Abstract
The exact pathogenesis of adenomyosis is still elusive. Among different reported concepts, direct invagination of gland cells from basalis endometrium deep into myometrium is the most widely accepted opinion in the development of adenomyosis. To address the mechanistic basis of this accepted concept, we investigated the role of hepatocyte growth factor (HGF) and estrogen in the occurrence of epithelial-mesenchymal transition (EMT) in human adenomyosis. Biopsy specimens from endometrium to myometrium were collected after hysterectomy from women with and without adenomyosis. The relationship between HGF and E-cadherin (epithelial cell marker)/N-cadherin (mesenchymal cell marker) was examined using endometrial epithelial cells (EECs) and tissues by qRT-PCR and immunohistochemistry. The gene and protein expressions of two transcriptional repressors of E-cadherin, SLUG and SNAIL, were examined using Ishikawa cells. HGF downregulated E-cadherin and upregulated N-cadherin mRNA expression in EECs, and an inverse relationship between HGF and E-cadherin was observed in basalis endometria of women with adenomyosis. HGF induced morphological changes and promoted migration of EECs. Ishikawa cells exhibited upregulation of SLUG/SNAIL gene expression in response to HGF and estrogen with an additive effect between them. HGF- and estrogen-promoted SLUG/SNAIL gene expression was significantly abrogated after pretreatment of cells with anti-HGF antibody or ICI 182720, an estrogen receptor antagonist. Our findings suggested that HGF either alone or in combination with estrogen may be involved in gland invagination deep into myometrium by inducing EMT in women with adenomyosis.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
9.1 Introduction
Adenomyosis is defined by the intramyometrial presence of gland and stroma derived from basalis endometrium surrounded by reactive hyperplastic or hypertrophic myometrium causing either focal or diffuse enlargement of the uterus [1, 2]. Adenomyosis occurs most likely during the fourth and fifth decades of life and after the completion of childbearing activity. However, recent development of diagnostic tools such as transvaginal ultrasonography and magnetic resonance imaging has disclosed that adenomyosis may occur in women of younger ages. The differences between endometriosis and adenomyosis are origin of endometrium (functionalis or basalis) and anatomic site of the lesion (outside or inside of the uterus) [2].
The clinical symptoms of adenomyosis are dysmenorrhea, menorrhagia, chronic pelvic pain, and subfertility and need proper treatment. Hysterectomy is the definitive treatment. In cases in which adenomyosis is located focally in either one of the uterine wall, uterine-preservation surgery such as adenomyomectomy is an alternative surgery that is applicable to those who desire to preserve their reproductive potential [3].
The exact physiopathology and pathogenesis of adenomyosis are still elusive. According to published literatures, there are different proposals for the pathogenesis of adenomyosis, such as de novo metaplasia of Müllerian remnants, invagination through vascular or lymphatic channel, and mechanical tissue injury or stress reaction at the endo-myometrial interface [1, 2, 4] (Fig. 9.1). The most widely accepted opinion is that adenomyosis develops as a downgrowth and invagination of the basalis endometrium into the myometrium [1]. However, a possible mechanism of gland invagination from basalis endometrium deep into underlying myometrium is unknown. Moreover, information on the biological differences between functionalis and basalis endometria, the two compartments of human endometrium, is still lacking.
Epithelial-mesenchymal transition (EMT) and its converse, mesenchymal-epithelial transition (MET), are concepts first defined by Elizabeth Hay some 45 years ago [5]. Once epithelial cells become competent to respond to EMT-inducing signals, these signals can promote disruption of the intercellular adhesion complexes and loss of the apicobasal polarity of the epithelial cells, a prime feature crucial for cells to leave the epithelium and achieve migration potentiality. Since, basalis endometrium and inner myometrium (junctional zone) are closely apposed without any intervening basement membrane, events of EMT might occur here.
