Abstract
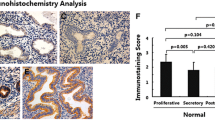
Epithelial—mesenchymal transition (EMT) has been reported to be involved in adenomyosis by promoting cell invasion and fibrogenesis. But few studies have identified critical factors that regulate EMT process during adenomyosis. The eukaryotic translation initiation factor 3 subunit e (eIF3e) protein is a component of the multisubunit eIF3 complex essential for cap-dependent translation initiation. The aim of this study was to investigate whether eIF3e is involved in EMT in adenomyosis. Ectopic endometrial tissue samples were collected from 40 premenopausal women with ultrasonographically diagnosed and histologically confirmed adenomyosis. As controls, endometrial samples were obtained from 40 cycling premenopausal women patients who underwent surgery for benign gynecologic disorders or cervical intraepithelial neoplasia but without endometriosis, adenomyosis, nor uterine fibroids. All tissue samples were subjected to immunohistochemistry analysis of eIF3e, transforming growth factor-β1 (TGF-β1), E-cadherin, vimentin, Snail, and proliferating cell nuclear antigen (PCNA). The epithelial component of ectopic endometrium showed significantly reduced immunoreactivity against eIF3e and E-cadherin but elevated immunoreactivity against TGF-β1, Snail, vimentin, and PCNA as compared with that of control endometrium (all P values <.05), and the difference was not affected by age, parity, or menstrual phase. The eIF3e staining levels correlated negatively with those of TGF-β1, vimentin, Snail, and PCNA (both P values <.05). These data suggest that decreased eIF3e expression may pave way for EMT in the development of adenomyosis through activating the TGF-β1 signaling pathway. Our study provided novel insights into the development and treatments of adenomyosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Benagiano G, Habiba M, Brosens I. The pathophysiology of uterine adenomyosis: an update. Fertil Steril. 2012;98(3):572–579.
Bergeron C, Amant F, Ferenczy A. Pathology and physiopathology of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4):511–521.
Farquhar C, Brosens I. Medical and surgical management of adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006;20(4):603–616.
Grow DR, Filer RB. Treatment of adenomyosis with long-term GnRH analogues: a case report. Obstet Gynecol. 1991;78(3 pt 2):538–539.
Shen M, Liu X, Zhang H, Guo SW. Transforming growth factor beta1 signaling coincides with epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation in the development of adenomyosis in mice. Hum Reprod. 2016;31(2):355–369.
Liu X, Shen M, Qi Q, Zhang H, Guo SW. Corroborating evidence for platelet-induced epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation in the development of adenomyosis. Hum Reprod. 2016;31(4):734–749.
Chen YJ, Li HY, Huang CH, et al. Oestrogen-induced epithelial-mesenchymal transition of endometrial epithelial cells contributes to the development of adenomyosis. J Pathol. 2010;222(3):261–270.
Oh SJ, Shin JH, Kim TH, et al. β-Catenin activation contributes to the pathogenesis of adenomyosis through epithelial-mesenchymal transition. J Pathol. 2013;231(2):210–222.
Khan KN, Kitajima M, Hiraki K, Fujishita A, Nakashima M, Masuzaki H. Involvement of hepatocyte growth factor-induced epithelial-mesenchymal transition in human adenomyosis. Biol Reprod. 2015;92(2):35.
Parasuraman P, Mulligan P, Walker JA, et al. Interaction of p190A RhoGAP with eIF3A and other translation preinitiation factors suggests a role in protein biosynthesis. J Biol Chem. 2017;292(7):2679–2689.
Hershey JW. The role of eIF3 and its individual subunits in cancer. Biochim Biophys Acta. 2015;1849(7):792–800.
Silvera D, Formenti SC, Schneider RJ. Translational control in cancer. Nat Rev Cancer. 2010;10(4):254–266.
Hershey JW. Regulation of protein synthesis and the role of eIF3 in cancer. Braz J Med Biol Res. 2010;43(10):920–930.
Gillis LD, Lewis SM. Decreased eIF3e/Int6 expression causes epithelial-to-mesenchymal transition in breast epithelial cells. Oncogene. 2013;32(31):3598–3605.
Chen L, Endler A, Uchida K, et al. Int6/eIF3e silencing promotes functional blood vessel outgrowth and enhances wound healing by upregulating hypoxia-induced factor 2alpha expression. Circulation. 2010;122(9):910–919.
Savagner P. Epithelial-mesenchymal transitions: from cell plasticity to concept elasticity. Curr Top Dev Biol. 2015;112:273–300.
