IMI, Rashid MI (2017) Optimization of food waste compost with the use of biochar. J Environ Manag. https://doi.org/10.1016/j.jenvman.2017.06.015
Warren JE, Bennett MA (1997) Seed hydration using the drum priming system. Hort Sci 32(7):1220–1221
Weltzien HC (1991) Biocontrol of foliar fungal disease with compost extracts. In: Andrews JH, Hirano SS (eds) Microbial ecology of leaves. Springer-Verlag, New York, pp 430–450
Wright B, Rowse H, Whipps JM (2003) Microbial population dynamics on seeds during drum and steeping priming. Plant Soil 255(2):631–640
Xu S, Hu J, Li Y, Ma W, Zheng Y, Zhu S (2011) Chilling tolerance in Nicotiana tabacum induced by seed priming with putrescine. Plant Growth Regul 63(3):279–290
Yadav PV, Kumari M, Ahmed Z (2011) Seed priming mediated germination improvement and tolerance to subsequent exposure to cold and salt stress in capsicum. Res J Seed Sci 4(3):125–136
Yan M (2015) Seed priming stimulate germination and early seedling growth of Chinese cabbage under drought stress. S Afr J Bot 99:88–92
Yogananda DK, Vyakarnahal BS, Shekhargouda M (2004) Effect of seed invigoration with growth regulations and micronutrients on germination and seedling vigour of bell pepper cv. California Wonder. Karnataka J Agri Sci 17(4):811–813
Zhou ZS, Guo K, Elbaz AA, Yang ZM (2009) Salicylic acid alleviates mercury toxicity by preventing oxidative stress in roots of Medicago sativa. Environ Exp Bot 65(1):27–34
Zhou J, Wang Y, Jahufer Z (2013) Location and chemical composition of semi-permeable layer of forage seeds. Bangladesh J Bot 42(1):23–30
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Waqas, M. et al. (2019). Advances in the Concept and Methods of Seed Priming. In: Hasanuzzaman, M., Fotopoulos, V. (eds) Priming and Pretreatment of Seeds and Seedlings. Springer, Singapore. https://doi.org/10.1007/978-981-13-8625-1_2
Download citation
DOI: https://doi.org/10.1007/978-981-13-8625-1_2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8624-4
Online ISBN: 978-981-13-8625-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)