E-cadherin is a multigene family of transmembrane glycoproteins and normally acts as an “adhesive zipper” that means E-cadherin maintains tight cell-cell or cell-matrix contact for epithelial cells [6]. It has been demonstrated that epithelial cells, potentially invasive in human endometriosis, lack the expression of E-cadherin [7]. Studies with breast cancer, bladder cancer, and hepatoma cells indicated that overexpression of hepatocyte growth factor (HGF) may downregulate cadherin-mediated cell-cell adhesion with consequent migration and invasion into mesenchymal cells [8]. Recent reports demonstrated that estrogen-induced epithelial-mesenchymal transition (EMT) of epithelial cells may contribute to the development of adenomyosis or may be involved in metastatic potential of cancer cells [9, 10]. As an estromedin and pleiotropic growth factor, we speculated that similar to estrogen, HGF might play a similar role in EMT and in the invasion of gland cells into myometrium in women with adenomyosis.
A number of transcriptional repressors of E-cadherin such as SLUG (a Snail-related zinc-finger transcription factor), SNAIL (another Snail-related zinc-finger transcription factor), SIP-1/Zeb-2, E12/E47, and Twist have been reported [10,11,12]. They are found to interact with the proximal E-boxes of E-cadherin promoter and induce DNA hypermethylation or histone deacetylation thereby causing decreased expression of E-cadherin [11, 12]. An inverse correlation between E-cadherin and SLUG/SNAIL expression has been described in different cell systems [10,11,12]. Ectopic expression of SLUG/SNAIL in epithelial cells caused EMT by the aid of migratory and invasive behaviors [11, 12]. The expression pattern of SLUG/SNAIL in epithelial cells in response to HGF and the additive effect between HGF and estrogen on their expression are not clearly described. We performed a series of experiments to establish that as an estromedin (regulated by estrogen) and pleiotropic growth factor, HGF might play a role in inducing EMT, and if it is true, HGF-induced EMT might be involved in the pathogenesis of adenomyosis.
9.2 Biological Differences Between Functionalis and Basalis Endometria
While endometriosis originates from the functionalis endometrium, origin of adenomyosis is from the basalis endometrium. Therefore, it is important to know the biological/functional differences between these two compartments of endometrium. Basalis endometrium can be distinguished from functional endometrium by defined histological criteria: (1) constant appearance of basalis throughout the menstrual cycle, (2) glands of basalis appear weakly proliferative, (3) basalis cells lack secretory change and the stroma is spindled and non-decidualized, and (4) 1 mm area from the endo-myometrial junction as an additional criteria independent of menstrual cycle [13, 14].
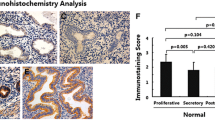
Considering biological differences, ER and PR expression was equally observed in gland cells and stromal cells of both functional endometrium and basalis endometrium. While there was no remarkable difference in the expression of ER and PR in these two layers of endometrium in the proliferative phase, a weak expression of ER and PR was observed in the basal layer during the secretory phases of women with adenomyosis and control women [15]. A strong expression of Ki-67 was observed in functionalis endometrium of proliferative phase and a weak expression in basalis endometrium throughout the menstrual cycle. The TUNEL-positive apoptotic cells were least in the proliferative phase, modest in the secretory phase, and highest in the menstrual phase as observed in the functionalis endometrium. A few TUNEL-positive apoptotic cells were observed in the basalis endometrium. A similar pattern of activated caspase-3 immunoexpression was found in the functionalis and basalis endometrium throughout the menstrual cycle [15]. These findings confirmed that the two compartments of endometrium are biologically different. A mixture of blood and tissues from the necrotic functionalis endometria are the constituents of the antegrade flow of menstruation. The surviving basalis endometria consequently may suffer a constant tissue stress reaction in response to cyclic breakdown of functionalis endometria.
9.3 Stress Reaction at the Endo-myometrial Interface
When we consider cyclic shedding of functionalis endometrium, cyclic tissue breakdown during menstrual phase may induce a constant stress reaction on or cause tissue injury at the endo-myometrial junction. In fact, as a marker of tissue stress reaction, we previously demonstrated higher expression of human heat shock protein 70 (HSP70) in eutopic/ectopic endometria and peritoneal fluid of women with endometriosis during the secretory phase and the menstrual phase of the cycle [16, 17]. Increased tissue expression of stress-reactive proteins including HSP70 may also occur at the endo-myometrial junction as a result of repeated curettage after abortion or after multiple pregnancies that have been traditionally considered as high-risk factors for adenomyosis [1].