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA. Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Invest. 2009;119(6):1438–1449.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139(5):871–890.
Ding D, Liu X, Duan J, Guo SW. Platelets are an unindicted culprit in the development of endometriosis: clinical and experimental evidence. Hum Reprod. 2015;30(4):812–832.
Wu Q, Ding D, Liu X, Guo SW. Evidence for a hypercoagulable state in women with ovarian endometriomas. Reprod Sci. 2015;22(9):1107–1114.
Zhu B, Chen Y, Shen X, Liu X, Guo SW. Anti-platelet therapy holds promises in treating adenomyosis: experimental evidence. Reprod Biol Endocrinol. 2016;14(1):66.
Siegler AM, Camilien L. Adenomyosis. J Reprod Med. 1994;39(11):841–853.
Valentini AL, Speca S, Gui B, Soglia G, Miccò M, Bonomo L. Adenomyosis: from the sign to the diagnosis. Imaging, diagnostic pitfalls and differential diagnosis: a pictorial review. Radiol Med. 2011;116(8):1267–1287.
Hernandez GC, Bujalil ML, de la Jara DJ, Mier CJ, Bouchan VP. Endometriosis and deficient intake of antioxidants molecules related to peripheral and peritoneal oxidative stress [in Spanish]. Ginecol Obstet Mex. 2006;74(1):20–28.
Wang-Tilz Y, Tilz C, Wang B, Tilz GP, Stefan H. Influence of lamotrigine and topiramate on MDR1 expression in difficult-to-treat temporal lobe epilepsy. Epilepsia. 2006;47(2):233–239.
Zhang Q, Duan J, Liu X, Guo SW. Platelets drive smooth muscle metaplasia and fibrogenesis in endometriosis through epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation. Mol Cell Endocrinol. 2016;428:1–16.
Zhang Q, Duan J, Olson M, Fazleabas A, Guo SW. Cellular changes consistent with epithelial-mesenchymal transition and fibroblast-to-myofibroblast transdifferentiation in the progression of experimental endometriosis in baboons. Reprod Sci. 2016;23(10):1409–1421.
Grund EM, Kagan D, Tran CA, et al. Tumor necrosis factor-alpha regulates inflammatory and mesenchymal responses via mitogen-activated protein kinase, p38, and nuclear factor kappaB in human endometriotic epithelial cells. Mol Pharmacol. 2008;73(5):1394–1404.
Matsuzaki S, Darcha C. Epithelial to mesenchymal transition-like and mesenchymal to epithelial transition-like processes might be involved in the pathogenesis of pelvic endometriosis. Hum Reprod. 2012;27(3):712–721.
Chen W, Ma Q, Suzuki H, Hartman R, Tang J, Zhang JH. Osteopontin reduced hypoxia-ischemia neonatal brain injury by suppression of apoptosis in a rat pup model. Stroke. 2011;42(3):764–769.
Herndon CN, Aghajanova L, Balayan S, et al. Global transcriptome abnormalities of the eutopic endometrium from women with adenomyosis. Reprod Sci. 2016;23(10):1289–1303.
Inagaki N, Ung L, Otani T, Wilkinson D, Lopata A. Uterine cavity matrix metalloproteinases and cytokines in patients with leiomyoma, adenomyosis or endometrial polyp. Eur J Obstet Gynecol Reprod Biol. 2003;111(2):197–203.
Desnoyers G, Frost LD, Courteau L, Wall ML, Lewis SM. Decreased eIF3e expression can mediate epithelial-to-mesenchymal transition through activation of the TGFbeta signaling pathway. Mol Cancer Res. 2015;13(10):1421–1430.
Suo J, Medina D, Herrera S, et al. Int6 reduction activates stromal fibroblasts to enhance transforming activity in breast epithelial cells. Cell Biosci. 2015;5:10.
Leconte M, Chouzenoux S, Nicco C, et al. Role of the CXCL12-CXCR4 axis in the development of deep rectal endometriosis. J Reprod Immunol. 2014;103:45–52.
Kawahara R, Matsuda M, Mori T. Increase in the number of integrinbeta1-immunoreactive monocyte-lineage cells in experimentally-induced adenomyosis in mice. Life Sci. 2003;73(7):907–916.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cai, X., Shen, M., Liu, X. et al. The Possible Role of Eukaryotic Translation Initiation Factor 3 Subunit e (eIF3e) in the Epithelial—Mesenchymal Transition in Adenomyosis. Reprod. Sci. 26, 377–385 (2019). https://doi.org/10.1177/1933719118773490
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719118773490