By immunohistochemical analysis, we observed a strong immunoexpression of HSP70 in basalis endometrium (gland+stroma) as well as in inner myometrium in women with adenomyosis than in control women. A similar moderate to strong HSP70 expression was found in glands cells and myometrium of both diffuse and focal adenomyotic lesions. A significantly higher quantitative-histogram (Q-H) score of HSP70 immunoreaction was found at the endo-myometrial junction (EMJ) of women with adenomyosis than in control women and in adenomyotic lesions of women with diffuse adenomyosis than in focal adenomyosis [18]. This indicates that an area of endo-myometrial interface may suffer from a constant tissue stress reaction in women with adenomyosis.
9.4 Effect of HGF on Basal EECs and E-Cadherin/N-Cadherin Expression
Expression of c-Met, a receptor of HGF, in endometrial epithelial cells (EECs) has already been described in a separate study [19]. A cell separation/scattering effect on cytokeratin-positive pre-confluent EECs was observed after treatment with HGF (at 50 and 100 ng/mL). A time-dependent increase in the mRNA expression of E-cadherin in basal EECs (HGF-untreated) was found with maximum gene expression at 24 and 48 h. This could be associated with cell-cell contact of EECs in response to overexpression of E-cadherin. Treatment with HGF on pre-confluent EECs significantly decreased mRNA expression of E-cadherin (epithelial cell marker) and induced concomitant increase in the mRNA expression of N-cadherin (mesenchymal cell marker) [18]. These differential expressions of E-cadherin and N-cadherin with consequent cell separation are the basic features of HGF-induced EMT. In fact, downregulation of E-cadherin resulting in the disruption of cell-cell contact is one of the components of EMT.
9.5 Expression of HGF, E-Cadherin/N-Cadherin in Functionalis and Basalis Endometria
A similar pattern of immunoexpression of HGF and E-cadherin was observed in gland cells of both functionalis and basalis endometria derived from control women. While no staining of N-cadherin was observed in either of these two layers, Vimentin (another mesenchymal cell marker) expression was increased. The protein expressions of HGF/E-cadherin/N-cadherin/Vimentin were equally observed in the functionalis layer of the endometrium derived from women with diffuse adenomyosis. However, a stronger expression of HGF and mild expression of E-cadherin was found in the basalis endometrium of women with diffuse adenomyosis. Comparing to expression of N-cadherin, a stronger expression of Vimentin was observed in functionalis/basalis endometrium [18].
A similar pattern of immunoexpression of HGF and E-cadherin was observed in the functionalis and basalis endometria derived from women with focal adenomyosis. While no difference in the expression of HGF and E-cadherin was observed in the functionalis and basalis endometria of contralateral side (no lesion) and functionalis layer of ipsilateral side (with lesion), a decreased expression of E-cadherin and moderate expression of HGF was observed in the basalis endometrium of ipsilateral side. That was confirmed by Q-H score analysis that showed a significantly more decreased expression of E-cadherin in the basalis layer than in the functionalis layer of the endometrium on the ipsilateral side [18]. It was interesting to observe that inverse relationship between HGF and E-cadherin was lost once adenomyotic lesion develops. This might be due to the hyperplastic or hypertrophic changes of surrounding smooth muscle cells causing realignment of migrating endometrial cells into glandular structure.
A constant tissue stress insult as manifested by higher expression of HSP70 in the basalis endometrium or in the inner myometrium may trigger the very early switch of EMT by higher expression of HGF and consequent lower expression of E-cadherin in the basalis endometrium. In fact, biological events of EMT at tissue level and morphological recognition of adenomyosis by image are different. We clinically diagnose adenomyosis by subjective and objective findings of each patient. Therefore, failure to diagnose adenomyosis by image cannot exclude the absence of biological events (stress reaction/EMT) at the tissue level. A silent variable degree of cyclic tissue stress reaction may occur at the endo-myometrial junction even in control women. Some of these women may time-dependently develop adenomyosis to be recognized either by histology or by image. We believe that the cause-effect of EMT in adenomyosis is still an open question for future investigation.
9.6 Tissue Concentration of HGF at the Endo-myometrial Junction
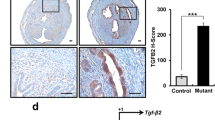
We collected tissue specimens from endo-myometrial interface after hysterectomy of women had operated for uterine myoma (control) and for focal and diffuse adenomyosis. These tissue specimens were cut into minute pieces and homogenized using Polytron homogenizer and tissue concentration of HGF was measured by ELISA. A significantly higher tissue content of HGF was found at the endo-myometrial interface derived from ipsilateral side of focal adenomyosis and anterior/posterior walls of diffuse adenomyosis when compared with similar tissues derived from control women or from contralateral endometrium of focal adenomyosis [18]. The gland cells, stromal cells, smooth muscle cells of the endometrium and myometrium as well as different immune cells have been reported to produce and secrete HGF in tissues and body fluids of women with different reproductive diseases [20, 21].
9.7 Effect of HGF/Estrogen on Changes in Cell Morphology and Cell Migration
We examined two other key cellular components of EMT in response to HGF and E2 such as cell migration and cell morphological change. Boyden’s chamber assay revealed that HGF was able to significantly migrate EECs from upper chamber to lower chamber with a dose of both 50 and 100 ng/mL. The expression of ER and PR in endometrial and endometriotic cells has been described previously [22, 23]. In addition to significant single effect of E2, an additive between HGF and E2 was observed in cell migration. This effect of HGF and E2 on cell migration was significantly abrogated after pretreatment of EECs with anti-HGF antibody or ICI 182720, an ER antagonist. Treatment with non-immune mouse IgG + HGF did not influence this migration. We found that HGF (100 ng/mL) induced a sequential and time-dependent change in EECs morphology, from cobblestone-like appearance (at 0 h) to a mesenchymal phenotype (elongated spindle-shaped cells) at 48 h [18]. Estrogen had similar effect on morphological change of EECs. These findings give us further information that in addition to function as estromedin growth factor (regulated by E2) [22] or as a pleiotropic growth factor [24] in endometriosis, HGF also exhibits its potential capacity to induce EMT in adenomyosis.
9.8 Effect of HGF and E2 on SLUG and SNAIL Expression in Ishikawa Cells
Ishikawa cells are a well-differentiated endometrial adenocarcinoma cell line that retains phenotype of endometrial epithelial cells and displays apical adhesiveness and a similar expression profile of different molecules to endometrium, under the control of estrogen and progesterone [25, 26]. In fact, different transcriptional repressors of E-cadherin (SLUG/SNAIL/Twist) are known to express in Ishikawa cells [27]. We used Ishikawa cells to investigate expression pattern of SLUG and SNAIL in response to HGF and estrogen. Both basal and estrogen (E2)-stimulated and ER-positive Ishikawa cells displayed c-Met (HGF receptor) expression. Individual treatment of Ishikawa cells with HGF and E2 upregulated mRNA expression of SLUG and SNAIL comparing to HGF/E2-untreated cells. In addition to individual significant effect, an additive effect between HGF and E2 was observed in the overexpression of SLUG and SNAIL mRNA. The individual effect of HGF and E2 was more prominent in the expression of SLUG gene comparing to SNAIL gene. HGF- and E2-promoted SLUG and SNAIL mRNA expression were significantly suppressed after pretreatment of cells with anti-HGF antibody or ICI 182720, an ER antagonist [18].
9.9 Expression of E-Cadherin and SLUG/SNAIL in Functionalis and Basalis Endometria
A similar pattern of E-cadherin and SLUG immunostaining was observed in both functionalis and basalis endometria derived from contralateral side (no lesion) and in functionalis endometrium derived from ipsilateral side (with lesion) of focal adenomyosis. In contrast, a mild expression of E-cadherin and moderate expression of SLUG was found in the basalis endometrium derived from ipsilateral side (with lesion) of focal adenomyosis [18]. SNAIL immunostaining was not observed in the functionalis or basalis endometria of either side of focal adenomyosis. Comparing to functionalis layer, a significantly decreased Q-H scores of E-cadherin was observed in the basalis layer with higher expression of SLUG in the ipsilateral side [18]. Similar to HGF and E-cadherin expression, no obvious difference in the expression of E-cadherin and SLUG was observed in gland cells of adenomyotic lesions. These results demonstrated that overexpression of SLUG with consequent downregulation of E-cadherin in response to HGF and estrogen may be involved in the disruption of cell-cell contact. This HGF-mediated cell separation effect together with increased cell migration/invasion and cell morphological change in the endometrium may be responsible for the gland invagination deep into the myometrium in women with adenomyosis.
9.10 Summary and Perspective
We demonstrated here that there are some fundamental biological differences between functional endometria and basal endometria of women with adenomyosis. Less ovarian steroid response and less apoptosis of cells of basal endometria indicate that there is substantial survival of cells in this compartment of endometria across the phases of the menstrual cycle. When there is mechanical tissue injury or tissue stress reaction at the endo-myometrial interface, these surviving cells of basal endometria may escape deep into myometrium resulting in the development of adenomyosis.
In an attempt to explore the mechanistic basis of this phenomenon, here we demonstrated a possible involvement of HGF-induced EMT in the pathogenesis of adenomyosis. A tissue stress insult may explain higher expression of HSP70 in the basalis endometrium or in the inner myometrium and may trigger the very early switch of EMT by higher expression of HGF and consequent lower expression of E-cadherin in the basalis endometrium. Unlike all previous studies, we used isolated epithelial cells and full thickness endometrium to myometrial tissues derived from women with both focal and diffuse adenomyosis after hysterectomy.
We found that higher tissue concentrations of HGF at the endo-myometrial interface triggered a sequential cascade of EMT such as cell migration and cell morphological changes (EECs) to a mesenchymal phenotype. Our findings of overexpression of HGF (EMT-inducing signal) and downregulation of E-cadherin with consequent disruption of tight cell-cell contact of EECs and resulting migration of EECs may be the basic components in the event of EMT in adenomyosis. The effect of HGF may be further supported by the action of local estrogen. Here we found that less expression of E-cadherin in response to HGF was well corresponded with migration through porous filters of EECs. Previous reports calculated the invasion capacity of EECs in relation to their motility taking into account that invasion also depends on motility [28, 29].
The event of HGF in EMT was mediated by upregulation of SLUG/SNAIL (transcriptional repressors of E-cadherin) in Ishikawa cells and downregulation of E-cadherin, an epithelial cell marker, and upregulation of N-cadherin/Vimentin, two mesenchymal cell markers. According to published literatures, lower tissue expression of E-cadherin and increased tissue expression of Vimentin or N-cadherin dictates a transitional cascade from an epithelial cell phenotype to mesenchymal cell phenotype [8, 21]. In other words, a phenomenon of EMT that was reported to be involved in the metastatic invasion of cancer cells [8] might also work in the pathogenesis of adenomyosis. We found that HGF either alone or in combination with estrogen was able to induce morphological changes of EECs from cobblestone appearance to spindle-shaped fibroblast-like cells. The functional specificity of HGF and E2 was confirmed by their neutralizing effect on EECs motility. These findings give us further information that in addition to function as estromedin growth factor (regulated by E2) [22] or as pleiotropic growth factor [23] in endometriosis, HGF also exhibits its potential capacity to induce EMT in adenomyosis.
It was interesting to observe that inverse relationship between HGF and E-cadherin was lost once adenomyotic lesion develops. This might be due to the hyperplastic or hypertrophic changes of surrounding smooth muscle cells causing realignment of migrating endometrial cells into glandular structure. A diagrammatic representation of the cascade of EMT in response to HGF and estrogen that may result in the development of adenomyosis is shown in Fig. 9.2.
A diagrammatic representation showing hepatocyte growth factor (HGF)- and estrogen (E2)-induced pathways in the occurrence of epithelial-mesenchymal transition (EMT) in human adenomyosis. Upregulation of SLUG and SNAIL, two transcriptional repressors of E-cadherin, in response to HGF and E2 is associated with decreased expression of E-cadherin (epithelial cell marker) and increased expression of N-cadherin/Vimentin (mesenchymal cell markers) causing disruption of tight cell-cell contact. These cellular events in endometrial cells and in intact tissues trigger morphological changes of endometrial epithelial cells (EECs) to a mesenchymal phenotype and induced increased cell migration, two essential components of EMT that was induced by HGF and E2 either alone or in combination. All these events of EMT may be involved in the invagination of glandular epithelial cells from basalis endometrium deep into the myometrium and finally result in the development of adenomyosis
We conclude that similar to estrogen [9], HGF may be involved in gland invagination deep into myometrium by inducing EMT at the endo-myometrial junction in women with adenomyosis. Since there is no basement membrane in between basal endometrium and inner myometrium (EMJ), the process of EMT can be better initiated here in response to tissue stress insult and/or local tissue inflammation and higher tissue content of HGF. Estrogen-induced EMT was found to be abrogated after treatment with raloxifene, a selective estrogen receptor modulator [9]. The findings of our current hypothesis warrant further investigation to examine the pattern of changes in the biological events of EMT in adenomyosis after treatment with estrogen-suppressing agent. Further studies are needed to strengthen our current findings.
References
Bergeron C, Amant F, Ferenczy A. Pathology and pathophysiology of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4):511–21.
Ferenczy A. Pathophysiology of adenomyosis. Hum Reprod Update. 1998;4(4):312–22.
Wang PH, Fuh JL, Chao HT, Liu WM, Cheng MH, Chao KC. Is the surgical approach beneficial to subfertile women with symptomatic extensive adenomyosis? J Obstet Gynecol Res. 2009;35:495–502.
Ridley JH. The histogenesis of endometriosis: a review of facts and fancies. Obstet Gynecol Surv. 1968;23:1–35.
Hay ED. Organization and fine structure of epithelium and mesenchyme in the development of chick embryo. In: Fleischmajer R, Billingham RE, editors. Epithelial-mesenchymal interactions. Baltimore: Williams & Wilkins; 1968. p. 31–55.
Shapiro L, Fannon AM, Kwong PD, Thompson A, Lehmann MS, Grubel G, Legrand JF, Als-Nielsen J, Colman DR, Hendrickson WA. Structural basis of cell-cell adhesion by cadherins. Nature. 1995;374:327–37.
Gaetje R, Kotzian S, Herrmann G, Baumann R, Starzinski-Powitz A. Nonmalignant epithelial cells, potentially invasive in human endometriosis, lacks the tumor suppressor molecule E-cadherin. Am J Pathol. 1997;150:461–7.
Hiscox S, Jiang WG. HGF/SF regulates the phosphorylation of β-catenin and cell-cell adhesion in cancer cells. Proc Am Assoc Cancer Res. 1998;39:500–1.
Chen YJ, Li HY, Huang CH, Twu NF, Yen MS, Wang PH, Chou TY, Liu YN, Chao KC, Yang MH. Estrogen-induced epithelial-mesenchymal transition of endometrial epithelial cells contributes to the development of adenomyosis. J Pathol. 2010;222:261–70.
Park SH, Cheung LWT, Wong AST, Leung PCK. Estrogen regulates Snail and Slug in the down-regulation of E-cadherin and induces mestastatic potential of ovarian cancer cells through estrogen receptorα. Mol Endocrinol. 2008;22:2085–98.
Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, Portillo F, Nieto MA. The transcriptional factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol. 2000;2:76–83.
Bolos V, Peinado H, Perez-Moreno MA, Fraga MF, Esteller M, Cano A. The transcription factor Slug represses E-cadherin expression and induces epithelial to mesenchymal transitions: a comparison with Snail and E47 repressors. J Cell Sci. 2003;116:499–511.
Hendrickson MR, et al. Normal histology of the uterus and fallopian tubes. In: Sternberg SS, editor. Histology for pathologists. 2nd ed. Philadelphia: Lippincott-Raven; 1997. p. 894–7.
Setoguchi T. Histology of endometrium (in Japanese). In: Suzuku S, editor. Textbook of practical histology. Tokyo: Nanzando Co. Ltd.; 1979. p. 218–41.
Khan KN, Fujishita A, Kitajima M, Masuzaki H, Nakashima M, Kitawaki J. Biological differences between functionalis and basalis endometria in women with and without adenomyosis. Eur J Obstet Gynecol Reprod Biol. 2016;203:49–55.
Khan KN, Kitajima M, Imamura T, Hiraki K, Fujishita A, Sekine I, Ishimaru T, Masuzaki H. Toll-like receptor 4 (TLR4)-mediated growth of endometriosis by human heat shock protein 70 (Hsp70). Hum Reprod. 2008;23(10):2210–9.
Imamura T, Khan KN, Fujishita A, Kitajima M, Hiraki K, Ishimaru T, Masuzaki H. Effect of GnRH agonist therapy on the expression of human heat shock protein 70 in eutopic and ectopic endometria of women with endometriosis. Eur J Obstet Gynecol Reprod Biol. 2014;180:16–23.
Khan KN, Kitajima M, Hiraki K, Fujishita A, Nakashima M, Masuzaki H. Involvement of hepatocyte growth factor-induced epithelial-mesenchymal transition in human adenomyosis. Biol Reprod. 2015;92(2):35.
Khan KN, Masuzaki H, Fujishita A, Kitajima M, Sekine I, Ishimaru T. Immunoexpression of hepatocyte growth factor and c-Met receptor in the eutopic endometrium predicts the activity of ectopic endometrium. Fertil Steril. 2003;79(1):173–81.
Khan KN, Kitajima M, Hiraki H, Fujishita A, Sekine I, Ishimaru T, Masuzaki H. Immunopathogenesis of pelvic endometriosis: role of hepatocyte growth factor, macrophages and ovarian steroids. Am J Reprod Immunol. 2008;60:383–404.
Khan KN, Masuzaki H, Fujishita A, Kitajima M, Kohno T, Sekine I, Matsuyama T, Ishimaru T. Regulation of hepatocyte growth factor by basal and stimulated-macrophages in women with endometriosis. Hum Reprod. 2005;20:49–60.
Khan KN, Masuzaki H, Fujishita A, Kitajima M, Sekine I, Matsuyama T, Ishimaru T. Estrogen and progesterone receptor expression in macrophages and regulation of hepatocyte growth factor by ovarian steroids in women with endometriosis. Hum Reprod. 2005;20:2004–13.
Fujishita A, Nakane PK, Koji T, Masuzaki H, Chavez RO, Yamabe T, Ishimaru T. Expression of estrogen and progesterone in endometrium and peritoneal endometriosis: an immunohistochemical and in situ hybridization study. Fertil Steril. 1997;67:856–64.
Khan KN, Kitajima M, Fujishita A, Ishimaru T, Sekine I, Masuzaki H. Multifunctional role of hepatocyte growth factor in the development of pelvic endometriosis. WES E-J. 2008;13–20.
Hannan NJ, Paiva P, Dimitriadis E, Salamonsen LA. Models for study of human embryo implantation: choice of cell lines? Biol Reprod. 2010;82:235–45.
Castelbaum AJ, Ying L, Samkuti SG, Sun J, IIesanmi AO, Lessey BA. Characterization of integrin expression in a well differentiated endometrial adenocarcinoma cell line (Ishikawa). J Clin Endocrinol Metab. 1997;82:136–42.
Montserrat N, Mozos A, Llobet D, Dolcet X, Pons C, Garcia de Herreros A, Matias-Guiu X, Prat J. Epithelial to mesenchymal transition in early stage endometrioid endometrial carcinoma. Hum Pathol. 2012;43:632–43.
Zeitvogel A, Baumann R, Starzinski-Powitz A. Identification of an invasive, N-cadherin-expressing epithelial cell type in endometriosis using a new cell culture model. Am J Pathol. 2001;159:1839–52.
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA. Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Invest. 2009;119:1438–49.
Acknowledgment
This work was supported in part by Grants-in-Aid for Scientific Research (grants 21592101 and 24592474) from the Japan Society of the Promotion of Science (to K.N.K.).
I gratefully thank Dr. Akira Fujishita and Dr. Koichi Hiraki of Saikeikai Nagasaki Hospital and Dr. Michio Kitajima of Nagasaki University Hospital for their kind assistance in sample collection and Prof. Masahiro Nakashima for his experimental advice. K.N.K. designed and supervised the project and edited/wrote the complete draft.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Khan, K.N. (2018). Role of Epithelial-Mesenchymal Transition in Human Adenomyosis: A New Insight into Its Pathogenesis. In: Sugino, N. (eds) Uterine Fibroids and Adenomyosis. Comprehensive Gynecology and Obstetrics. Springer, Singapore. https://doi.org/10.1007/978-981-10-7167-6_9
Download citation
DOI: https://doi.org/10.1007/978-981-10-7167-6_9
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-7166-9
Online ISBN: 978-981-10-7167-6
eBook Packages: MedicineMedicine (R0)